Journal of Multidisciplinary Clinical Case Reports Volume 1 (2021), Article ID: JMCCR-101
https://doi.org/10.33790/jmccr1100101Review Article
Small Airway Disease / Bronchiolitis: Case Presentation and Review of Literature
Wafaa A. Elatre, MD, PhD
Assistant Professor of Clinical Pathology, Department of Pathology, Keck School of Medicine of USC, HC4 1450 San Pablo Street, Health Sciences Campus, Los Angeles, United States.
Corresponding Author Details: Wafaa A. Elatre, MD, PhD, Assistant Professor of Clinical Pathology, Department of Pathology, Keck School of Medicine of USC, HC4 1450 San Pablo Street, Health Sciences Campus, Los Angeles, United States. E-mail: wafaa.elatre@med.usc.edu
Received date: 04th August, 2021
Accepted date: 22th August, 2021
Published date: 31th August, 2021
Citation: Elatre, W.A. (2021). Small Airway Disease / Bronchiolitis: Case presentation and review of literature. J Multidiscip Clin Case Rep, 1(1): 101.
Copyright: ©2021, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Introduction
Small airways constitute the quiet zone between the conducting and the respiratory lung zones [1]. Still, Small Airway Disease (SAD) can be a presenting manifestation in multiple diseases includes connective tissue disorders such as rheumatoid arthritis, exposure to tobacco smoke, air borne pollutants, mineral dusts, and viral respiratory infections. It may develop following bone marrow transplantation and in recipients of heart-lung or lung transplantation [2].
We are presenting a case of small airway disease/bronchiolitis with through literature review.
History and clinical presentation
A 35-year-old man had dyspnea on exertion (DOE), cough and episodic fever for 6 years. He had smoked ½ packs per day (½ PPD) x 3 years. His sister has asthma.
