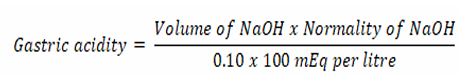Journal of Basic and Applied Pharmaceutical Science Volume 1 (2023), Article ID: JBAPS-101
https://doi.org/10.33790/jbaps1100101Research Article
Study of Anti-Ulcerogenic Effect of Methanol Extract and Fractions of the Leaves of Ocimum Gratissimum (Lamiaceae), on Ethanol-Induced Ulcer in Rats
Okorie Ndidiamaka Hannah1*, Ali Ibeabuchi Jude1, Ugodi Gerald Walter1, Ujam Nonye Treasure2, Okorie Chigozie Peace3 and Atuzu Chibueze Valantine1
1Department of Pharmaceutical Chemistry, Faculty of Pharmaceutical Sciences, Enugu State University of Science and Technology, Enugu State, Nigeria.
2Department of Pharmaceutical Microbiology, Faculty of Pharmaceutical Sciences, Enugu State University of Science and Technology Enugu State, Nigeria.
3Department of Medical Biochemistry, Faculty of Basic Medicinal Sciences. University of Nigeria Nsukka. Enugu State Nigeira.
Corresponding Author Details: Okorie Ndidiamaka Hannah, Department of Pharmaceutical Chemistry, Enugu State University of Science and Technology, Nigeria.
Received date: 10th August, 2023
Accepted date: 11th September, 2023
Published date: 13th September, 2023
Citation: Hannah, O. N., Jude, A. I., Walter, U.G., Treasure, U. N., Peace, O. C., & Valantine, A. C., (2023). Study of Anti Ulcerogenic Effect of Methanol Extract and Fractions of the Leaves of Ocimum Gratissimum (Lamiaceae), on Ethanol Induced Ulcer in Rats. J Basic Appl Pharm Sci, 1(1): 101.
Copyright:©2023, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Peptic ulcer has been a ravaging gastrointestinal disorder in clinical practice. The symptomatic effect is the perforation of the stomach lining. The study was aimed at evaluating the anti-ulcer activity and phytochemical constituents of the leaves of Ocimum gratissimum. The powdered leaves were macerated in 95%v/v of methanol for 72 h, and the dried extract obtained was washed with n-hexane (Hex), ethyl acetate (EtOAc) and butanol (Buta) successively using the solvent partition method to fractionate. Phytochemical constituents were evaluated using validated methods. Acute toxicity was performed using a modified Lorke’s method. Antiulcer studies were conducted using an ethanol-induced ulcer in rat model. The rats received 200, 400 and 20 mg per kg body weight of O.gratissimum extract, its fractions and omeprazole respectively once daily for 14 days. The extraction yielded 3.3 % methanol extract, 16.8 % n-hexane 24.4 % ethyl acetate and 5.2 % butanol fractions. The phytochemical constituents of the extracts are alkaloids, flavonoids, tannins, terpenoids, carbohydrates, steroids and saponin. There was no mortality even at an oral dose of 5000 mg of extract/kg of mouse in the acute toxicity evaluation. The extract and its fractions (200 and 400 mg/kg doses) exhibited dose-dependent ulcer curative. The Hex fraction (400 mg/kg) demonstrated the best anti-ulcer effect with 78.5 % and ulcer index (UI) of 0.60±0.24 when compared to the standard with 50% and 1.40±0.24 respectively. A significant reduction (p< 0.05) in the UI was observed in rats treated with the O. gratissimumextract and its fractions.
Keywords: Ocimum gratissimum, Omeprazole, ethanol-induced ulcer, peptic ulcer, toxicity, wistar rats, anti-ulcer
Introduction
The study of peptic ulcer disease, its management and treatment had been of huge concern in the clinical field. Studies have revealed Helicobacter pylori bacteria and pain-relieving nonsteroidal anti inflammatory drugs (NSAIDs) as the major risk factors in ulcer [1]. Other disease conditions associated with peptic ulcers include severe abdominal pains, weight loss, vomiting, bloating, and heartburn [2-4].
