Journal of Case Reports in Cancer Volume 1 (2020), Article ID: JCRC-104
https://doi.org/10.33790/jcrc1100104Review Article
Extraskeletal Osteosarcoma Arising Within A Primary Atrial Myxoma
Ronit Farahmandian*,Dhruvi Panchal*, Marjan Ladan, Hanieh Zham, Daryoush Javidi, Sanford H Barsky**
Cancer Center and Institute for Personalized Medicine, California University of Science and Medicine, 1501 Violet Street, Colton, CA 92324,United States.
Corresponding Author Details: Sanford H Barsky**, Cancer Center and Institute for Personalized Medicine, California University of Science and Medicine, 1501 Violet Street, Colton, CA 92324, United States. E-mail: BarskyS@cusm.org
Received date: 30th April, 2020
Accepted date: 23th May, 2020
Published date: 26th May, 2020
Citation: Farahmandian, R., Panchal, D., Ladan, M., Zham, H., Javidi, D., & Barsky, S.H. (2020). Extraskeletal Osteosarcoma Arising Within A Primary Atrial Myxoma. J Case Reports Cancer 1(1):104.
Copyright: ©2020, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Primary cardiac extraskeletal osteosarcomas (EOS) are very rare neoplasms and those that have been reported previously have been thought to have arisen de novo and their prognosis has been uniformly poor. Complete resection of these tumors usually are very complex and challenging with distant metastases and local recurrences very common despite the use of adjuvant therapies which have been largely ineffective. To date, fewer than 60 cases have been reported in the literature. We report here another case of primary cardiac extraskeletal osteosarcoma but unlike the previously reported cases, this case arose within a preexisting atrial myxoma. Although the progression of atrial myxomas to various sarcomas and other malignancies have been rarely observed and previously reported, this is the first known case of an atrial myxoma progressing specifically to an extraskeletal cardiac osteosarcoma. The endocardial gelatinous tumoral mass consisted of a polypoid fragment composed of 80% atrial myxoma, 15% transitional zone, and 5% extraskeletal osteosarcoma. Areas were recognized as such by routine diagnostic histopathology with confirmatory immunocytochemistry. Unlike the other cardiac extraskeletal osteosarcomas reported in the literature, this case exhibited successful tumor extirpation, no cardiac recurrences, no distant metastases and the patient has been diseasefree for over 3 years. In general, benign neoplasms do not progress into malignant ones and when they rarely do, exhibit the biological behavior of the malignant component. In other words, the malignant component dominates. In this case, the biology of the benign atrial myxoma component dominated and therefore illustrated not only a unique case of cardiac extraskeletal osteosarcoma but also uniquely benign biology as well.
Key words: Endocardial Neoplasms, Cardiac Neoplasms, Benign Transformation to Malignancy
Introduction
Although cardiac tissues have one of the highest concentrations of environmental carcinogen-induced DNA adducts of all tissues [1], the heart is one of the rarest sites of both benign and malignant neoplastic transformation [2]. Although nearly 20% of all cardiac neoplasms are malignant with the majority of these consisting of sarcomas [3], these numbers must not belie the rareness of cardiac neoplasms in general. Although hemangiosarcomas or angiosarcomas are the most common cardiac sarcoma which predominately arise within the right atrium, extraskeletal osteosarcomas usually arise with the left atrium (LA) [3-6]. All cardiac sarcomas comprise a group of aggressive tumors with a very high rate of recurrence and dismal outcome despite surgical resection and oncologic treatments [7]. Early manifestations of cardiac neoplasms including sarcomas are unfortunately nonspecific only standard treatment when feasible is complete surgical resection. No benefits for modalities like chemotherapy and radiotherapy have been proven yet. Extraskeletal cardiac osteosarcomas have been reported previously as single isolated case reports, the vast majority of which exhibited very poor survival [8-35]. Herein we report another case of extraskeletal osteosarcoma but one which uniquely arose within a preexisting atrial myxoma and one which has exhibited both prolonged disease-free and overall survival and probable cure.
Case presentation
A 35-year-old Iranian otherwise healthy man was referred to the hospital with a one month history of palpitation, dyspnea on exertion, and chest discomfort. His vital signs and physical exam were unremarkable. Echocardiogram showed a heterogenous round shape mobile mass measuring 41mm x 28 mm, in the LA originating from the anterior mitral valve and protruding through the mitral valve in diastole (suggestive of either atrial myxoma or papillary fibroelastoma) (Figure 1). Computed Topography Angiography (CTA) of coronary arteries (CA) showed CA-score of 0 and a filling defect in the LA with a 35 mm protrusion to the mitral valve and attachment to the lateral left atrial wall, which also suggested atrial myxoma. A diagnosis of left atrial mass with mild mitral regurgitation and mild tricuspid insufficiency was made. Flow velocity measurements (FVM) showed that the atrial mass was producing obstruction to the flow at the mitral level (Peak Gradient = 22.11 mmHg) (Figure 2). Due to the outflow obstructive nature of the tumor, urgent cardiac surgical intervention was indicated.
