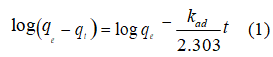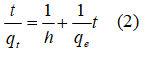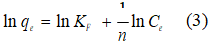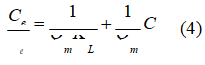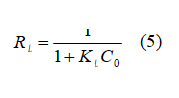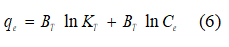Journal of Environmental Engineering and Critical Challenges Volume 1 (2022), Article ID: JEECC-101
https://doi.org/10.33790/jeecc1100101Research Article
Assessment of Diclofenac Adsorption into Activated Sludge: Mechanism and Thermodynamic
Luana Dandara Barreto Torres1, Mirieli dos Santos2, Silmara Silva Queiróz3, Lucia Helena Gomes Coelho4
1MSc.,Student Environmental Science & Technology post-graduation course, Center of Engineering, Modelling and Applied Social Sciences, Federal University of ABC, Address: Avenida dos Estados, 5001, Santo André, SP,
2MSc. Environmental Science & Technology post-graduation course, Center of Engineering, Modelling and Applied Social Sciences, Federal University of ABC, Address: Avenida dos Estados, 5001, Santo André, SP, 09210-580, Brazil. ORCID: 0000-0002-9942-7180.
3Colaborator, Environmental Science & Technology post-graduation course, Center of Engineering, Modelling and 14 Applied Social Sciences, Federal University of ABC, Address: Avenida dos Estados, 5001, Santo André, SP, 09210-580, Brazil. ORCID: 0000-0002-9942-7180.
4*Ph.D., Professor, Environmental Science & Technology post-graduation course, Center of Engineering, Modelling and Applied Social Sciences, Federal University of ABC, Address: Avenida dos Estados, 5001, Santo André, SP, 09210-580, Brazil
Corresponding Author: Lucia Helena Gomes Coelho, Ph.D., Professor, Environmental Science & Technology post-graduation course, Center of Engineering, Modelling and Applied Social Sciences, Federal University of ABC, Address: Avenida dos Estados, 5001, Santo André, SP, 09210-580, Brazil
Received date: 03rd October, 2022
Accepted date: 22nd October, 2022
Published date: 24th October, 2022
Citation: Torres, L.D.B., Santos, M., Queiróz, S.S., Coelho,L.H.G. (2022). Assessment of Diclofenac Adsorption into Activated Sludge: Mechanism and Thermodynamic. J Envir Engin Criti Challen, 1(1): 101.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
This study aimed at understanding and characterizing the sorption process of Diclofenac (DCF) onto activated sludge under conditions similar to those of a conventional Wastewater Treatment Plant (WWTP). Two experiments were performed: kinetics test, composed of six identical pairs of control/sample with the same DCF concentration stirred under constant rotation for different intervals; and an adsorption thermodynamics test with one pair of control samples and six duplicate samples with DCF concentrations from 5 - 100 mg L-1 stirred under constant rotation for the same period. DCF concentrations in the supernatant were measured in time spectrophotometrically at 282 nm. The adsorption of diclofenac onto the activated sludge reached its equilibrium after approximately 2 hours. The results showed that the process was best described by the Freundlich and Langmuir isotherm models, which suggest that the adsorption of DCF onto the activated sludge is a non-favorable second order reaction with a monolayer coverage. The Temkin isotherm model suggests that the heat of adsorption of the DCF molecules in the layer decreases linearly as a result of the increased surface coverage and follows a chemisorption mechanism. The amount of DCF adsorbed onto the sludge particles varied between 17 and 44%, with the adsorption ratio of DCF per gram of sludge being considerably low (< 1 mg per g of sludge). This might be a result of the longer hydraulic retention times (HRT) adopted, considering that DCF may be toxic to the sludge microbiota, interfering with DCF removal from the supernatant. Thus, improving diclofenac removal by aerobic WWTP might require an adjustment of HRT to enhance sorption onto the sludge and reduce its impact on bacterial community.
Keywords: Activated Sludge, Adsorption, Diclofenac, Freundlich Isotherm, Langmuir Isotherm, Tamkin Isotherm
Introduction
The presence of micro contaminants in the environment has attracted the attention of the scientific community over the past years due to its harmful effects on living beings, resulting in aquatic toxicity, genotoxicity, endocrine disruption in wild animals, and selection of resistant pathogen bacteria [1]. These contaminants include drugs of different categories, such as analgesics, antibiotics, lipidic regulators, synthetic hormones, anti-inflammatories and others [2].
Pharmaceutical products have been found in different environmental compartments worldwide, including surface and groundwaters, soil, sediment, wastewater, and tap water [3-6]. The bioactivity of pharmaceutical products, even in low concentrations, can result in an increase in their bioaccumulation and ecotoxicity in biota and induce adverse effects in human beings due to unaware re-ingestion [7-10].