A positron emission tomography scan (PFTs) showed moderate to severe obstruction with FEV1 of 31% and the DLCO was 64%.
High-resolution computed tomography (HRCT) showed scattered centrilobular and tree-in-bud nodularity in both lungs, bronchial wall thickening and subtle mosaic attenuation consistent with a component of air-trapping.
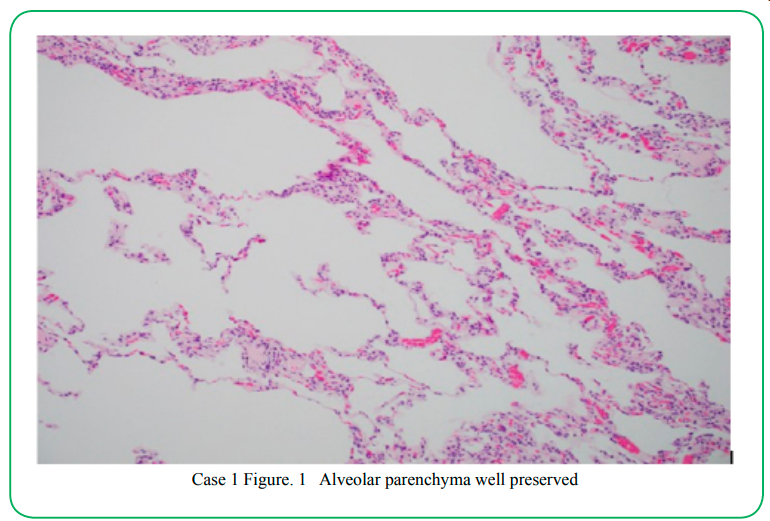
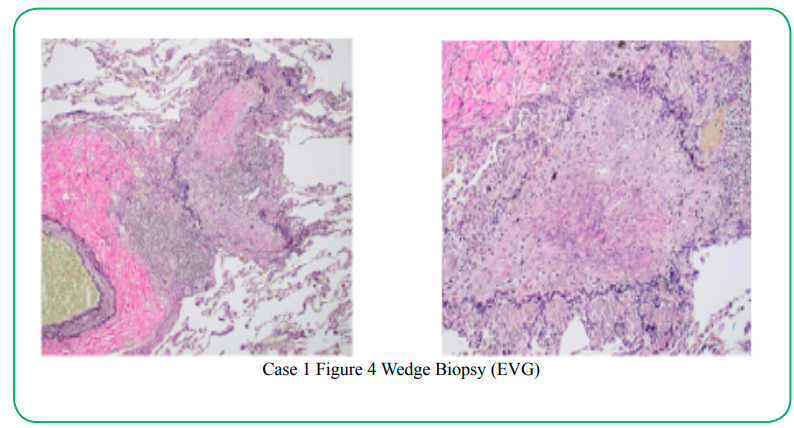
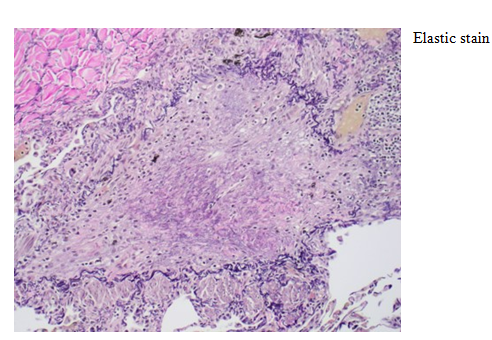
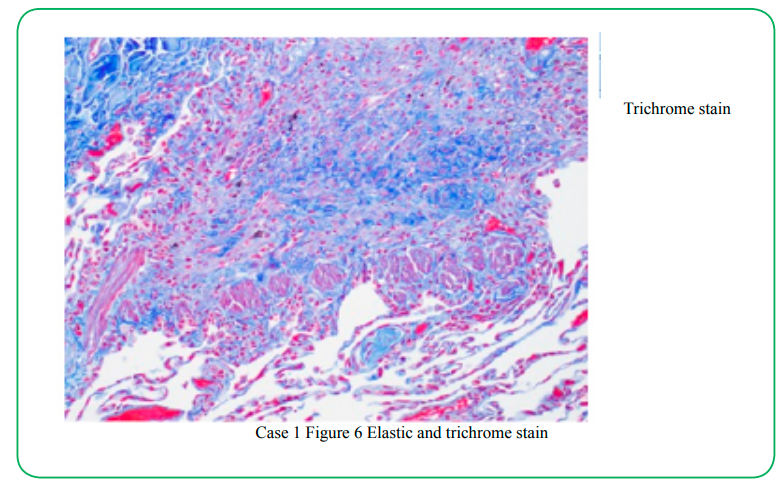
Lungwedge biopsy was performed under video-assisted thoracoscopy (VATS). On microscopic examination, lung architecture was preserved (Fig.1). Pulmonary arteries showed Pulmonary arteriosclerosis with adventitial thickening and non-descript multifocal perivascular scarring (Fig 2). Histologic changes of the bronchioleswere highlighted by elastic and trichrome special stains (fig 4-6) and consisting of hypertrophy of the smooth muscles of the bronchioles, peribronchiolar inflammatory infiltrates, accumulation of mucus in the bronchiolar lumenand bronchiolar scarring. There is a concentric narrowing of the lumen of the bronchioles by the inflammatory fibrosis.(Fig. 2,3)

Case 1: Wedge Biopsy Figure 2. Pulmonary arteriosclerosis with adventitial thickening and non-descript multifocal perivascular scarring
No granuloma was seen in lung parenchyma or in peribronchiolar areas. Mild inflammatory infiltrates wereseen around airways. No pathogen was identified on Grocott and PAS staining and on biopsy cultures. This histological pattern was consistent with Obliterative Bronchiolitis (OB). The lack of marked interstitial inflammation and absence of granuloma ruled out other diagnoses such as sarcoidosis or hypersensitivity pneumonia.