There is an increased search for treatment for this ravaging disease because Pylori infections affect about half of the world’s human population. Due to the considered advantages of products from natural sources, it has become pertinent for researchers to search for the treatment of peptic ulcers from plant sources [5,6]. The effectiveness of the bioactive ingredients found in herbal medicines to act as natural healers and their accessibility, perceived safety, availability and affordability may be responsible for the rising preference for using herbal medicines over conventional medications [7,8].
One of the recently found medicinal plants, Ocimum gratissimum, popularly called smell leaf, has served as potential source of novel medication or as complementary therapy for a variety of illnesses. It is well distributed across Africa, Asia, and South America and is also a member of the Lamiaceae family [9]. It is a perennial herbaceous plant with a high economic value and has a potent aroma. Vital bioactive substances contained in the plant include tannins, flavonoids, phenols, calcium, phosphorous, iron, vitamin A, and more, all of which are essential for human health [10,11]. In traditional medicine, O. graissmum is used pharmacologically to treat a variety of illnesses, including cough, fever, pneumonia, diarrhoea, inflammation, aches, anaemia and bacterial and fungal infections [9]. It is used in the production of foods including fish, soup, meat and stew as a natural flavouring ingredient, condiment, or vegetable. Due to its use in traditional medicine in the management of gastric ulcers and related ailments, this study, therefore, evaluated the qualitative phytochemical constituents and anti-ulcer properties of O. graissmum. The methanol extract and its fractions were used to assay the anti-ulcer activity of O. gratissimum using in-vivo models.
Materials and methods
Materials
Chemicals and reagent
All the reagents used are of analytical standard, and included methanol, n-hexane (GHTECH Guandong Sci-Tech., Co.). ,acetic acid, ethyl acetate (BDH Chemicals., Poole England), mercuric iodide, potassium iodide, hydrochloric acid, chloroform (JHD), ferric chloride, Million’s reagent, alpha naphthol, Benedict’s solution, cupric ions, dilute hydrochloric acid (HCl), ammonia, sodium hydroxide (NaOH), Mayer reagent, sulphuric acid, omeprazole (Ciron drugs and pharmaceuticals Pvt. Ltd,India), Fehling Solution B (BDH Chemicals Ltd., Poole England).
Experimental animals
The guidelines from the National Research Council (NRC) for the care and use of laboratory animals 8th edition were followed while carrying out animal experiments [12]. Healthy mixed sexes of Wistar Albino rats were procured from Animal Farmof the University of Nigeria, Nsukka. The animals were acclimatized to the environmental conditions for 14 days, housed in aluminum cages in softwood shavings as beddings at temperature/humidity of 25 ± 2 °C/24.5-26%, with unrestricted access to pelleted poultry feed and drinking water ad libitum.
Methods
Collection and authentication of plant material
The fresh leaves of O. gratissimum were purchased from Eke Agbani, Enugu, Nigeria in September 2022 and were authenticated by Mr. Patrick Obi, a taxonomist at the Pharmacognosy Department, Enugu State University of Science and Technology, Nigeria. A voucher specimen, ID FP/Cog/17020, was deposited at the herbarium of the Department. The leaves were subjected to air-drying for 21 days at 25 ± 2 °C and the reduced into powder using a mechanical grinder. The fine powder was preserved in a moisture-free air-tight container and used for phytochemical analysis and anti-ulcer evaluation.
Preparation of plant extract
A 750 g of pulverized sample was macerated in 3 liters of methanol (analytical grade) for 72 h and agitated intermittently. The mixture was pre-filtered using a funnel clogged with cotton wool, filtered through a Whatman No. 1 filter paper and the filtrates concentrated under vacuum at a reduced temperature to furnish methanol extract. The dried extract was then stored into an airtight container.
Fractionation of methanol extract
A 25 g of dried methanol extract was dispersed in 150 ml of 10 % aqueous methanol using liquid-liquid fractionation, and 200 ml of the solvents starting with n-hexane, ethyl acetate and butanol respectively were introduced into the dissolved extract. Shaken very well and allowed to separate into two layers. The fraction of each solvent was collected at each point and was concentrated under vacuum at a reduced temperature using a rotary evaporator to obtain a dried fraction. The crude fractions were tested for the presence of secondary metabolites such as alkaloids, steroids, tannins, flavonoids, glycosides, anthraquinone, reducing sugar, carbohydrate, terpenoids and saponins using a standard method [13].