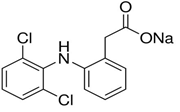
One of the main sources of pharmaceuticals in the environment is municipal wastewater discharge containing drug residues not completely removed by conventional wastewater treatment processes [5]. Other contributing sources are industries, farms, hospitals, and healthcare facilities [11]. One of the most frequently detected pharmaceuticals in waters and urban wastes is diclofenac (DCF), also named 2-[(2,6-dichlorophenyl)amino]benzene acetic acid (Figure 1), used mostly in the forms of sodium and potassium salts or methyl ester. DCF is the most abundant of the active compounds found in water, being identified in samples collected before and after treatment in Wastewater Treatment Plants (WWTP) [12,13].
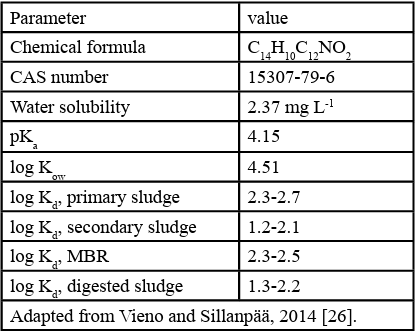
According to Lonappan et al. (2018), DCF requires special attention due to its acute toxicity, extensive use in the treatment of several symptoms, ease of purchase and no prescription required in many countries [14]. The physical chemical properties of DCF are presented in Table 1.
DCF’s annual consumption is 1443 ± 58 tons worldwide [15] and was one of the compounds included in the first list limited by the European Union (EU) through Directive 2015/495 to achieve their associated risks as well as determine mitigation measures [16-18]. After an analysis of 6698 samples from 25 EU countries during 3 years, DCF was identified in waterbodies in concentrations ranging between 27 and 47 ng L-1. Although DCF was removed from the EU’s watch list for substances of emerging concern in May 2018 [19], it remains a monitored compound in within the member countries. However, in a different scenario such as in developing countries, monitoring of emerging pollutants is still insufficient and underestimated by public authorities [20], which reinforces the importance of studies aiming at quantifying and removing DCF from natural and residual waters.
DCF removal in WWTP varies significantly between 5% and 81%, depending on the type of biological treatment and drug concentration in the affluent [14,21-25]. In conventional activated sludge treatment plants, this rate may be lower due to the inefficiency of biological processes in the degradation of DCFpresent in wastewaters [26]. This ineffectiveness may lead to the ubiquitous presence of DCF in surface water, causing ecotoxic effects in many aquatic and terrestrial organisms [27-29].
Diclofenac is classified as a recalcitrant compound (kbio < 0.1 L kg-1 SS d-1) when considering a first order decay constant in WWTPs [30]. Quintana et al. (2005) [31] investigated the biodegradation of diclofenac by activated sludge and found no transformation of diclofenac over 28 days even when diclofenac was the only source of carbon. The poor biodegradation of diclofenac was confirmed by Kimura et al. (2007) [32] in batch elimination experiments using sludge from WWTPs and MBR. Thus, like other recalcitrant compounds, DCF removal efficiencies above 40% were obtained for systems operating under several days of hydraulic retention times (HRT) and longer or infinite solids retention times (SRT) [33.34]. Compounds with hydrophobic aromatic groups such as DCF had been reported as being easily adsorbed by microorganism [35]; also, hydrophobicity may enhance sludge adsorption, thus requiring longer HRT for biological degradation efficiency [33,[36].
In a conventional WWTP, the pollutants (adsorbates) can be removed from the affluent by adsorption into the activated sludge (adsorbent) followed by biodegradation process [7]. Adsorption in biological wastewater bulk is a complex process that depends on the physical-chemical properties of the adsorbate (such as molecular charge and hydrophobicity) as well as on the properties of the adsorbent (superficial charge, specific surface area, oxidation state, and organic matter content of the sludge or biofilm) [36,37].
The activated sludge consists of a complex consortium of bacteria, protozoans, fungi, and micro metazoans that consume organic and inorganic compounds present in the influent for metabolic processing. Each type of sludge (primary, secondary or biofilm) shows a different behaviour regarding its interaction with pollutants [1]. The electrostatic interaction between sludge and pollutants is remarkably influenced by the pH media, once its slight variation can either protonate (positive or neutral charge) or deprotonate (negative or neutral charge) the molecules of pollutants, therefore driving the nature of its interaction with the sludge particles [14,39].
Nevertheless, studies evaluating the main removal mechanisms are still limited and a better understanding of the kinetics and thermodynamic aspects of the DFC sorption process in sludge can assist in comprehending and drafting solutions for the removal of this compound from wastewater through biological processes. Therefore, this study aims at understanding the sorption kinetics and thermodynamics of DCF onto activated sludge through data adjustment to adsorption models, with the intent to help optimize the conventional treatment configuration for the removal of DCF from wastewater. The hypothesis of the present work is that DCF is retained in aerobic biological treatment systems by sorption onto the sludge, such that the saturation of the active sites present on the sludge surface may limit the treatment efficiency in terms of DCF removal.
