Journal of Case Reports in Cancer Volume 1 (2020), Article ID: JCRC-103
https://doi.org/10.33790/jcrc1100103Review Article
Utility of111In-octreotide for the Characterization of an Extra-axial Lesion with uptake of 18F-choline PET-CT in a Patient with Adenocarcinoma of the Prostate
Ana Álvarez-Gracia2*,Virginia Vallejos Arroyo1,Gabriela Antelo Redondo2,Sergio Lafuente Carrasco1,Anabel Mañes García1,Gloria Moragas Freixa1,Salvador Villà Freixa2
1Hospital Universitario Germans Trias y Pujol. Nuclear Medicine Department. Spain.
2Hospital Universitario Germans Trias y Pujol. Radiation Oncology Department. Institut Català d´Oncologia. Badalona. Spain.
Corresponding Author Details: Ana Álvarez-Gracia, Radiation Oncologist. ICO Badalona, UAB, Spain. E-mail: ana.alvarez@iconcologia.net
Received date: 30th April, 2020
Accepted date: 23th May, 2020
Published date: 26th May, 2020
Citation: Gracia, A.A. (2020). Utility of 111In-octreotide for the characterization of an extra-axial lesion with uptake of 18F-choline PET-CT in a patient with adenocarcinoma of the prostate. J Case Reports Cancer 1(1):103.
Copyright: ©2020, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
We present a case of a 76-year-old male patient diagnosed with prostatic adenocarcinoma that in clinical follow-up an extra-axial brain lesion located in clivus was detected due to a 18F -choline PET scan. However nor MRI or a18F -choline PET-CT could exclude the metastatic origin nor confirm a meningioma lesion.
Our finding suggests the relevance and clinical implication derived from the performance of a complementary brain SPECT-CT with 111 In-octreotide to characterize the extra-axial lesion.
Key words: Prostate Cancer; 111In-octreotide SPECT-CT; 18F-choline PET-CT; Meningioma
Background
Meningiomas account approximately 20% of primary brain tumors and nearly 1-2% are incidentally postmortem findings. Meningiomas are considered hormone-sensitive tumors and express, among others, receptors for prolactin, androgens and estrogens [1]. Consequently, luteinizing hormone-releasing hormone (LHRH) analogues and other hormone treatments, commonly used in patients with prostate cancer, can stimulate their growth and produce neurological symptoms [2]. According this, in the intracranial lesions detected with 18F-Choline Positron Emission Tomography (PET) -Computed Tomography (CT) in the context of prostate cancer, the presence of meningiomas should be taken into account [3].
18F-Choline PET-CT has a high diagnostic accuracy in patient management to detect the early clinical relapse and facilitates the re-staging patients with prostatic cancer [4,5]. The current data published indicates that 18F-choline can be fixed on meningiomas. Also, the choline metabolism rate valued by this radioactive drug has been used to differentiate between typical and atypical meningiomas [3,6]. Therefore, in the context of a biochemical relapse in prostate adenocarcinoma and a single 18F-choline uptake in the brain, the differential diagnosis between a meningioma and a possible brain metastasis may be difficult.
On the other hand, numerous publications have reported overexpression of somatostatin receptors in meningiomas and the usefulness of 111In-octreotide in the non-invasive differential diagnosis of this type of lesions [7]. In addition, there are no conclusive data in the literature about an uptake of 111In-octreotide in brain metastases, except for tumors of neuroendocrine strain or with somatostatin receptors overexpression.
We present a patient with prostate adenocarcinoma in whose evolution presented a biochemical relapse and the finding of 18F-choline uptake in an extra-axial brain lesion located in clivus with no conclusive MRI for metastases. Our findings highlight the relevance and clinical implication derived from the performance of a cerebral SPECT-CT with 111In-octreotide to the lesion characterization.