Pharmacological evaluation
Acute toxicity study:
The extract was tested for acute toxicity in mice using the Lorke [14] protocol. The tests were conducted in two phases. The phase one evaluated the toxic range of the extract. The mice were randomly placed in 3 groups (n = 3) and each group received10, 100 and 1000 mg of the extract per kg body weight of mouse. The mice were closely monitored for 24 h for behavioral changes and any death. With no death encountered in phase one, the second phase became inevitable. Since there were no deaths recorded in phase one, three fresh groups of mice; (n=1), were dosed 1600, 2900, and 5000 mg of the extract per kg body weight of mouse. The mice were monitored for signs of acute intoxication or death for 24 h. The median lethal dose (LD50) was calculated using Lorke’s formula [15].
LD50 = √(a×b),
(a = least lethal dose, b = highest safe dose).
Preparation of stock solutions
A 1 g of crude methanol extract, n-hexane and ethyl acetate fractions of O. grattisimum leaves were weighed out into three different glass containers with a stopper. A 10 ml of distilled water was poured into each of the glass bottle containers corked tight and then shaken thoroughly until the extract was completely dissolved in the vehicle. A 20 mg of omeprazole in 5 ml vehicle of stock solution was prepared. The bottle was corked tight, and shaken until the powder was completely dissolved.
Induction of ulcer using ethanol model
Fifty-six adult albino rats of both sexes (150-180 g) were randomly distributed into 8 groups (n = 7) and were kept in 8 different cages labeled as group 1 to group 8. The rats were fasted but allowed free access to water, 24 h before the day of ulcer induction. The rats were orally administered 1 ml of ethanol to induce gastric ulceration. After an hour, two rats were randomly picked from each group and sacrificed to check for gastric ulceration. The gastric juice in the stomach of each rat was emptied into dispensing bottle where the respective pH was taken using the pH meter and then the ulceration in the stomach was observed using a 10x magnifier lens. The rats were treated with the calculated doses of omeprazole (positive control), plant extract and fractions (200 mg and 400 mg) once daily for 14 days.
7 h after the last treatment, all the treated rats were sacrificed by cervical dislocation, and dissected following standard protocol. The gastric juice in the stomach of each rat was emptied into dispensing bottle and its pH was taken using the pH meter.
The grouping and administration of the extract, fractions and control drug are as follows:
Group 1: Negative control; 1 ml ethanol alone
Group 2: Positive control; 1ml ethanol + 20 mg of omeprazole / kg body weight of mouse
Group 3: 1 ml ethanol + 200 mg extract/kg body weight of mouse
Group 4: 1 ml ethanol + 400 mg extract/kg body weight of mouse
Group 5: 1 ml ethanol + 200 mg of Hex fraction per kg body weight of mouse
Group 6: 1 ml ethanol + 400 mg of Hex fraction per kg body weight of mouse
Group 7: 1 ml ethanol + 200 mg of EtOAc fraction per kg body weight of mouse
Group 8: 1 ml ethanol + 400 mg of EtOAc fraction per kg body weight of mouse
Measurement of pH
A 1 ml aliquot of gastric juice was mixed with distilled water (1 ml) and pH of the solution was measured using a pH meter.
Measurement of free acidity
One millilitre of gastric juice was pipetted into a conical flask (100 ml). Three drops of phenolphthalein indicator were added and titrated with 0.25N NaOH until the pink colour disappeared and the colour of the solution turned to yellowish orange.
Free acidity = NAVA= NBVB
Estimation of total gastric acidity
A 1 ml of gastric juice in a conical flask was titrated against 0.10 mol Lֿ-1sodium hydroxide solution using phenolphthalein indicator until the colourless solution changed to light pink. The volume of sodium hydroxide that neutralized the gastric juice was recorded. The total acidity of gastric juice was calculated thus:
Measurement of ulcer score and ulcer index (UI)
The blood clots and the gastric contents of the opened stomachs of the rats were removed by rinsing with distilled water and thereafter examined for the formation of ulcers using a 10x magnification lens. The Ulcer score, Ulcer index (UI) and percentage curative were obtained from the number of ulcers counted [16] using subjective indices of 0 to 5 (Table 1).