Diagnosis: Obliterative Bronchiolitis (OB)/Constrictive Bronchiolitis (CB)
Constrictive bronchiolitis, also known as bronchiolitis obliterans, is characterized by submucosal and peribronchiolar fibrosis that causes concentric narrowing of the bronchiolar lumen. This lesion is characterized by "a peribronchiolar fibrotic process that surrounds rather than fills the lumen", resulting in extrinsic compression and obliteration of the airway. Constrictive bronchiolitis is mostly seen following inhalation injury, transplantation (eg.bone marrow, lung), or in the context of rheumatoid lung or inflammatory bowel disease. Symptoms include progressive onset of cough and dyspnea associated with hypoxemia at rest or with exercise. Crackles may be present. Pulmonary function tests show a progressive and irreversible airflow limitation. Findings on inspiratory CT scan include centrilobular bronchial wall thickening, bronchiolar dilation, tree-in-bud patternand a mosaic ground-glass attenuation pattern [3,4].
Discussion and differential diagnosis of bronchiolitis:
Bronchioles are small airways (internal diameter of 2 mm or less) that do not contain cartilage in their walls [5]. These airways consist of membranous and terminal bronchioles that are purely air conducting and respiratory bronchioles containing alveoli in their walls [6-8]. Respiratory bronchioles communicate directly with alveolar ducts and are in the range of 0.5mm or less in diameter [5]. The acinus or primary pulmonary lobule (the basic unit of gas exchange) consists of one terminal bronchiole, two to five generations of respiratory bronchioles, alveolar ducts, alveolar sacs, and alveoli [6,9].
Histologically, from the lumen to the adventitia, bronchioles have a lining mucosa (including Clara, ciliated, and basal cells), a basement membrane region, a thin lamina propria, an elastic tissue membrane, a layer of smooth muscle, and an adventitial connective tissue layer that is attached to the surrounding alveolar and perivascular interstitium [10,11]. Lymphoid tissue usually is not present in the normal adult lung. In chronic inflammatory conditions, however, lymphoid follicles along small airways form part of the mucosa-associated lymphoid tissue reaction. When germinal centers are present, the term "follicular bronchiolitis" is appropriate [11]. The epithelium overlying lymphoid follicles represents specialized "lymphoepithelium." Direct communications occur between bronchioles and surrounding alveoli (Lambert's canals), and it is through these canals that bronchiolar epithelium may grow in the healing of bronchiolitis [10]. This has been called lambertosis, bronchiolarization of alveoli, and peribronchiolar metaplasia [5].
Microscopic patterns of bronchiolitis are diverse and can be broadly separated into acute and chronic categories [5]. Acute lesions are sometimes characterized by epithelial necrosis. Chronic bronchiolitis is typically accompanied by variable degrees of intralumenal or peribronchiolar fibrosis ranging from proliferation of fibroblasts and myofibroblasts to collagen scarring. Reparative or reactive hyperplasia of Clara cells and/or smooth muscle often accompanies fibrosis in various forms of chronic bronchiolitis [12]. Several patterns of primary bronchiolitis have been delineated, including constrictive bronchiolitis (also called obliterative bronchiolitis or bronchiolitis obliterans), acute bronchiolitis, diffuse panbronchiolitis, respiratory bronchiolitis, mineral dust airway disease, and follicular bronchiolitis. Only the first three are typically associated with evidence of airflow limitation [13].
Radiologic imaging of the chest, especially HRCT, is a very useful tool in the diagnostic evaluation of a patient with suspected small airways disease [14-17]. Chest radiography demonstrates normal findings or mainly hyperinflation in purely obstructive bronchiolar lesions such as constrictive bronchiolitis [15-17]. In other primary bronchiolar disorders, small [15-17] nodules or reticulonodular infiltrates may be observed. In distal acinar interstitial diseases with secondary bronchiolar involvement, chest radiography usually demonstrates features of the underlying parenchymal disease process such as cryptogenic organizing pneumonia or hypersensitivity pneumonitis.
HRCT at full expiration should routinely be performed when airway disease is suspected or documented clinically. Because visibility on CT is limited to airways more than 2 mm in diameter, normal bronchioles cannot be seen on CT scans [15-17]. However, diseased bronchioles with dilated lumen (> 2 mm in diameter) or thickened walls can be visualized [16,17]. Aside from demonstrating evidence of bronchiolar disease, HRCT of the chest helps identify those cases in which an interstitial lung disease or large airway disease may be the predominant underlying process.