The patient was operated through a median sternotomy incision. Upon bicaval cannulation and connection to cardiopulmonary bypass, the left atrium was opened and a large mass with hemorrhagic cysts protruding from a dense pedicle attached to the posterior left atrial wall and posterior mitral leaflet was found. The polypoid mass was largely exophytic without evidence of gross invasion of the underlying endocardium or myocardium. The polypoid mass was totally excised and dissected meticulously through the endocardium and down to the superficial myometrium. Additional endocardial margins were excised to ensure adequate surgical extirpation. After the closure of the atriotomy and deairing, the patient came off bypass: cross clamp 44 min; bypass 70 min; Temp 34° C. The patient had an uneventful recovery and was discharged from the hospital on the 5th postoperative day.
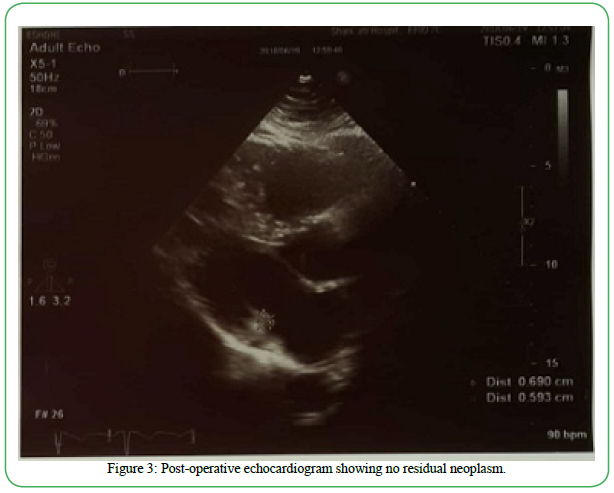
Postoperatively, whole body scintigraphy was performed, which did not detect any evidence of active osteoblastic bony metastasis. Additionally, spiral abdominal and pelvic CT scan with contrast found no abnormalities in the liver, gallbladder, spleen, pancreas,urinary bladder, or kidneys. Spiral thoracic CT scan with contrast found no evidence of mediastinal lymphadenopathy or pleural effusion. It also reported the thoracic aorta, pulmonary artery, and veins to be unremarkable. Moreover, positron emission tomography/x computed tomography (CT) scan using F-18 Fluorodeoxyglucose (FDG) found no abnormal reuptake in head and neck, lung fields, mediastinum, axillary regions, chest wall, breasts, liver, spleen, adrenal glands, pancreas, pelvis, and musculoskeletal system. Chest X-Ray, ultrasound (abdomen, retroperitoneum and pelvic), complete blood count, comprehensive metabolic panel and urinalysis were unremarkable. Post-operative echocardiogram did not find any residual mass (Figure 3).
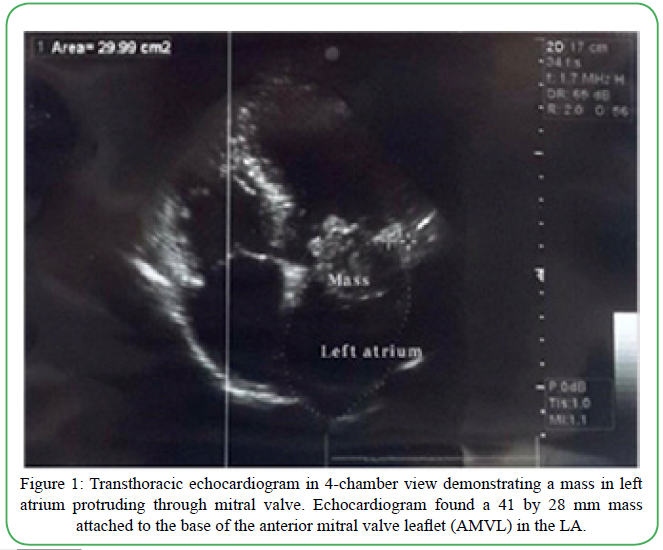
Figure 1. Transthoracic echocardiogram in 4-chamber view demonstrating a mass in left atrium protruding through mitral valve. Echocardiogram found a 41 by 28 mm mass attached to the base of the anterior mitral valve leaflet (AMVL) in the LA.
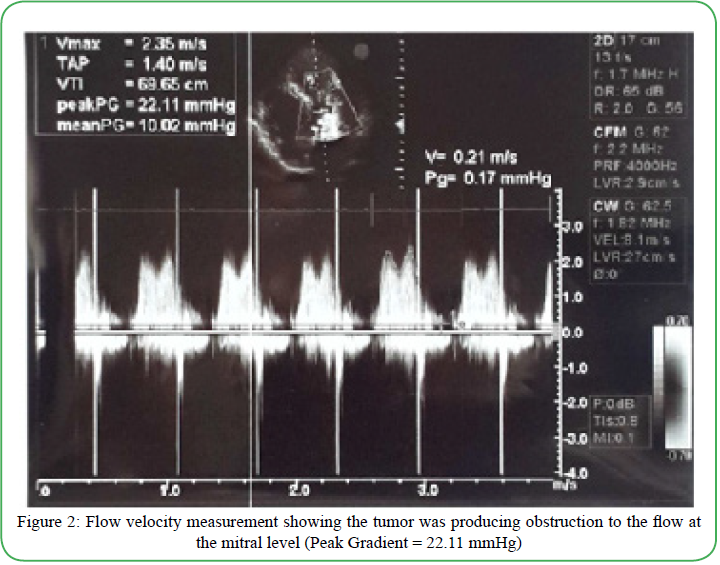
Figure 2. Flow velocity measurement showing the tumor was producing obstruction to the flow at the mitral level (Peak Gradient = 22.11 mmHg)
Materials and Methods
Institutional Review Board (IRB) approval: IRB approval was sought from the California University of Science and Medicine (CUSM). This case was waived from Human Subjects Protection Review with the assurances that all patient information was deidentified by CUSM’s IRB (HS-2020-10).