Statistical analysis
Data were expressed as mean ± standard error of the mean and statistically evaluated using one-way analysis of variance (ANOVA), followed by a 2-sided Dunnett post hoc comparison test was conducted. A p < 0.05 was considered to be significant.
Results
Yields of O. gratissimum extraction
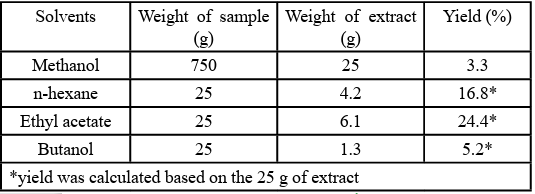
A 750 g of O. gratissimum yielded 25 g of extract. The 25 g of the extract furnished 4.2, 6.1 and 1.3 g of EtOAc, Hex and Buta fractions respectively. The yield of the methanol extract, ethyl acetate and butanol fraction was calculated to be 14.7, 3.0 and 7.8% respectively as seen in Table 2.
Phytochemical analysis of O. gratissimum
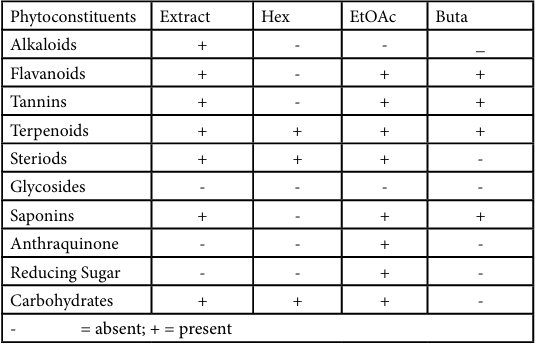
The phytochemical studies on O. gratissimum revealed the presence of alkaloids, flavonoids, tannins, terpenoids, steroids, saponins and carbohydrates as observed in Table 3.
Acute toxicity of O. gratissimum
It is shown in the result below that the methanol extract was not toxic even at a dose of 5000 mg/kg as there was no sign of behavioral changes or physical alteration that led to the death of any of the mice.
Anti-ulcer activity of O. gratissimum
The curative effect of O. gratissimum on the ethanol-induced ulcer model reported in Table 4 shows that the methanol extract and fractions significantly reduced gastric lesions when compared with the positive control.
Results are expressed as mean ± SEM. A significant difference is set at *p<0.05 level represent a significant difference in UI compared with the untreated control. One-way, ANOVA followed by Dunnett with the untreated control. One-way, ANOVA followed by Dunnett
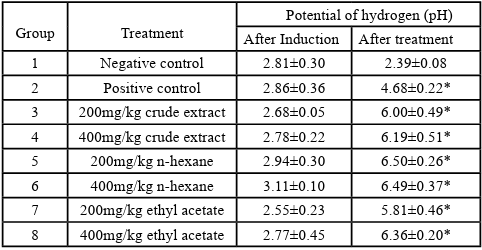
Effect of O. gratissimum on the mean potential of hydrogen
Table 6 below showed an increase in the pH as a result of the treatment with the methanol and fractions of O. gratissimum.
Results are shown as mean ± SEM. A significant difference is set at *p<0.05 level represent a significant difference in the mean potential of hydrogen (pH) when compared with the negative controll. One way, ANOVA followed by Dunnett
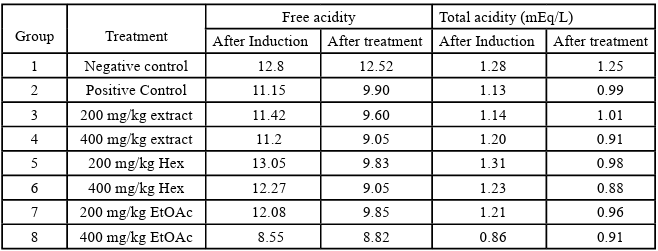
Effect of O. gratissimum on the free and total gastric acidity
Table 7 below shows a decrease in the free and total acidity as a result of the treatment with the extract and fractions of O. gratissimum.
Discussion
One of the ulcer-inducing models in experimental rats is absolute ethanol and it was adopted in this study. The induction normally causes extensive damage of the gastric mucosa and other biological process mediated by free radicals [17]. These events ultimately leads to the destruction of lipid membranes [18-20].