Features of bronchiolar disease on HRCT can be broadly categorized into direct and indirect signs [15-17]. Direct CT findings of bronchiolar disease include bronchiolar wall thickening, bronchiolar dilatation (bronchiolectasis), and luminal impaction that render affected airways directly visible in the lung periphery [16,17]. Bronchiolar wall thickening may occur due to inflammation or fibrosis. Bronchiolar luminal impaction with secretions or fibrotic material manifests as 2 to 4 mm nodular and linear branching centrilobular opacities on CT. The “tree-in-bud” pattern represents a form of bronchiolar impaction in which branching linear structures have more than one contiguous branching site [17].
Indirect signs of bronchiolar disease on CT include subsegmental atelectasis and air trapping [15-17]. Air trapping due to small airway disease often results in a “mosaic pattern” of lung attenuation (multilobular, geographic density differences of the lung parenchyma), which, however, is not specific for bronchiolar diseases [16]. In bronchiolar diseases, the mosaic pattern is caused by hypoventilation of alveoli distal to bronchiolar obstruction (cicatricial scarring of many bronchioles), which leads to secondary vasoconstriction (consequently, under perfused lung) and is seen on CT scans as areas of decreased attenuation. Uninvolved segments of lung show normal or increased perfusion with resulting normal or increased attenuation, respectively. Paired CT scans performed in inspiration and expiration are useful for distinguishing bronchiolar disease from pulmonary vascular disease and some diffuse infiltrative diseases that may also cause a mosaic pattern. In bronchiolar disease, the lucent regions of lung seen at inspiration remain lucent at expiration due to air trapping and show little increase in lung attenuation or decrease in volume as seen for primary vascular lung disease (figure 7).

(Figure 7) Mosaic attenuation (hyperlucent pulmonary lobules alternating with normal/ increased density) and ground glass due to air-trapping (arrowheads) in expiratory HRCT.
There have been several proposed classifications of small airways disease, including, for example, one of the initial classifications by Myers and Colby in 1993 which classify small airways disease into seven categories [Myers and Colby.9]
1) Acute (infectious) bronchiolitis
2) Bronchiolitis obliterans-organizing pneumonia; cryptogenic organizing pneumonia
3) Constrictive (obliterative) bronchiolitis; bronchiolitis obliterans
4) Adult bronchiolitis Respiratory (smoker’s) bronchiolitis-associated interstitial lung disease
5) Mineral dust airways disease
6) Follicular bronchiolitis
7) Diffuse panbronchiolitis.
A relatively recent 2006 classification by Katzenstein [9,18,19] divide small airway disease into four types: [Data were derived from Katzenstein] [20].
1) Cellular bronchiolitis Constrictive bronchiolitis obliterans
2) Nonspecific chronic bronchiolitis Respiratory (smoker’s) bronchiolitis
3) Follicular bronchiolitis
4) Diffuse panbronchiolitis
Another recent classification that is descriptive of the various pathologic patterns was publishedIn 2008, it consists of 15 categories [Cagle et al] [21].
1) Cellular bronchiolitis
2) Acute bronchiolitis
3) Acute and chronic bronchiolitis
4) Chronic bronchiolitis, with or without fibrosis
5) Subtypes of cellular bronchiolitis
6) Follicular bronchiolitis
7) Diffuse panbronchiolitis
8) Bronchiolitis obliterans with intraluminal polyps
9) Constrictive bronchiolitis
10) Respiratory bronchiolitis
11) Mineral dust-associated airway disease
12) Peribronchiolar metaplasia
13) Bronchiolocentric nodules
14) Asthmatic-type changes
15)Chronic bronchitis/emphysema-associated small airways changes.
One might classify these diseases according to whether the histologic patterns, along with clinical and radiologic correlation, show changes diagnostic of a particular disease; however, except for diffuse idiopathic neuroendocrine cell hyperplasia and diffuse panbronchiolitis, and arguably follicular bronchiolitis and respiratory bronchiolitis-interstitial lung disease, the histologic changes, such as cellular bronchiolitis and fibrotic changes, are typically not specific for a distinctive etiology. One could also classify small airways diseases for whether the disease is primary to the small airways or secondary to other lung disease, such as smoking or asthma [18].