Histological studies: Paraffin-embedded tissues of the extirpated tissues were processed according to standard protocols involving dehydration, paraffin embedding, 15 micron sectioning and staining with hematoxylin and eosin as well as cutting sections that were left unstained.
Immunocytochemical studies: Sections of the extirpated tissues were then treated by target antigen retrieval solution (DAKO, Carpinteria, CA) in a steamer for 40 min and allowed to cool for 20 min and rinsed in phosphate buffered saline (PBS). After treatment with 0.1% Triton X-100 in PBS for 5 min, tissue sections were incubated with 5% normal donkey serum in PBS for 1 hour and then treated to block endogenous peroxidase. The sections were then incubated with the respective primary antibodies at the recommended dilutions of the manufacturer: polyclonal rabbit anti-CD31, polyclonal rabbit antivimentin, polyclonal rabbit anti-alkaline phosphatase and polyclonal rabbit anti-osteocalcin (Abcam, Cambridge, MA). Tissue sections were then washed three times in PBS for 5 min each and incubated with the appropriate secondary antibodies of DAB-conjugated goat anti-rabbit (Abcam). Chromogen detection was done by a horseradish peroxidase polymer-based detection system counterstained with haematoxylin. For the immunocytochemical studies, the slides were viewed with an Olympus microscope with attached digital camera.
Results
The extirpated mass consisted of a polypoid mass exhibiting cystic spaces, and hemorrhage and protruding from a dense pedicle attached to an underlying layer of the endocardium (Figure 4). The mass measured 38 mm x 30mm x 20mm. Microscopically 80% of the mass consisted of areas of classic atrial myxoma comprised of spindle and stellate shaped cells embedded in a myxoid stroma as well as blood vessels (Figure 5). Underlying normal appearing endocardium was present. The atrial myxoma transitioned into more cellular and less myxoid areas with a higher cell density and with cells gradually enlarging in nuclear size and nuclear/cytoplasmic ratio. These transitional areas comprised approximately 15% of the mass. In adjacent areas the histology transitioned to frank EOS, osteoblastic subtype, with large malignant nuclei embedded in malignant osteoid, having the appearance of “woven bone”. The osteosarcoma areas comprised approximately 5% of the overall mass. The osteosarcoma areas were present in the middle regions of the pedunculated mass well away from the underlying stalk and underlying endocardium. The osteosarcoma areas did not exhibit stalk invasion but were present within and surrounded by areas of atrial myxoma. The diagnostic areas identified on routine hematoxylin and eosin staining were confirmed by supportive immunocytochemistry (Figure 5). Specifically, the areas of atrial myxoma consisting of spindle cells embedded in a myxomatous stroma were vimentin and calretinin positive (data not shown). In addition, vascular endothelial cells illustrating vasoformative structures were observed within the atrial myxoma that were strongly CD31 positive (Figure 5). The regions of osteosarcoma were strongly alkaline phosphatase positive (Figure 5) and osteocalcin positive (data not shown) whereas the adjacent areas of atrial myxoma were completely negative.
Discussion
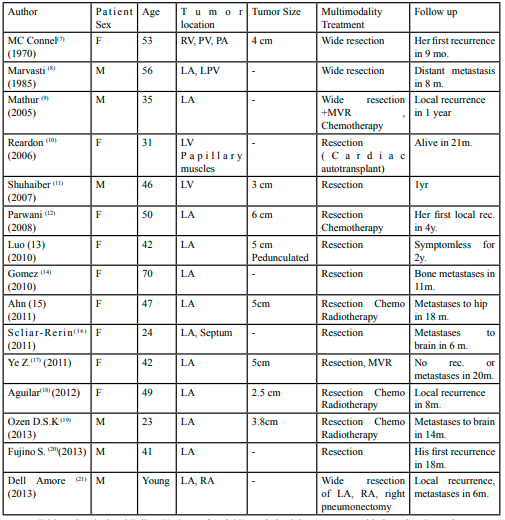