Medicinal plants have shown promising results in handling peptic ulcers from previous studies [3,4,21]. The yields of the extraction and fractionation methods obtained were 3.3 % of the powdered plant material, 16.8, 24.4 and 5.2 % of the methanol extract. The yields evidently showed that there were more non-polar constituents of the plant which could be attributed to volatile oils and other terpenes than polar constituents as shown in Table 2. The low yield of butanol fraction could be attributed to the presence of some volatile compounds [11].
The results of phytochemical screening of O. gratissimum extract and its fractions in Table 3, revealed the presence of tannins, alkaloids, saponins, flavonoids, steroids, carbohydrates and terpenoids. These findings were similar to a previous report by Talabi and Makanjuola [22] where the O. gratissimum water extract revealed the abundance of tannins, flavonoids, saponins, alkaloids, steroids and phlorotannin, glycoside, chalcones, anthraquinone and phenols. Hence, the significant ant-ulcer effect of O. gratissimum may be associated with these metabolites present [23,24].
The acute toxicity study of the extract of O. gratissimum revealed that there was no lethality in the first and second phases as shown in Table 3. This justifies the use of the leaves for cooking different kinds of food in Nigeria and also the treatment of different health conditions [9,18].
The effect of O. gratissimum on the ulceration index of ethanol induced ulcer in rats is shown in Figure 3 The manifestation of ulcer through ethanol induction was inhibited to a very reasonable degree by pretreatment of the experimental animals with omeprazole (the reference drug) and the different doses of O. gratissimum extract and fractions as evident in the reduction of the ulcer index. The results show that after induction of the ulcer, all the rats in all the groups had an ulcer index of 3.00±0.00. The plot shows that after the fourteen day treatment with the different fractions and the extract of the plant, a dose-dependent decrease for both the extract and its fractions was observed in all the treated groups. The same was also observed for the group treated with the standard drug. The results further revealed that the group which received 400 mg/kg body weight of n-hexane fraction had the least ulcer index (0.60±0.24), thus the highest antiulcer effect followed by 400 mg/kg body weight of ethyl acetate and crude extract which obtained a mean of 1.20±0.20 and 1.20±0.37 respectively. This dose-dependent anti-ulcer activity of the extract, ethyl acetate and n-hexane is in agreement with other previous works done on the anti-ulcer property [21,23]. The negative control group however continued to record a high ulcer index by the end of the treatment indicating that the ulcer did not resolve. The result reveals that there was a significant reduction in the mean ulcer index of all the groups treated with the plant extract and the standard drug when compared to the untreated control group that was not treated but induced with ethanol at p<0.05 level of significance.
The higher reduction in UI observed for 400 mg/kg body weight dose of the n-hexane fraction compared with omeprazole 20 mg agreed with a report by Mostofa et al., [25] where the 400 mg/kg extract of Phyllanthus niruri L. leaves elicited a more significant ulcer inhibition than omeprazole.
The result of Figure 3 shows that the group treated with Hex fraction recorded the highest percentage curative effect on gastric erosion (78.6%), followed by the EtOAc (400 mg/kg body weight), extract (57.10%), omeprazole and 200 mg/kg body weight Hex (50.0%), ethyl acetate fraction (200 mg/kg body weight) gave 42.90% and extract 200 mg/kg body weight (35.70%) reduction. Finally, the negative control recorded no cure for gastric erosion. The extract, Hex and EtOAc fractions showing higher anti-ulcer activity when compared with omeprazole is also supported by other previous works [3,4]
Table 6 shows the effect of O. gratissimum on the potential of hydrogen (pH) of induced ulcers in rats. The results show that after induction of the ulcer, all the rats in all the groups had relatively uniform low pH. However, the plot shows that after the fourteen day treatment with the different fractions and the extract of the plant, an increase in the mean pH was observed in all the treated groups. The same was also observed for the group treated with omeprazole. This indicates that the plant can inhibit gastric secretion which consequently protect the stomach environment from ulceration. Meanwhile, the negative control group, however, continued to record low pH by the end of the treatment indicating that the ulcer did not resolve [26].