Methods
Chemicals
Diclofenac (2-[(2,6-Dichlorophenyl)amino]benzeneacetic) sodium salt purchased from Sigma-Aldrich, purity ≥ 98.5% (HPLC grade) was used in all experiments. Activated sludge provided by the Wastewater Treatment Plant Parque Andreense (Santo André/ SP - Brazil) was used as substrate for all experiments. All standard solutions and the synthetic effluent used to feed the sludge were prepared with ultrapure (Mili-Q) water. Methanol 99,98% (HPLC grade) was used for the preparation of 150 mg L -1 diclofenac stock solution 1:5 methanol:water.
Sludge characterization
The activated sludge was acclimated in a pilot batch bioreactor for approximately 1 week prior to use, kept under 4 mL s-1 air flowrate and fed with 250 mL of a synthetic effluent on a daily basis, whose composition is detailed in the work of Matsubara et al. [34]. The sludge was characterized by total suspended solids (TSS), total fixed solids (TFS) and suspended volatile solids (SVS) [40]. All analyses were performed in duplicate.
DCF spectrophotometric analysis
A spectrophotometer model NOVA 1800UV was used to track the changes in DCF concentration over time, through the readings of absorbance of UV light at 282 nm - optimum absorption peak for DCF in methanol 20% v/v [42]. A 5-cm quartz cuvette was used for all readings. A calibration curve was built with standard solutions at DCF concentration range from 2 to 30 mg L-1.
Adsorption kinetic and isotherms
DCF content sorbed onto the sludge was calculated by the difference between the initial and equilibrium mass concentrations found through the absorbance readings. The mass of adsorbate in each flask was calculated using the concentration of TSS, considering the different volumes of sludge added to each beaker.
For the kinetics evaluation, glass flasks were used: half of them filled with 150 mL of activated sludge for control assay and the other half filled with 15 mL of DCF solution added to 135 mL of activated sludge, as to have a final concentration of 15 mg L-1 in each flask. The beakers were submitted to agitation on an orbital shaker at room temperature (25 ± 1) ºC under 150 rpm. At set interval times (10, 20, 30, 40, 60, and 285 minutes), a pair of assays (control and DCF spiked sample) were removed from shaking for the readings.
Prior to the readings, all sample aliquots (control and spiked solutions) were centrifuged at 700 rpm for 5 minutes and filtered through 0勀Ṍ勀 µm fiberglass filters. All beakers were time-monitored for pH, Dissolved Oxygen (DO) and temperature variation. DO was monitored with a multiparametric probe (Hach, model HQD) and pH and temperature were monitored with a standard benchtop pH meter (Micronal).
To investigate the controlling mechanism of adsorption processes such as mass transfer and chemical reactions, pseudo-first-order and pseudo-second-order kinetic models were applied in an attempt to model the kinetics of DCF adsorption onto the activated sludge. The pseudo-first-order rate equation is given by:
where qe is the equilibrium adsorption capacity (mg g-1), qt is the adsorption capacity at time t (mg g-1), and kad (min−1) is the pseudo first order rate constant of the adsorption. The rate constant, kad and correlation coefficients for different concentrations of DCF were calculated from the linear plots of log (qe− qt) versus t. The pseudo second-order model is expressed by the following equation [43]:
where h (mg g-1 min-1) can be regarded as the initial adsorption rate and k is the pseudo-second-order rate constant of adsorption (g mg-1 min-1). A plot of t/qt versus t should yield a straight line if pseudo second-order kinetics is applicable and qe and h can be determined from the slope and intercept of the plot, respectively.
For isotherms evaluation, 14 flasks were set up: 2 for control and 6 duplicates filed with 150 mL of the mixture sludge + DCF, stirred on an orbital shaker at 150 rpm. Each flask received a different volume of DCF to keep the concentrations within the following range: 2, 5, 10, 15, 25, and 50 mg L-1. All the systems were left stirring for 2 hours. Aliquots of 20 mL of each sample were centrifuged, filtered, and analyzed spectrophotometrically.
To evaluate data adjustment to the Freundlich, Langmuir, and Temkin isotherm models, the variables qe(mass of DCF adsorbed in the equilibrium) and Ce (concentration of DCF in the equilibrium) were linearized as follows, with equation 3 corresponding to the Freundlich model:
where KF is the distribution coefficient (mg g-1) and n is an empirical parameter that indicates process favoring ( 1 ≤ n ≤ 10 indicates a favorable adsorption for the Freundlich model).
Equation 4 corresponds to the Langmuir model and its favourability is given by the Separation Factor (RL) in equation 5
where C0 is the initial concentration (mg L-1), Qm is the maximum adsorption capacity to form a single layer and is related to the energy of adsorption (L mg-1); and KL is the Langmuir constant, which indicates the theoretical adsorption capacity onto the mono-layer (L g-1 c).
Equation 6 corresponds to the Temkim model, which considers the effects of indirect adsorbate/adsorbate interactions on the adsorption process:
For Temkim’s model, BT is a constant which is related to the heat of sorption (J mol-1) and KT is the isotherm constant (L g-1).
After plotting the data, the linear adjustment was applied using Microcal Origin 8.0 software to obtain the equation of each curve and the coefficients of determination (R2).