Although cardiac tissues have one of the highest concentrations of environmental carcinogen-induced DNA adducts of all tissues [1], the heart is one of the rarest sites of both benign and malignant neoplastic transformation [2]. Primary cardiac extraskeletal osteosarcomas are very rare neoplasms and those that have been reported previously have been thought to have arisen de novo and their prognosis has been uniformly poor [3-35]. Osteosarcomas most often arise in bone, ie., the skeleton. Nevertheless, rarely osteosarcomas can arise in extraskeletal sites. These unique neoplasms account for less than 2% of all sarcomas and usually arise in the thighs, buttocks, upper extremities, retroperitoneum, and trunk [4]. The heart and specifically the endocardium is the rarest of sites for extraskeletal osteosarcomas [5]. When they occur, they usually involve the left atrium. However, most extraskeletal osteosarcomas are widely infiltrative and invasive lesions that easily gain access to the vasculature and metastasize hematogenously [3-35]. The vast majority of extraskeletal osteosarcomas, whatever site they occur, have a uniformly poor prognosis. This is especially true of extraskeletal osteosarcomas that arise from the endocardium. EOS, like all heart sarcomas, usually occurs in younger patients and has a high histologic grade [3-35]. Symptoms are usually nonspecific such as chest pain, dyspnea, fatigue, and tachyarrhythmia which are related to the obstructive nature of the tumor or invasion into the conductive system of the heart. The standard treatment for all cardiac sarcomas in order to achieve longer survival benefits is complete surgical resection [3,5,22,23]. Chemotherapy and immunotherapy do not appear to improve survival when there is residual or recurrent tumor or metastasis. Although radiotherapy may increase overall survival in non-cardiac extraskeletal osteosarcomas, its role in cardiac sarcomas is not clear [33]. All the reported cases of cardiac endocardial osteosarcomas belong to three common subtypes (osteoblastic, chondroblastic and fibroblastic) [24]. In the literature, fewer than 60 cases of primary cardiac osteosarcomas have been reported [3-35]. We reviewed the vast majority of these cases to get a better handle on their natural history. The detailed records of 42 patients who had undergone surgery for biopsy, debulking or complete resection were available to us. We specifically followed up on 21 patients who had undergone complete tumor resection in the hopes of cure. In 6 patients local recurrences or distant metastases happened as early as 3 months after surgery [23,26-29]. The characteristics of 15 patients with a survival of more than 6 months are summarized (Table 1). In 9 patients the tumor was not resectable and only a biopsy was performed. In 12 patients only a debulking operation could be performed. The overall results of all patients were dismal. No patient survived more than 9 months.
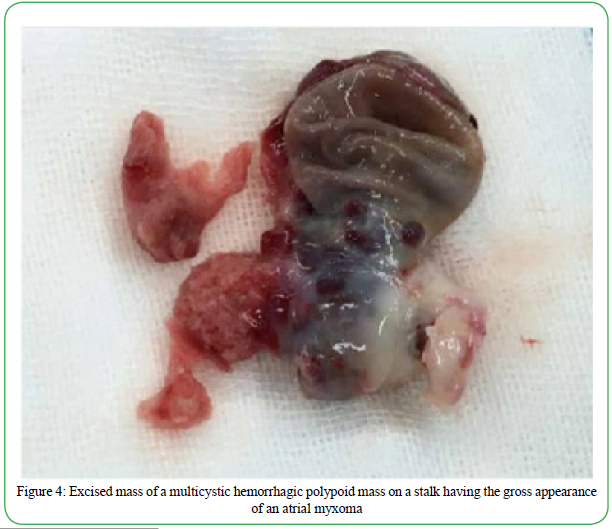
Figure 4. Excised mass of a multicystic hemorrhagic polypoid mass on a stalk having the gross appearance of an atrial myxoma
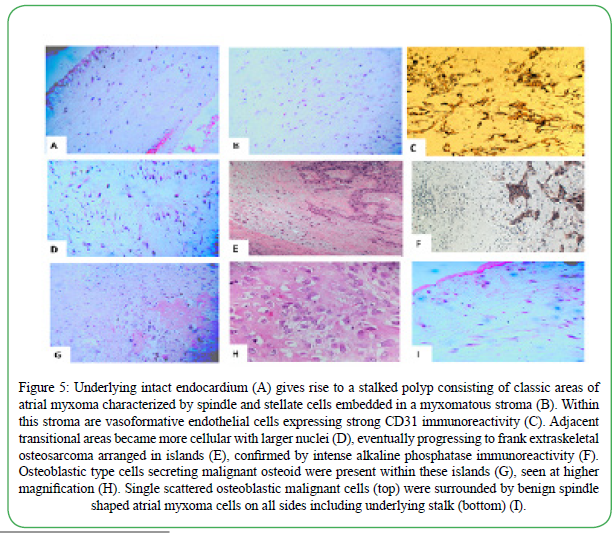
Figure 5. Underlying intact endocardium (A) gives rise to a stalked polyp consisting of classic areas of atrial myxoma characterized by spindle and stellate cells embedded in a myxomatous stroma (B). Within this stroma are vasoformative endothelial cells expressing strong CD31 immunoreactivity (C). Adjacent transitional areas became more cellular with larger nuclei (D), eventually progressing to frank extraskeletal osteosarcoma arranged in islands (E), confirmed by intense alkaline phosphatase immunoreactivity (F). Osteoblastic type cells secreting malignant osteoid were present within these islands (G), seen at higher magnification (H). Single scattered osteoblastic malignant cells (top) were surrounded by benign spindle shaped atrial myxoma cells on all sides including underlying stalk (bottom) (I).