The efficacy of the O. gratissimum was also evaluated in terms of acidity value (Table 7). The values for the free and total gastric acidity were relatively the same in all the groups’ post-ulcer induction; however, there was a significant reduction after treatment with the doses of O. gratissimum and omeprazole. The untreated control groups were the highest with 12.52N and 1.25 mEq L-1in the other groups. The free acidity for each treated group was lower than the positive control group 9.90N treated with 20 mg/kg omeprazole. The treated groups for the total gastric acidity also had lower values than omeprazole except for the 200 mg/kg body weight crude extract (1.01 mEqLֿ1) with slightly higher values. This is per the report of Lee et al. (2022) where the total gastric acidity of each treatment group treated with an extract of Cinnamomum cassia was lower than the control group.
Conclusion
The study has shown that O. gratissimum demonstrated significant ulcer healing effect on ethanol-induced gastric ulcers. This is evident as there was a comparable antiulcer activity of omeprazole and the extract and its fractions against the ethanol-induced ulcer in rats. Traditionally, O. gratissimum has been useful in the management of gastric ulcers and related ailments. The study validates this ethnomedicinal information and further elucidated the connections between the ulcer healing properties of O. gratissimum and its constituentsecondary metabolites.
Funding
There was no external funding to carry out this research
Acknowledgments
Warm and candid appreciation from the authors to the Faculty of Pharmaceutical Sciences ESUT for their technical support during the research work
Conflicts of Interest:
No conflict of interest as declared by the authors.
References
Anthony, K. (2018). Types of Ulcers. Healthline. Retrieved March 3, 2023, from https://www.healthline.com/health/types of-ulcers.View
Tarasconi, A., Coccolini, F., Biffl, W. L., Tomasoni, M., Ansaloni, L., Picetti, E., and Catena, F. (2020). Perforated and bleeding peptic ulcer: WSES guidelines. World J Emerg Surg.15:1–24.View
Okorie, N. H., Mbah, C. J., Ugodi, G. W., and Magbo, C.C. (2022). Investigation into the Gastroprotective Properties of Alstoniaboonei (De Wild) Leaves and Stem Bark. Trop J Nat Prod Res.; 6(3):403-407.
Okorie, N. H., Ali, I. J., Nnadi, C. O. and Ani, D. I. Anti-ulcer Activity of Crude (2023). Extract and Fractions of Acanthus montanus (Nees) T. Anderson on Indomethacin-Induced Ulcer. Trop J Nat Prod Res.; 7(5):3017-3021View
Al-Sayed, E., Michel, H. E., Khattab, M. A., El-Shazly, M., & Singab, A. N. (2020). Protective role of casuarinin from melaleuca leucadendra against ethanol-induced gastric ulcer in rats. Planta Med.86:32–44.View
Ahmad, M., Aslam, B., Muhammad, F., Mohsin, M., & Raza, A., (2019). Gastroprotective and antioxidant potential of Euphorbia prostrata against aspirin-induced gastric ulcers in rabbits. Pak J Pharm Sci.; 32:7–13.View
Diovu, E. O., Ayoka, T. O., Onah, C. M., and Nnadi, C.O., (2023). Biochemical and histological insights of 1,4-polyisoprene isolated from Sphenocentrum jollyanum Pierre (Menispermaceae) stem in wound healing activity in streptozotocin-induced diabetic rats. J. Ethnopharmacol. 307(11): 6248. View
Ugwuokpe, A. L., Onah, C. M., Nnadi, C. O., and Omeje, E. O., (2022). Evaluation of Antimicrobial and Immunomodulatory Activities of Extract of Beta vulgaris Linn (Chenopodiaceae) Root Tuber. Tropical J.Nat Pro Res. 6(2): 256-259. View
Akara, E. U., Okezie, E., Ude, VC., Uche-Ikonne, C., Eke, G., & Ugbogu, A. E. (2021). Ocimum gratissimum leaf extract ameliorates phenylhydrazine-induced anaemia and toxicity in Wistar rats. Drug Metab. Pers. Ther. 36(4):311-320.View