Case report
We present a case of a 76-year-old male patient diagnosed with intermediate-risk prostate adenocarcinoma in November 2007 (PSA 6.7 ng / ml, Gleason 4 + 3 and T2b by prostate MRI). Radical prostatectomy was performed in January 2008, and pathological analysis resulted on prostate adenocarcinoma Gleason 4 + 3, with perineural permeation and free margins, pT3a. The post-operative PSA was 0.1 ng/mL.
During the follow-up, he presented a biochemical relapse in May 2009 with PSA of 0.6 ng/mL and was considered for Radiotherapy to surgical bed. We administered 70 Gyin 35 fractions of 2 Gy per fraction, ending in August 2009. After Radiotherapy, the patient reached nadir PSA of 0.17 ng/mL in July 2010. The patient continued analytical controls every six months, detecting biochemical progression in April 2013 (PSA of 3.84 ng/mL), without locoregional or distant radiological recurrence. Hormone blockage was initiated with biannual LHRH analog, achieving biochemical response until November 2015 when biochemical progression was detected. He was considered to complete the hormonal blockade by adding an antiandrogen (bicalutamide). In June 2016, resistance to hormonal treatment was evident (PSA 3.6 ng/mL with testosterone at castration levels). Bone scintigraphy was requested, which ruled out bone metastasis. In a subsequent control performed in February 2017 it was a new increase in PSA (7.83 ng/mL), and a 18F-choline PET-CT (Figure 1) was requested. It observed one hypermetabolic focus in the left posterior margin of the clivus (SUVmax 6.3 g/mL), without clear morphological translation on CT-scan. In consequence it was recommended to assess the finding in a Magnetic Resonance Imaging (MRI).
The MRI showed an extra-axial image in the posterior fossa, with a base of meningeal implantation in the left aspect of the clivus, sized 10 x 10 x 20 mm, with intense and homogeneous gadolinium uptake, and was insinuated to the entrance of the internal auditory canal without clear distortion of cranial nerves or evident mass effect on the protuberance. It was concluded that the lesion could correspond to the one mentioned in the PET-CT and radiologically it was consistent with meningioma (Figure 2.).
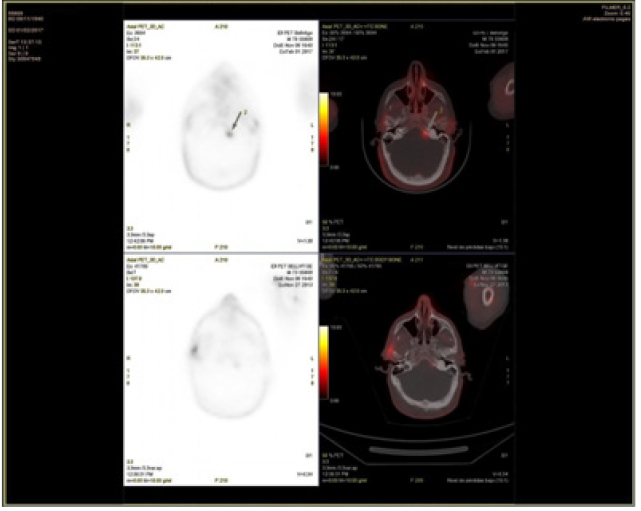
Figure 1. Hypermetabolic focus in left posterior border of clivus (SUVmax: 6,3), not evident in CT-scan, not being able to exclude metastasic disease.