According to Table 1, the median disease-free survival was 16 months (range: 6-48 months). In patients who underwent debulking surgery, the mean overall survival was only 3 months (range: 0-9 months). Cardiac or endocardial osteosarcomas are aggressive tumors with dismal outcomes with rare exceptions [18]. Nowrangi et al reported a unique case of slow-growing primary left ventricle osteosarcoma who survived for 6 years without any resection [30]. Parwani et al reported a left atrial osteosarcoma in a woman who survived for 5 years with multiple surgeries for resection of local recurrences and distant metastases [12]. The overall survival for cardiac sarcomas were considerably shorter than non-cardiac sarcomas (6 months v >93 months (p<0.001) [3,6]. Bakaeen’s results were exceptionally longer than other survival reports [22]. It seems that microscopically negative surgical margins may improve disease-free survival rate slightly. Li et al reported recurrences after a median of 6 months in the margin positive group v 36 months in the margin negative group (p<0.001) [31]. Ramlawi et al, however, did not find any recurrence or survival benefit in the microscopically negative surgical margin group [35]. There are rare case reports of heart transplantation and autotransplantation in local primary cardiac sarcomas without any survival benefits [34,35].
But our patient has survived over 36 months and is currently disease-free and presumably cured. Our patient also differs from the other case reports of cardiac or endocardial EOS because it arose within a preexisting atrial myxoma.
To prove our observations, we conducted detailed histological and immunocytochemical analyses of the extirpated neoplasm. We searched for tumor markers characteristic of both atrial myxomas and osteosarcomas. While no one marker was specific for either, the markers obtained confirmed the diagnoses of both histological areas [37-42]. Furthermore, transitional areas were in evidence showing a progressive increase in cellular density, nuclear size, and atypia with increased nuclear/cytoplasmic ratio. Two markers strongly positive in osteosarcoma but negative in atrial myxoma included osteocalcin and alkaline phosphatase [39,42]. Osteocalcin (OCN) is known to be a bone tissue-specific protein [39]. Alkaline Phosphatase (ALP) is a ubiquitous enzyme present in all tissues but is mainly concentrated in the liver, kidney, placenta, and bone. In the musculoskeletal system, ALP is abundant in osteoblasts and is considered to play a role in the mineralization of newly formed bone. ALP is a sensitive and specific marker in identifying osteosarcoma [42]. In our case, ALP was highly discriminatory in recognizing and distinguishing the areas of osteosarcoma from the areas of atrial myxoma.
Atrial myxomas are nearly always benign but rare case reports have appeared in the literature of progression of benign atrial myxoma to myosarcoma, malignant fibrous histiocytoma, myofibroblastic sarcoma, undifferentiated sarcoma, thymoma, B-cell lymphoma, malignant giant cell tumor and angiofibroliposarcoma [43-52]. Our case is the first reported case of progression of atrial myxoma to extraskeletal osteosarcoma. In nearly all of the previously reported cases of progression of atrial myxoma to malignancy, the patients either recurred locally or distantly metastasized and did not survive [43-52]. Generally, benign neoplasms do not progress into malignant ones and when they rarely do, exhibit the biological behavior of the malignant component. In other words, the malignant component dominates. In our case, the biology of the benign atrial myxoma component seems to have dominated. It could be argued that since the osteosarcoma region was within the surrounding atrial myxoma which comprised the vast majority of the polypoid lesion, that the osteosarcoma could not invade the stalk of the polyp and/or gain access to the vasculature. However, this possible reason remains speculative. We really do not know the reason or mechanism behind the progression of any atrial myxoma to malignancy and certainly do not know the mechanism of our case progressing to EOS. We hope to conduct a study in the near future of a series of cases of atrial myxomas progressing to various malignancies to find common mechanisms and further examine our case against this background. In summary, our case report illustrates not only a unique case of atrial myxoma progressing to an EOS but also a case where the benign biology of the former dominated.
Author Contributions
All authors made an intellectual contribution to the work. Ronit Farahmandian and Dhruvi Panchal equally conducted a critical literature review and interpretation of the data. M Ladan and H Zham recorded critical patient data and wrote drafts of the manuscript. Daryoush Javidi treated and managed the patient and wrote drafts of the manuscript. Sanford H. Barsky interpreted the pathology and conducted the ancillary immunocytochemical studies and wrote portions of the final manuscript which was reviewed by all the authors.
List of Abbreviations
AMVL, anterior mitral valve leaflet; CT, computed tomography; CTA, computed tomography angiography; coronary artery (CA); DAB, 3,3′-diaminobenzidine tetrahydrochloride; EOS, extraskeletal osteosarcoma; FDG, fluorodeoxyglucose; IRB, institutional review board; LA, left atrium; PBS, phosphate buffered saline.
Acknowledgements
The authors wish to thank CUSM Instructional and Informational Technology Services for enabling videoconferencing coauthor communications and Louise Borda for providing operational assistance in the creation of this manuscript.
Conflicts of Interest
The authors declare that they, at the present time, have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. None of the sources of support listed influenced the collection, analysis and interpretation of data, the generation of the hypothesis, the writing of the manuscript or the decision to submit the manuscript for publication.
Funding
The work was supported by the California University of Science and Medicine and the Dr. Carolyn S. Glaubensklee Endowed Cancer Center Directorship.
Conflict of interests:
The authors declare no conflict of interest.