Constrictive bronchiolitis (CB) (or obliterative bronchiolitis [OB])
refers to inflammation and fibrosis occurring predominantly in the walls and contiguous tissues of membranous and respiratory bronchioles, with resultant narrowing of their lumens [9].
Histologic Diagnosis: the lesions are patchy, even in individuals with severe respiratory impartment. Therefore, sampling error, either at the time of bronchoscopy or on the part of the pathologist if inadequate numbers of specimens or levels are examined, may lead to an inconclusive diagnosis.
Paradis et al [22] recommended a minimum of six parenchyma-containing tissue fragments to adequately evaluate an allograft lung biopsy for CB, with the further qualification that each specimen should be fluoroscopically localized to within 1 to 2 cm from the pleural surface to ensure that bronchioles are included in the sample. A subsequent report suggested that at least five fragments of alveolated tissue, each with at least one bronchiole and 100 alveoli, be obtained [23]. In advanced stages, the presence of extensive subepithelial and peribronchiolar fibrosis may impede obtaining adequate endoscopically directed biopsy specimens [24].
The primary histologic hallmark of CB is the presence of submucosal bronchiolar fibrosis [9,23,25,26]. The initial event is believed to be bronchiolar inflammation with resulting epithelial necrosis.
The inflammation can extend through the mucosa, submucosa and into the peribronchiolar parenchyma, and it is quickly associated with young, myxoid fibrous connective tissue that compromises or obliterates the bronchiolar lumen (Figure 8). Constrictive bronchiolitis with an inflammatory component is referred to as "active" in both the 1990 Working Formulation for the Diagnosis of Heart and Lung Rejection (Banff criteria) and in the 1995 Revised Working Formulation 2523

(Figure 8) Initial stage probably consists of submucosal lymphocytic inflammation, with resulting epithelial necrosis - rarely seen in lung transplants but may be seen in non transplant setting. Termed “active OB” in Banff lung rejection criteria.
The bronchiolar changes are progressive, and in "pure" CB they involve only the membranous and terminal (respiratory) bronchioles. Eventually, dense fibrous eosinophilic plaques form in the bronchioles. They may be eccentric, concentric, or completely obliterate the lumen [4].
When peribronchiolar inflammation is absent, CB (OB) is referred to as "inactive" in the Working Formulation [23,25]. In transplant patients, CB is often inactive at initial diagnosis.
Sever cases of inactive CB with complete cicatricial destruction of bronchioles can be difficult to diagnose in hematoxylin-eosin-stained sections, manifesting as focal or patchy fibrosis with atelectasis. The histologic clue is the absence of the distal bronchial tree, which in the normal lung should be distributed in proximity to the arterial tree. In this setting, staining for elastica will frequently demonstrate residual bronchiolar elastic fibers within the scarred areas. For this reason, elastic stains should be performed on all tissue blocks when there is clinical suspicion of CB [4](Figure 9-12).
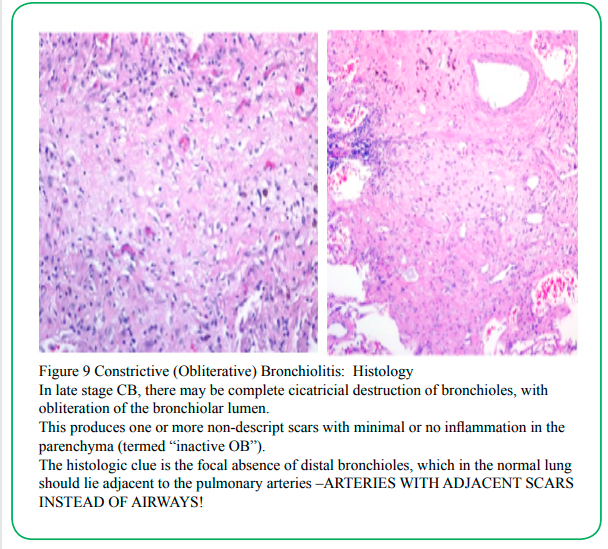
(Figure 9) Constrictive (Obliterative) Bronchiolitis: Histology In late stage CB, there may be complete cicatricial destruction of bronchioles, with obliteration of the bronchiolar lumen. This produces one or more non-descript scars with minimal or no inflammation in the parenchyma (termed “inactive OB”). The histologic clue is the focal absence of distal bronchioles, which in the normal lung should lie adjacent to the pulmonary arteries –ARTERIES WITH ADJACENT SCARS INSTEAD OF AIRWAYS!