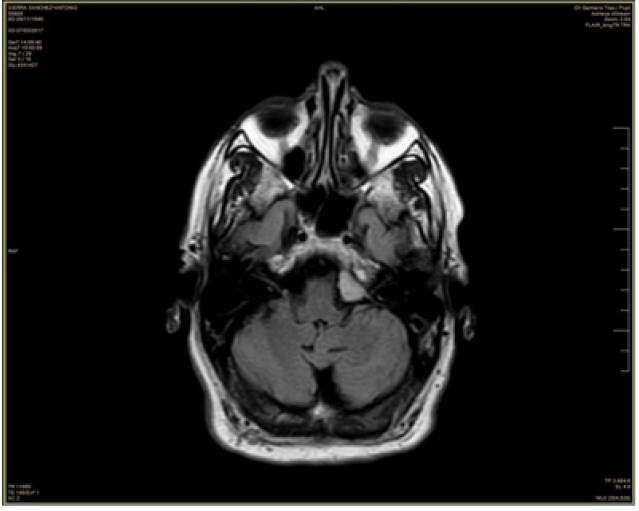
Figure 2. MRI shows an extra-axial image in the posterior fossa, with a meningeal implantation base on the left aspect of the clivus. It shows intense and homogeneous gadolinium uptake.
Given the findings of 18F-choline PET-CT, brain MRI and confirmed biochemical progression resistant to castration, the case was evaluated in the Neuro-Oncology Tumors Committee of our center, which decided to perform a SPECT-CT with 111In-octreotide to be able to clarify the differential diagnosis between a brain metastasis or meningioma in this location.
The cerebral SPECT-CT with somatostatin receptors -6.71 mCi of 111In-octreotide- identified intense focal fixation in clivus, which correlated anatomically with the finding of the brain PET-CT and MRI, suggesting the presence of a lesion with overexpression of somatostatin receptors, compatible with meningioma (Figure 3).
In subsequent MRI controls, extra-axial lesion persists centered on the left border, has not changed its diameters and neither the appearance of edema or other underlying bone infiltration are appreciated.
Both, the iconography and the evolution experienced by the patient supported the diagnosis of meningioma.
Discussion
The incidental uptake of 18F-choline in meningiomas has been reported in approximately 1% of patients with prostate cancer [3]. The disruption of choline metabolism in meningiomas is an evidence from in vivo and in vitro studies. The product derived from the enzyme choline kinase activity is the increase in phosphocholine. The high ratio between phosphocholine and glycerophosphocholine is responsible for cellular transformation and cell immortalization and, in addition, this ratio is higher in atypical meningiomas [8].
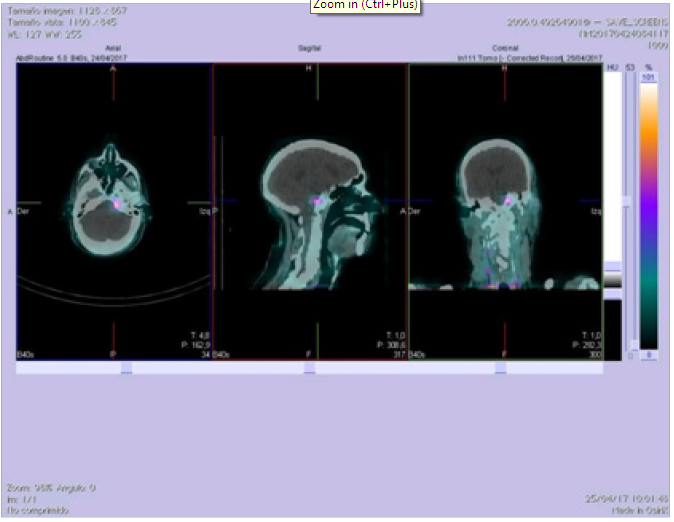
Figure 3. Intense focal fixation of 111In-Octreotide in the posterior fossa extra-axial lesion, which would suggest the presence of a probable lesion with somatostatin receptor overexpression, compatible with the suspicion of meningioma.

Figure 4. In MRI control, extra-axial lesion persisted. Regarding the previous study, its diameters had not changed, and the appearance of edema or other underlying bone involvement was not appreciated. No other extra-axial lesions or other relevant changes with respect to the previous study.
The LHRH analogues and antiandrogenic treatment received by patients with prostate cancer could trigger the growth and symptoms of an unknown meningioma, due to their hormone sensitivity and their greater expression of prolactin, androgens and estrogens’ receptors.