References
DPhillips, D. H. (2002). Smoking-related DNA and protein adducts in human tissues. Carcinogenesis, 23(12), 1979–2004. https://doi.org/10.1093/carcin/23.12.1979View
Singhal, P., Luk, A., Rao, V., & Butany, J. (2014). Molecular basis of cardiac myxomas. International Journal of Molecular Sciences, 15(1), 1315–1337. https://doi.org/10.3390/ ijms15011315View
Hamidi, M., Moody, J. S., Weigel, T. L., & Kozak, K. R. (2010). Primary Cardiac Sarcoma. Annals of Thoracic Surgery. https:// doi.org/10.1016/j.athoracsur.2010.03.065 View
Longhi, A., Bielack, S. S., Grimer, R., Whelan, J., Windhager, R., Leithner, A., Gronchi, A., Biau, D., Jutte, P., Krieg, A. H., Klenke, F. M., Grignani, G., Donati, D. M., Capanna, R., Casanova, J., Gerrand, C., Bisogno, G., Hecker-Nolting, S., De Lisa, M., … Ferrari, S. (2017). Extraskeletal osteosarcoma: A European Musculoskeletal Oncology Society study on 266 patients. European Journal of Cancer, 74, 9–16. https://doi. org/10.1016/j.ejca.2016.12.016View
Zhang, P. J., Brooks, J. S., Goldblum, J. R., Yoder, B., Seethala, R., Pawel, B., Gorman, J. H., Gorman, R. C., Huang, J. H., Acker, M., & Narula, N. (2008). Primary cardiac sarcomas: a clinicopathologic analysis of a series with follow-up information in 17 patients and emphasis on long-term survival. Human Pathology. https://doi.org/10.1016/j.humpath.2008.01.019View
Silverman, N. A. (1980). Primary cardiac tumors. Annals of Surgery. https://doi.org/10.1097/00007611-198210000-00023
McConnell, T. H. (1970). Bony and cartilaginous tumors of the heart and great vessels. Report of an osteosarcoma of the pulmonary artery. Cancer. https://doi.org/10.1002/1097- 0142(197003)25:3<611::AID-CNCR2820250316>3.0.CO;2-EView
Marvasti, M. A., Bove, E. L., Obeid, A. I., Bowser, M. A., & Parker, F. B. (1985). Primary Osteosarcoma of Left Atrium: Complete Surgical Excision. Annals of Thoracic Surgery. https://doi.org/10.1016/S0003-4975(10)60079-3View
Mathur, A., Yadav, R. S., Sharma, A., Khanna, H., & Arsiwala, S. (2005). Osteosarcoma of the left atrium. Indian Journal of Thoracic and Cardiovascular Surgery. https://doi.org/10.1007/ s12055-005-0007-5View
Reardon, M. J., Walkes, J. C. M., DeFelice, C. A., & Wojciechowski, Z. (2006). Cardiac autotransplantation for surgical resection of a primary malignant left ventricular tumor. Texas Heart Institute Journal.View
Shuhaiber, J., Cabrera, J., & Nemeh, H. (2007). Treatment of a case of primary osteosarcoma of the left heart: a case report. The Heart Surgery Forum. https://doi.org/10.1532/HSF98.20061081View
Parwani, A. V., Esposito, N., & Rao, U. N. M. (2008). Primary cardiac osteosarcoma with recurrent episodes and unusual patterns of metastatic spread. Cardiovascular Pathology. https:// doi.org/10.1016/j.carpath.2008.02.003View
Luo, H., Lei, Y., Su, C., Cai, L., Wang, T., Zou, J., & Chen, Z. (2010). Primary cardiac osteosarcoma in a 42-year-old woman. Journal of Cardiothoracic Surgery. https://doi.org/10.1186/1749- 8090-5-120View
Gomez-Rubin, M. C., Rios, J. C., Dobarro, D., Sanchez- Recalde, A., Bret-Zurita, M., Filgueiras, D., Moreno-Yanguela, M., Mate, I., Nistal, M., & Lopez-Sendon, J. (2010). A recidivant primary cardiac osteosarcoma: the role of bone scans. Cardiovascular pathology : the official journal of the Society for Cardiovascular Pathology, 19(1), 55–58. https://doi. org/10.1016/j.carpath.2008.07.005View
Ahn, S., Choi, J. A., Chung, J. H., Choi, H., Chun, E. J., Choi, S. Il, & Kang, H. S. (2011). MR imaging findings of a primary cardiac osteosarcoma and its bone metastasis with histopathologic correlation. Korean Journal of Radiology. https://doi.org/10.3348/kjr.2011.12.1.135View
Scliar-Rerin, J., Targa-Martins, R., Bizarro-Barra, M., Nigri- Oliveira, R., Garcia-Bolze, C. D., & Serafini Osvaldo, A. (2011). Left atrial primary cardiac osteosarcoma. Gaceta Mexicana de Oncologia.