Results and Discussion
DCF adsorption kinetic
Adsorption of DCF in natural sludge showed no statistical difference with sterilized, with only 5% greater removal after 4 hours of reaction, indicating low biodegradation contribution. Microbial degradation of diclofenac by activated sludge microbiota does not lead to its complete elimination but produces other metabolites and transformation products, among them 1-(2,6-dichlorophenyl)-1,3 dihydro-2H-indol-2-one and 2,3-dichloro-N-(phenyl)aniline, which are also considered such as pollutants [41].
The activated sludge configuration is the most used in WWTP for biological treatment; however, the results obtained indicate that and the rate of elimination of diclofenac by this process is little efficient, not exceeding 50 % [44-46], with only a small portion of complete mineralization (21.8 - 28.5%) [47]. However, experimental conditions such as temperature, pH, stirring and sludge concentration can be adapted to achieve more satisfactory results.
Fan et al. (2014) [41]evaluated the biodegradability of the diclofenac on two 2 types of sludge; sterilized and activated. Results of batch adsorption experiments showed that contributions of sludge adsorption and biodegradation after 6 hours were 14.9% and 4.8%, respectively. The authors suggested that this difference in removal efficiency by sterilized and activated sludge is not related to biodegradation process; rather, it may be due to the sterilizing effect on the activated sludge, which can be caused changes in the characteristics of the substrates and might contribute to the removal of diclofenac by adsorption. Similar results were obtained in this study with higher dispersion of the flocks in the sterile sludge, which could increase the contact area and, consequently, the number of active sites for DCF adsorption. Thereby, Fan’s study indicates that the activated sludge may be able to adsorb and transform the pollutant although very long reaction times may be required, which can be a hinderance for WWTPs [41].
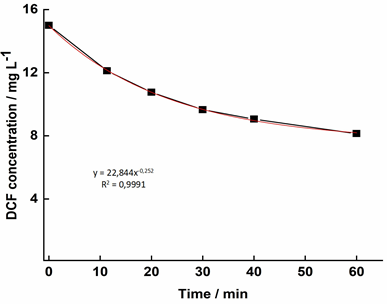
In this study, DCF concentration decayed faster along the first 40 minutes, reaching the equilibrium after two hours (Figure 2). This indicated that the adsorption kinetics of diclofenac on activated sludge follows the pseudo first-ordermodel, with a 0.99% correlation coefficient (R2). The tendency observed in the adsorption kinetics curve implies that equilibrium is reached before 2 hours. For this reason, this was the time set to the adsorption thermodynamics test. Kabak and Basibuyuk (2012) [7] adopted 160 minutes for this test, reaching equilibrium after only 20 min. One possible explanation for such rapid saturation is that the authors used 40 mg L-1 as the initial DCF concentration, as well as smaller total volatile solids (TVS) in activated sludge (3.000 mg L-1 whereas approximately 8.400 mg L-1 were used in the present study). Therefore, a higher ratio DCF/sludge might have accelerated the process.
It may be worth mentioning that an assay submitted to 4h of shaking showed an abnormally higher absorbance reading, corresponding to a concentration close to that of the first sample (around 10 mg L-1). This was the opposite of what was expected, since it was assumed that the concentration of DCF in the supernatant would decay with time. To confront this finding, a solids test was performed after 2h and 4h of shaking, under the hypothesis that part of the solids may have dissolved in the supernatant, causing the absorbance readings to shoot up after a longer shaking period. The hypothesis was confirmed, given that, after 2h and 4h of shaking, the suspended solids content decayed by 7.5 and 12%, respectively. However, even after correcting the value of qe for this point in the curve, the redissolution was not enough 2 to explain why the absorbance value was so high. Another hypothesis is that other organic compounds adsorbed to the sludge may have solubilized into the bulk interfering with the readings and causing an phenomenom otherwise known as matrix effect.
No time variation was observed for DO or pH (Figure 3a and 3b, respectively). The pH values around 8 were similar to other studies with DCF and sewage sludge [7,48]. Regarding DO measurements, Figure 3b shows that the oxygen concentration is lower in the samples with DCF, indicating that the concentration applied (15 mg L-1) may present some toxicity to the microbiota in the sludge. Kabak and Basibuyuk (2012) [7] found that DCF started showing toxicity to the bacteria in the sludge above 10 mg L-1. Similarly, concentrations higher than 50 mg L-1 presented toxic effect with an inhibition of microorganisms’ activity in the activated sludge [48].
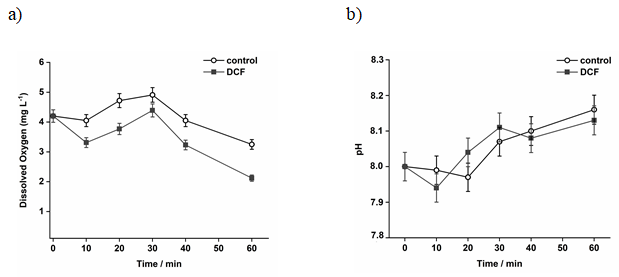
Figure 3: Parameters variation with time for control system and DCF 15 mg a) Dissolved oxygen (mg L-1); b) pH.