On the other hand, it is increasingly to perform a PET-CT with 18F-choline in patients with biochemical relapse in prostate cancer and, in this context, it is frequent finding incidentaloma in brain that should be characterized. One of the advantages offered by PET-CT with 18F-choline with respect to 18F-FDG is the absence of physiological brain activity, who facilitates the identification of avid intra or extra-axial lesions, including gliomas, medulloblastomas, and meningiomas [3,9,10].
Although MRI findings demonstrate classical semiology for meningioma, in our case the context of biochemical relapse and the presence of a single focus of brain activity in the PET-CT with 18F-choline did not rule out a metastatic lesion. In neuro-oncology committee of our center it was decided to perform a SPECT-CT with 111In-octreotide that allowed to reveal overexpression of somatostatin receptors and characterize the lesion as a meningioma.
Conflict of interests:
The authors declare no conflict of interest.
References
Davis, C. (1990). Meningiomas and sex hormones. Eur J Cancer 26(8):859-60.View
Lee, K.L., & Terris, MK. (2003). Luteinizing hormone-releasing hormone agonists and meningioma: a treatment dilemma. Urology. Aug; 62(2):351.View
Fallanca, F., Giovacchini, G., Picchio, M., Bettinardi, V., Messa, C., & Fazio, F.F. (2009). Incidental detection by [11C]choline PET/CT of meningiomas in prostate cancer patients.Q J Nucl Med Mol Imaging. Aug;53(4):417-21.View
Picchio, M., Messa, C., Landoni, C., Gianolli, L., Sironi, S., Brioschi, M., Matarrese, M., Matei, DV., De Cobelli, F., Del Maschio, A., Rocco, F., Rigatti, P., & Fazio, F. (2003). Value of [11C]choline-positron emission tomography for re-staging prostate cancer: a comparison with [18F]fluorodeoxyglucosepositron emission tomography. J Urol. 169(4):1337-40.View
Krause, B.J., Souvatzoglou, M., Tuncel, M., Herrmann, K., Buck, A.K., Praus, C., Schuster, T., Geinitz, H., Treiber, U., Schwaiger, M. (2008). The detection rate of [11C]choline- PET/CT depends on the serum PSA-value in patients with biochemical recurrence of prostate cancer. Eur J Nucl Med Mol Imaging. Jan; 35(1):18-23.View
Ohtani, T., Kurihara, H., Ishiuchi, S., Saito, N., Oriuchi, N., Inoue, T., & Sasaki, T. (2001). Brain tumour imaging with carbon-11 choline: comparison with FDG PET and gadoliniumenhanced MR imaging. Eur J Nucl Med. 28(11):1664-70.View
Nathoo, N., Ugokwe, K., Chang, A.S., Li, L., Ross, J., Suh, J.H., Vogelbaum, M.A., & Barnett, G.H. (2007). The role of 111indium-octreotide brain scintigraphy in the diagnosis of cranial, dural-based meningiomas. J Neurooncol. 81(2):167-74. Epub 2006 Jul 19.View
Bhakoo, K.K., Williams, S.R., Florian, C.L., Land, H., & Noble, M.D. (1996). Immortalization and transformation are associated with specific alterations in choline metabolism.Cancer Res. 15;56(20):4630-5. View
Welle, C.L., Cullen, E.L., Peller, P.J., Lowe, V.J., Murphy, R.C., Johnson, G.B., & Binkovitz, L.A. (2016). 11C-Choline PET/ CT in Recurrent Prostate Cancer and Non Prostatic Neoplastic Processes. Radiographics. 36(1):279-92.View
Giovacchini, G., Fallanca, F., Landoni, C., Gianolli, L., Picozzi, P., Attuati, L., Terreni, M., Picchio, M., Messa, C., & Fazio, F. (2009). C-11 choline versus F-18 fluorodeoxyglucose for imaging meningiomas: an initial experience. Clin Nucl Med. 34(1):7-10.View