Ye, Z., Shi, H., Peng, T., & Han, A. (2011). Clinical and pathological features of high grade primary cardiac osteosarcoma. Interactive CardioVascular and Thoracic Surgery. https://doi.org/10.1510/icvts.2010.249300View
Aguilar, C. A., Donet, J. A., Galarreta, C. I., & Yabar, A. (2013). A primary cardiac osteosarcoma: Case report and review of the literature. Journal of Cardiology Cases. https://doi. org/10.1016/j.jccase.2012.09.010View
Özen, D. S. K., Öztürk, M. A., Selcukbiricik, F., Esatoglu, S. N., Turna, Z. H., Beyaz, P., Dervisoglu, S., & Özgüroglu, M. (2013). Primary osteosarcoma of the heart: Experience of an unusual case. Case Reports in Oncology. https://doi. org/10.1159/000351123View
Fujino, S., Miyoshi, N., Ohue, M., Noura, S., Hamamoto, S., Oshima, K., Araki, N., Tomita, Y., & Yano, M. (2014). Primary osteosarcoma of the heart with long-term survival: A case report of laparoscopic resection of a metastatic sarcoma in the intestine. Oncology Letters. https://doi.org/10.3892/ol.2014.2405View
Dell’Amore, A., Asadi, N., Caroli, G., Dolci, G., Bini, A., & Stella, F. (2014). Recurrent primary cardiac osteosarcoma: A case report and literature review. General Thoracic and Cardiovascular Surgery. https://doi.org/10.1007/s11748-013- 0236-2View
Bakaeen, F. G., Jaroszewski, D. E., Rice, D. C., Walsh, G. L., Vaporciyan, A. A., Swisher, S. S., . . . Reardon, M. J. (2009). Outcomes after surgical resection of cardiac sarcoma in the multimodality treatment era. The Journal of Thoracic and Cardiovascular Surgery, 137(6), 1454-1460. doi:10.1016/j. jtcvs.2008.11.026View
Burke, A. P., & Virmani, R. (1991). Osteosarcomas of the heart. American Journal of Surgical Pathology. https://doi. org/10.1097/00000478-199103000-00009View
Wang, J., Liu, B., Gao, H., Li, Y., Zhao, P., & Liu, X. (2016). Primary Cardiac Osteosarcoma. Heart, Lung and Circulation, 25(7), 698-704. doi:10.1016/j.hlc.2016.01.006View
Siontis, B. L., Zhao, L., Leja, M., Mchugh, J. B., Shango, M. M., Baker, L. H., . . . Chugh, R. (2019). Primary Cardiac Sarcoma: A Rare, Aggressive Malignancy with a High Propensity for Brain Metastases. Sarcoma, 2019, 1-6. doi:10.1155/2019/1960593View
Dan, M. S., & Hodge, F. A. (1997). Osteogenic Sarcoma of the Left Atrium. The Annals of Thoracic Surgery, 63(6), 1766-1768. doi:10.1016/s0003-4975(97)00130-6View
Jahns, R., Kenn, W., Stolte, M., & Inselmann, G. (1998). A primary osteosarcoma of the heart as a cause of recurrent peripheral arterial emboli. Annals of Oncology, 9(7), 775-778. doi:10.1093/oxfordjournals.annonc.a010995View
Takeuchi, I., Kawaguchi, T., Kimura, Y., Kojima, J., Shimamura, H., Shimizu, N., & Izumi, T. (2007). Primary Cardiac Osteosarcoma in a Young Man with Severe Congestive Heart Failure. Internal Medicine, 46(10), 649-652. doi:10.2169/ internalmedicine.46.6267View
Farhoud, M., & Bakdash, H. (2014). Unique mitral valve mass: Think beyond vegetation. Avicenna Journal of Medicine. https:// doi.org/10.4103/2231-0770.140661View
Nowrangi, S. K., Ammash, N. M., Edwards, W. D., Breen, J. F., & Edmonson, J. H. (2000). Calcified Left Ventricular Mass: Unusual Clinical, Echocardiographic, and Computed Tomographic Findings of Primary Cardiac Osteosarcoma. Mayo Clinic Proceedings, 75(7), 743-747. doi:10.4065/75.7.743View
Li, H., Xu, D., Chen, Z., Ding, W., Hong, T., Chen, H., . . . Wang, C. (2014). Prognostic Analysis for Survival After Resections of Localized Primary Cardiac Sarcomas: A Single-Institution Experience. The Annals of Thoracic Surgery, 97(4), 1379-1385. doi:10.1016/j.athoracsur.2013.12.030View
Liu, J. J., Liu, S., Wang, J. G., Zhu, W., Hua, Y. Q., Sun, W., & Cai, Z. D. (2013). Telangiectatic osteosarcoma: A review of literature. In OncoTargets and Therapy. https://doi.org/10.2147/ OTT.S41351View
Wang, H., Miao, R., Jacobson, A., Harmon, D., Choy, E., Hornicek, F., . . . Chen, Y. E. (2018). Extraskeletal osteosarcoma: A large series treated at a single institution. Rare Tumors, 10, 203636131774965. doi:10.1177/2036361317749651View
Li, H., Yang, S., Chen, H., Yang, Z., Hong, T., Hou, Y., & Wang, C. (2016). Survival after heart transplantation for non-metastatic primary cardiac sarcoma. Journal of Cardiothoracic Surgery, 11(1). doi:10.1186/s13019-016-0540-xView