DCF Adsorption Isotherms
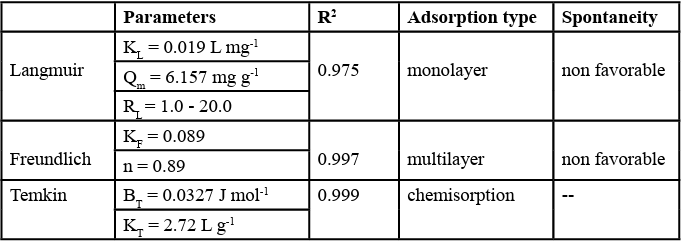
The parameters presented in Table 2 show that the Freundlich’s isotherm provided better adjustment to the experimental data than the Langmuir’s isotherm, with an R-squared (R2) of 0,997, meaning that the Freundlich’s model can explain about 99,7% of the thermodynamic behaviour of the system. This suggests that the adsorption is likely to happen on multilayers, assuming that the adsorbent (sludge) has a heterogeneous composition, with active sites distributed exponentially [49]. The parameter n = 0.89 indicates that the adsorption process of DCF onto activated sludge in a monolayer configuration is non-favourable, which implies that some energy input is needed for this type of reaction to take place. For the Langmuir model, the values of RL ranged from 1.04 to 20.00, indicating that the adsorption process is also energetically non favourable (RL> 1) As to Temkin’s isotherm model, it takes into account the effects of indirect adsorbate/adsorbate interactions on the sorption process. The linear adjustment for this model showed a R-squared (R2) of 0,999, indicating that the heat of adsorption of all molecules in the layer decreases linearly as a result of an increased surface coverage and that the sorption process follows a chemisorption mechanism [50].
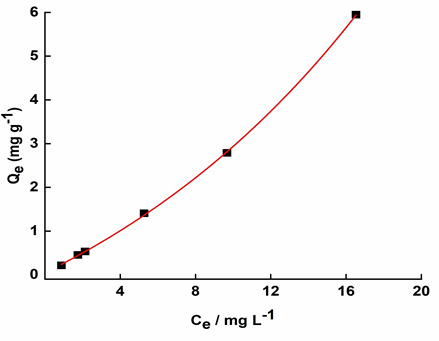
Figure 4 shows the isotherm built with the collected data. The shape of the graph (convex function) indicates that the adsorption process of DCF onto the activated sludge in non-favourable, revealing that the mass of DCF retained per mass unit of sludge is low even for higher concentrations of the drug in the liquid phase.
The mass of DCF adsorbed to the sludge varied between 17 and 44%. This percentage is related to the octanol-water partition coefficient (Kow) of the drug. Compounds with high log Kow> 3 tend to have higher affinity for organic matter, indicating that they are likely to accumulate in soil, sediment and living organisms. This can also be implied from the low solubility of DCF in water (2.37 mg L-1). Morissette et al. (2015) [51] showed that pollutants with a log Kow< 3 had little or no adsorption to sewage sludge, while compounds with log Kow> 3 had 30 to 90% of its mass adsorbed to the sludge in the first 48 hours, which shows some consistency with our results. The authors also presented evidence that the adsorption of DCF onto sludge is pH dependent, leading to conclude that a change in pH and adsorbent surface charge could optimise the results. Since pH conditions in both experiments were above DCF’s pKa value (7.93 - 8.12 > 4.15), it is likely that operating below this range would provoke a change to the compound’s surface charge, which could result in better adsorption outcomes.
One factor that alters the removal of pharmaceutical compounds in systems is the functional group of the molecule. Forinstamce, compounds with electronic groups that strongly donate electrons (hydroxyl groups, for example) show higher rates of biodegradation and, consequently, higher removal efficiencies in aerobic biological treatment systems [41,45]. On the other hand, electron receptors supposedly show some restrictions to biodegradation. DCF, for instance, contains groups of acceptors such as halogen, amine and carboxylic groups, which hinders its potential for biodegradation. Marco-Urrea et al., 2010 [52] suggests that DCF is primarily degraded by the hydroxylation of the aromatic fragment 1-amino-2 unsubstituted, which impairs further degradation of this by-product.
This is concerning, given that DCF’s persistence might affect bacterial community in the activated sludge process. Kraiger et al (2011) [53] demonstrated that diclofenac by-products affect the structure of the nitrite-oxidizing bacterial community, which strongly influence the microbial diversity and affects WWTPs performance.