Venuprasad, M. P., Kumar Kandikattu, H., Razack, S., & Khanum, F. (2014). Phytochemical analysis of Ocimum gratissimum by LC-ESI-MS/MS and its antioxidant and anxiolytic effects. South African Journal of Botany, 92, 151 158. https://doi.org/10.1016/j.sajb.2014.02.010View
Melo, R. S., Azevedo, A. M. A., Pereira, A. M. G., Rocha, R. R., Cavalcante, R. M. B., Matos, M. N. C., Lopes, PHR, Gomes, GA., Rodrigues, TH.S., Dos Santos, H.S., Ponte, I.L., Costa, RA., Brito, Júnior GS, and Carneiro, VA. (2019). Chemical composition and antimicrobial effectiveness of ocimum gratissimum L. Essential oil against multidrug-resistant isolates of Staphylococcus aureus and Escherichia coli. Molecules 3864, 2–17.View
National Research Council (2011). Guide for the care and use of laboratory Animals: Eight Edition. Washington DC. The National Academies Press.View
Harborne, J.B., and Baxter, H. (1992). Phytochemical methods. A guide to modern technique of plant analysis. Chapman and Hill, London. 279-282.View
Lorke, D., (1983). A new approach to practical acute toxicity testing. Arch. Toxicol., 54275-287.View
Abu, D. E., Tim, J. N., Imo-Obasi, P., Ayoka, T. O., & Nnadi, C. O. (2022). In-vivo sub-chronic toxicological evaluation of extract of Vernonia glaberrima leaves in experimental rats. Notulae Scientia Biologicae, 14(2), 11181View
Alok, B., Rajeev, Vivek, D., & Miyaz, A., (2012). Evaluation of anti-ulcer activity of Citrullus lanatus seed extract in Wister albino rats. International J. pharm and Pharm sci, 4(5): 135 139.View
Prasenjit, M., Tanaya, G., and Prasanta, K.M. (2015). Anti peptic ulcer activity of Tea leaves. SMU.Med.J.2, 192-214.View
Ayoka, T. O., & Nnadi CO. (2022). Lesser-known leafy vegetables of Southeastern Nigeria (Vitex doniana and Zanthoxylum zanthoxyloides). Notulae Scientia Biologicae, 14(2), 11777. View
Ayoka, T. O., Nwachukwu, N., Ene, A. C., Igwe, C. U., Nnadi, C. O., (2023). Hepatocurative and Histopathological Evaluations in Albino Rats Exposed to Vitex Doniana Alkaloids. Letters in Appl Nano Biosci. 12(2):56. View
Ekeanyanwu, R. C., Ejiogu, R. N., and Egbogu, M. C., (2016). Lipid peroxidation and non-enzymatic antioxidants status in hypertension in diabetic and non-diabetic patients in Nigeria: a comparative study. Biomed. Res. 27, 250-256View
Okorie, N. H., Ezealisiji, K. M., and Obieze, C. J., (2022). Evaluation of anti-ulcer activity of crude methanol extract of aerial parts of costus afer in ethanol-induced and indomethacin induced ulcer models. J.Pharm and Allied Sci 19 (3); 3722 – 3731View
Talabi, J. Y., and Makanjuola, S. A. (2017). Proximate, phytochemical, and in vitro antimicrobial properties of dried leaves from Ocimum gratissimum. Prev. Nutr. Food Sci. 22 (3), 191–194.View
Jayachitra, C., Jamana, S., Ali, M. A., Paulsamy, S., and Al-Hemaid Fahad, M. A., (2018). Evaluation of traditional medicinal plant, Cissus setosa Roxb. (Vitaceae), for anti-ulcer property. Saudi J. Biol Sci 25 (3); 273-297View
Lozano, E., Barrera, P., Spina, R., and Sosa, M. A., (2016). Terpenoid derivatives as potential trypanocidal agents. Med. Chem.6 (4); 319-321View
Mostofa, R., Ahmed, S., Rahman, S.,Begum, M.M., Riazul, H.T and Amir, H. (2017). Evaluation of Anti-ulcer & Anti inflammatory Activity of Phyllanthus niruri L. leaves in experimental rats. BMC Complement and Altern Med 17:267. View
Ahmed, O. (2022). Evaluation of anti-gastric ulcer activity of aqueous and 80% methanol leaf extracts of Urtica simensis in rats. Metabol Open, 14(10):100172.View