Ramlawi, B., Al-Jabbari, O., Blau, L. N., Davies, M. G., Bruckner, B. A., Blackmon, S. H., . . . Reardon, M. J. (2014). Autotransplantation for the Resection of Complex Left Heart Tumors. The Annals of Thoracic Surgery, 98(3), 863-868. doi:10.1016/j.athoracsur.2014.04.125View
Terracciano, L. M., Mhawech, P., Suess, K., D’Armiento, M., Lehmann, F. S., Jundt, G., Moch, H., Sauter, G., & Mihatsch, M. J. (2000). Calretinin as a marker for cardiac myxoma: Diagnostic and histogenetic considerations. American Journal of Clinical Pathology, 114(5), 754–759. https://doi.org/10.1309/ NR6G-T872-F090-LBRWView
Song, X., Liu, D., Cui, J., Zhou, M., Wang, H., Liu, N., Qi, X., & Li, Z. (2016). Identification of Stem-Like Cells in Atrial Myxoma by Markers CD44, CD19, and CD45. Stem Cells International, 2016. https://doi.org/10.1155/2016/2059584View
Evola, F. R., Costarella, L., Pavone, V., Caff, G., Cannavò, L., Sessa, A., Avondo, S., & Sessa, G. (2017). Biomarkers of osteosarcoma, chondrosarcoma, and ewing sarcoma. Frontiers in Pharmacology, 8(APR), 1–14. https://doi.org/10.3389/ fphar.2017.00150View
Fanburg-Smith, J. C., Bratthauer, G. L., & Miettinen, M. (1999). Osteocalcin and osteonectin immunoreactivity in extraskeletal osteosarcoma: A study of 28 cases. Human Pathology, 30(1), 32–38. https://doi.org/10.1016/S0046-8177(99)90297-6View
Wu, D., Chen, K., Bai, Y., Zhu, X., Chen, Z., Wang, C., Zhao, Y., & Li, M. (2014). Screening of diagnostic markers for osteosarcoma. Molecular Medicine Reports, 10(5), 2415–2420. https://doi.org/10.3892/mmr.2014.2546View
Schulz, A., Jundt, G., Berghauser, K. H., Gehron-Robey, P., & Termine, J. D. (1988). Immunohistochemical study of osteonectin in various types of osteosarcoma. American Journal of Pathology, 132(2), 233–238.View
Mavrogenis, A., & Papagelopoulos, P. (2014). Soft Tissue Tumors: Extraskeletal osteosarcoma. Atlas of Genetics and Cytogenetics in Oncology and Haematology, 18(6), 443–446. https://doi.org/10.4267/2042/53774View
Kasugai, T., Sakurai, M., Yutani, C., Hirota, S., Waki, N., Adachi, S., & Kitamura, Y. (1990). Sequential MaIignant Transformation of Cardiac Myxoma. Pathology International, 40(9), 687-692. doi:10.1111/j.1440-1827.1990.tb01617.xView
Lian, D., Zhiyun, X., & Zhigang, L. (2010). Relapse of a cardiac tumor from myxoma to low-grade malignant myofibroblastic sarcoma: Malignant transformation or metachronous lesion? Journal of Medical Colleges of PLA, 25(1), 50–53. https://doi. org/10.1016/S1000-1948(10)60017-2View
Todo, T., Usui, M., & Nagashima, K. (1992). Cerebral metastasis of malignant cardiac myxoma. Surgical Neurology, 37(5), 374–379.View
Park, C. K., Cho, Y. A., Kim, M., & Shim, H. S. (2018). Malignant lymphoma arising in cardiac myxoma, presenting with peripheral arterial emboli. Cardiovascular Pathology, 32, 26–29. https://doi.org/10.1016/j.carpath.2017.10.001View
Kusumi, T., Minakawa, M., Fukui, K., Saito, S., Ohashi, M., Sato, F., Fukuda, I., & Kijima, H. (2009). Cardiac tumor comprising two components including typical myxoma and atypical hypercellularity suggesting a malignant change. Cardiovascular Pathology, 18(6), 369–374. https://doi. org/10.1016/j.carpath.2008.05.002View
Miller DV, Tazelaar HD, Handy JR, Young DA, Hernandez JC. Thymoma arising within cardiac myxoma. Am J Surg Pathol. 2005;29(9):1208‐1213. doi:10.1097/01. pas.0000158398.65190.7bView
Sakovich VA, Grinshteĭn IuI, Vershinin IV. [Malignant transformation of left atrial myxoma] Klinicheskaia Meditsina. 2004 ;82(2):65-67.View
Eckhardt, B.P., Dommann-Scherrer, C.C., Stuckmann, G. et al. Giant cardiac myxoma with malignant transformed glandular structures. Eur Radiol 13, 2099–2102 (2003). https://doi. org/10.1007/s00330-002-1663-0View
Nguyen, Anita, and Wael I. Awad. “Cardiac Sarcoma Arising From Malignant Transformation of a Preexisting Atrial Myxoma.” The Annals of Thoracic Surgery, vol. 101, no. 4, 2016, pp. 1571–1573., doi:10.1016/j.athoracsur.2015.05.129.View
Seo IS, Warner TF, Colyer RA, Winkler RF. Metastasizing atrial myxoma. Am J Surg Pathol. 1980;4(4):391‐399. doi:10.1097/00000478-198008000-00008View