Conclusions
The adsorption of diclofenac onto the activated sludge reached its equilibrium after approximately 2 hours and proved to be energetically unfavourable, meaning that some energy input is necessary for the reaction to take place. The amount of DCF 2 adsorbed on the sludge particles varied between 17 and 44% and the adsorption ratio of DCF per gram of sludge in both experiments was considerably low (less than 1 mg per g of sludge). Thus, different operational conditions (such as apH, temperature, higher ratio DCF/sludge and the addition of salts) might be evaluated to optimise the outcomes. Moreover, effective process for the removal of a persistent contaminant such as DCF and its by-products in conventional WWTPs may be influenced by HRT increase; however, longer contact times of these compounds with the biomass might interfere in DCF’s elimination due to microbiota toxic effects. Thus, diclofenac removal by aerobic WWTP might reach higher efficiencies adjusting HRT in order to enhance the drug’s sorption onto the sludge and reduce the impact of the pharmaceutical residues on the sludges’s bacterial community.
Acknowledgements
The authors thank the Coordination for the Improvement of Higher Education Personnel (Capes) and São Paulo Research Foundation (FAPESP) (Grant number: 16/23684-0). We also wish to thank the Multiuser Experimental Central of UFABC (CEM-UFABC) for laboratory and equipment support. We thank the employees of ETE Jesus Netto for their aid in effluent sampling.
Conflict of interest:
The authors declare no conflict of interest.
References
Castro, L,V., Brandt, E.M.F., Campos, A.C.V., Aquino, S.F., Werner, D., Afonso, R.J.C.F., Filho, C.R.M. (2018). Behavior of Micropollutants in Polishing Units that Combine Sorption and Biodegradation Mechanisms to Improve the Quality of Activated Sludge Effluent. Water Air Soil Pollut, 229(189), 1-11.View
Jelic, A., Gros, M., Ginebreba, A., Sanches R.C. (2011). Occurrence, partition and removal of pharmaceuticals in sewage water and sludge during wastewater treatment. Water research, 45, 1165-1176. View
Rodrigo A.R., Yolanda P., (2020). Analysis of emerging and related pollutants in aquatic biota, Trends in Environmental Analytical Chemistry.View
Angeles, L.F., Mullen, R. A., Huang, I. J., Wilson, C., Khunjar, W., Sirotkin, H. I., McElroy, A. E., Aga, D. S. (2020). Assessing pharmaceutical removal and reduction in toxicity provided by advanced wastewater treatment systems. Environ. Sci.: Water Res. Technol., 2020, 6, 62View
Kumirska, J. , Łukaszewicz, P. , Caban, M., Migowska, N., Pleni, A., Bielinska, A. B., Czerwicka, M., Qi, F., Piotr S. (2019). Determination of twenty pharmaceutical contaminants in soil using ultrasound-assisted extraction with gas chromatography mass spectrometric detection. Chemosphere 232 (2019) 232 242.View
Puckowski, A., Mioduszewska, K., Łukaszewicz, P., Borecka, M., Caban, M., Maszkowska, J., Stepnowski, P. (2016). Bioaccumulation and analytics of pharmaceutical residues in the environment: a review. Journal of Pharmaceutical Biomedical Analysis, 127, 232–255.View
Kabak, H., Basibuyuk, M. (2012). Diklofenak ve Parasetamol’ün Aktif Çamur Tarafından Adsorbsiyonu [Adsorption of Diclofenac and Paracetamol by Activated Sludge]. Ekoloji, 21(85), 41-48.View
Shore, R.F., Taggart, M.A., Smits, J., Mateo, R., Richards, N.L., Fryday, S. (2014). Detection and drivers of exposure and effects of pharmaceuticals in higher vertebrates. Philosophical Transactions of the Royal Society B, 369, 1-10.View
Hofman-Caris, C.H.M., Siegers, W.G., Van, M.K., Man, A.W.A., Hofman, J.A.M.H. (2017). Removal of pharmaceuticals from WWTP effluent: Removal of EfOM followed by advanced oxidation. Chemical Engineering Journal, 327, 514–521.View
Almeida D. L., Pavanello A., Saavedra, L. P., Pereira, T. S., Castro-Prado, M. A. A., Mathias, P. C. F. (2019). Environmental monitoring and the developmental origins of health and disease. Journal of Developmental Origins of Health and Disease.View
Langford K. H., Thomas K. V. (2009). Determination of pharmaceutical compounds in hospital effluents and their contribution to wastewater treatment works. Environ Intern. 35:766–770. doi: 10.1016/j.envint.2009.02.007.View
Opriș, O., Lung, I., Soran, M. L., Ciorîță, A., Copolovici, L. (2020). Investigating the effects of non-steroidal anti inflammatory drugs (NSAIDs) on the composition and ultrastructure of green leafy vegetables with important nutritional values. Plant Physiology and Biochemistry 151 (2020) 342–351.View
Heberer, T. (2002). Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: a review of recent research data. Toxicology Letters, 131, 5- 7.View
Lonappan, L., Rouissi, T., Brar, S.K., Verma, M., Surampalli, R.Y. (2018). An insight into the adsorption of diclofenac on different biochars: Mechanisms, surface chemistry, and thermodynamics. Bioresource Technology, 249, 386-394.View
NICE Non-Steroidal Anti-Inflammatory Drugs; Guidance and Guidelines. (accessed on 21 July 2020); Available online: https://www.nice.org.uk/advice/ktt13/chapter/evidence-context.View
Carvalho, R. N., Ceriani, L., Ippolito, A., Lettieri, T. (2015). JRC Technical Report: Development of the first Watch List under the Environmental Quality Standards Directive. EUR 27142. [s.l.] Europeian Comission.View
Commission Implementing Decision (EU) 2015/495 OF 20 MARCH. List of substances for monitoring at Union level in the field of water policy. Official Journal of the European Union, Vol. 2015, n. 78, p. 40–42, 2015.View
Tiedeken, E. J. et al. (2017). Monitoring, sources, receptors, and control measures for three European Union watch list substances of emerging concern in receiving waters –A 20 Year systematic review. Science of the Total Environment, v. 574, p. 1140–1163.View
Loos, R., Marinov, D., Sanseverino, I., Napierska, D., Lettieri, T. (2018). Review of the 1st Watch List under the Water Framework Directive and recommendations for the 2nd Watch List, EUR 29173 EN, Publications Office of the European Union,Luxembourg, ISBN 978-92-79- 81839-4, doi:10.2760/614367, JRC 111198.View
Machado K. C., Grassi M. T., Vidal C. et al. (2016). A preliminary nationwide survey of the presence of emerging contaminants in drinking and source waters in Brazil. Science of the Total Environment 572, 138–146.View
Santos, L.H.M.L.M., Araújo, A.N., Fachini, A., Pena, A., Matos, C.D., Montenegro, M.C.B.S.M. (2010). Ecotoxicological aspects related to the presence of pharmaceuticals in the aquatic environment. Journal of Hazardous Materials, 175, 45-95.View
Vendenyapina, M.D., Borisova, D.A., Simakova, A.P., Proshina, L.P.,VEDENYAPIN, A.A. (2013). Adsorption of Diclofenac Sodium from Aqueous Solutions on Expanded Graphite. Solid Fuel Chemistry, 47(1), 59–63.View
Luo, Y., Guo, W., Ngo, H.H., Nghiem, L.D., Hai, F.I., Zhang, J., Liang, S., Wang, X.C. (2014). A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci. Total Environ, 473(474), 619–641.View
Tran, N.H., Reinhard, M., Gin, K.Y.H. (2018). Occurrence and fate of emerging contaminants in municipal wastewater treatment plants from different geographical regions-a review. Water Research, 133, 182–207.View
Lonappana, L. Pulicharla R., Rouissi T., Brar S.K., Verma M., Surampalli R. Y., Valero, J. R. (2016). Diclofenac in municipal wastewater treatment plant: quantification using laser diode thermal desorption—atmospheric pressure chemical ionization—tandem mass spectrometry approach in comparison with an established liquid chromatography-electrospray ionization–tandem mass spectrometry method. Journal of Chromatography A. 10.1016/j.chroma.2016.01.030.View
Vieno, N., Sillanpää, M. (2014). Fate of diclofenac in municipal wastewater treatment plant — A review. Environment International, 69, 28-39.View
Yoon, J., Ryu, J., Oh. J., Choi, B.G., Snyder, S.A. (2010). Occurrence of endocrine disrupting compounds, pharmaceuticals, and personal care products in the Han River (Seoul, South Korea). Science of the Total Environment, 408, 636-643.View
Vulliet, E., Cren-Olive, C., Grenier-Loustalot, M.F. (2011). Occurrence of pharmaceuticals and hormones in drinking water treated from surface waters. Environmental Chemistry Letters, v.9, p.102-114.View
Lapworth, D.J., Baran, N., Stuart, M.E., Ward, R.S. (2012). Emerging organic contaminants in g r o u n dwater: a review of sources, fate and occurrence. Environmental Pollution,163,287-303.View
Joss, A., Zabaczynski, S., Göbel, A., Hoffmann, B., Löffler, D., McArdell, C.S., Ternes, T.A., Thomsen, A., Siegrist, H., (2006). Biological degradation of pharmaceuticals in municipal wastewater treatment: proposing a classification scheme. Water Res. 40, 1686–1696.View
Quintana, B.J., Weiss, S., Reemtsma, T., 2005. Pathways and metabolites of microbialdegradation of selected acidic pharmaceutical and their occurrence in municipal wastewater treated by a membrane bioreactor. Water Res. 39, 2654–2664.View
Kimura, K., Hara, H., Watanabe, Y., (2007). Elimination of selected acidic pharmaceuticals from municipal wastewater by an activated sludge system and membrane bioreactors. Environ. Sci. Technol. 41, 3708–3714.View
Tambosi, J.L., Sena, R.F., Favier, M., Gebhardt, W., José, H.J., Schröder, H.F., Moreira, R.F.P.M., (2010). Removal of pharmaceutical compounds in membrane bioreactors (MBR) applying submerged membranes. Desalination. 261, 1-2, 148 156.
Matsubara, Milena Emy; Helwig, Karin; Hunter, Colin; Roberts, Joanne; Subtil, Eduardo Lucas; Coelho, Lúcia Helena Gomes. Amoxicillin removal by pre-denitrification membrane bioreactor (A/O-MBR): Performance evaluation, degradation by-products, and antibiotic resistant bacteria. Ecotoxicology And Environmental Safety, v. 192, p. 110258, 2020.View
Carballa, M., Omil, F., Lema, J.M., Llompart, M., García-Jares, C., Rodríguez, I.,Gomez, M., Ternes, T., (2004). Behavior of pharmaceuticals, cosmetics and hormones in a sewage treatment plant. Water Res. 38, 2918 e 2926.View
Grandclement, C., Seyssiecq, I., Piram, A., Chung, P. W.W., Vanot, G., Tiliacos, N., Roche, N., Doumenq, P. (2017). From the conventional biological wastewater treatment to hybrid processes, the evaluation of organic micropollutant removal: A review. Water Research 111, 297 e 317.View
Cherik, D., Benali, M., Louhab, K. (2015). Occurrence, ecotoxicology, removal of diclofenac by adsorption on activated carbon and biodegradation and its effect on bacterial community: A review. World Scientific News, 16, 116-144.View
Mailler, R., Gasperi, J., Rocher, V., Gilbert-Pawlik, S., Geara Matta, D.,Moilleron, R., Chebbo, G. (2013). Biofiltration vs conventional activated sludge plants: what about priority and emerging pollutants removal?. Environmental Science and Pollution Research, 21(8), 5379-5390.View
Margot, J., Rossi, L.; Barry, D. A., Holliger, C. (2015). A review of the fate of micropollutants in wastewater treatment plants. Wires Water, 2, 457-487.View
Standard Methods for the Examination of Water and Wastewater (1998). 20th edn, American Public Health Association/American Water Works Association/Water Environment Federation, Washington DC, USA.View
Fan, H., Li, J., Zhang, L., Feng, L., (2014). Contribution of sludge adsorption and biodegradation to the removal of five pharmaceuticals in a submerged membrane bioreactor. Biochem. Eng. J. 88, 101 e 107.View
Cipiana, U., Yllmaz, B. (2014). Determination of Diclofenac in Pharmaceutical Preparations by UV- and First-Order Derivative Spectrophotometry Methods. Indian Journal of Novel Drug Delivery, 6(1), 25-31.View
Ho. Y.s., McKay, G. (1999) The Sorption Of Lead(Ii) Ions On Peat. Wat. Res., 33(2), 578-584.View
Kosjek, Tina & Heath, Ester & Pérez, Sandra & Petrovic, Mira & Barceló, Damia. (2009). Metabolism Studies of Diclofenac and Clofibric Acid in Activated Sludge Bioreactors Using Liquid Chromatography with Quadrupole Time-of-flight Mass Spectrometry. Journal of Hydrology. 372. 109-117.View
Petrie, B., McAadam, E.J., Scrimshaw, M.D., Lester, J.N., Cartmelli, E. (2013). Fate of drugs during wastewater treatment. Trends in Analytical Chemistry, 49, 145-159.View
Suárez, S., Reif, R., Lema, J.M., Omil, F. (2012). Mass balance of pharmaceutical and personal care products in a pilot-scale single-sludge system: Influence of T, SRT and recirculation ratio. Chemosphere, 89, 164-171.View
Radjenovic, J., Matosic, M., Mijatovic, I., Petrovic, M., 2007. Membrane Bioreactor (MBR) as an Advanced Wastewater Treatment Technology. Springer-Verlag Berlin Heidelberg, 06 11, 5(Part S/2), p. 37–101.View
Rubiano, J.C., Perdomo, M.P., Pramparo, L. (2017). Respirometric study of Diclofenac biodegradation. Entre Ciencia e Ingeniería, 21, 9-13.View
Saadi, R., Saadi, Z., Fazaeli, R., Fardi, N.E. (2015). Monolayer and multilayer adsorption isotherm models for sorption from aqueous media. Korean J. Chem. Eng,32(5), 787-799.View
D. Ringot, B. Lerzy, K. Chaplain, J.-P. Bonhoure, E. Auclair, and Y. Larondelle, (2007).“In vitro biosorption of ochratoxin A on the yeast industry by-products: comparison of isotherm models,” Bioresource Technology, vol. 98, no. 9, pp. 1812–1821.View
Morissette, M.F., Duy, S.V., Arp, H.P.H., Sauvé, S. (2013). Sorption and desorption of diverse contaminants of varying polarity in wastewater sludge with and without alum. The Royal Society of Chemistry, 1(3), 1-11.View
Marco-Urrea E, Pérez-Trujillo M, Cruz-Morato C, Caminal G, Vicent T (2010c) Degradation of the drug sodium diclofenac by Trametes versicolor pellets and identification of some intermediates by NMR. J Hazard Mater 176:836–842.View
Nitrification activity and community structure of nitrite oxidizing bacteria in the bioreactors operated with addition of pharmaceuticals. Barbara Kraigher, Ines Mandic-Mulec. Journal of Hazardous Materials 188 (2011) 78-84.View