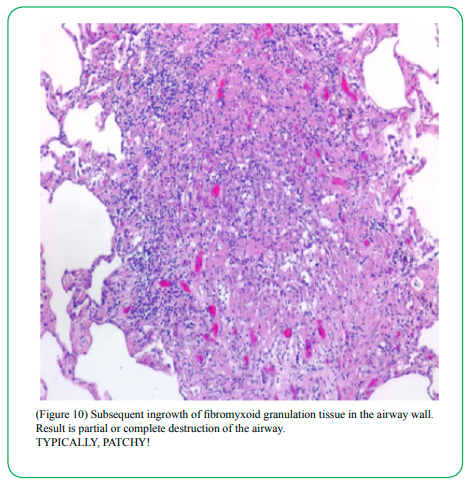
(Figure 10) Subsequent ingrowth of fibromyxoid granulation tissue in the airway wall. Result is partial or complete destruction of the airway. TYPICALLY, PATCHY!
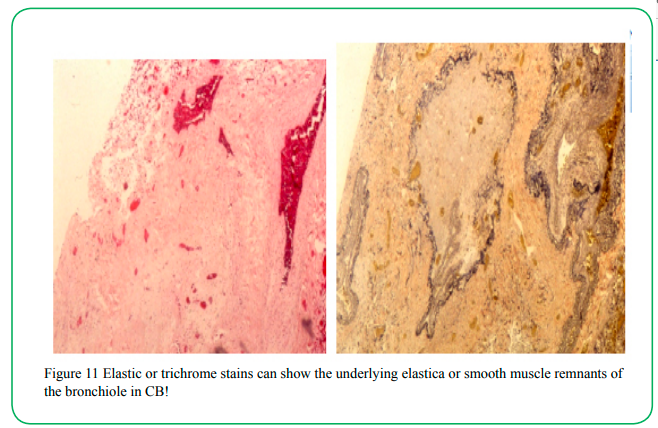
(Figure 11) Elastic or trichrome stains can show the underlying elastica or smooth muscle remnants of the bronchiole in CB!
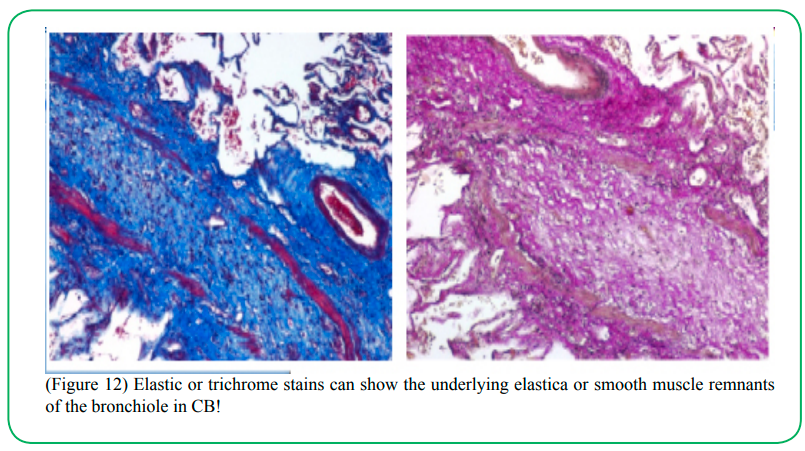
(Figure 12) Elastic or trichrome stains can show the underlying elastica or smooth muscle remnants of the bronchiole in CB!
Known causes and associations with constrictive bronchiolitis include connective tissue disorders (most common), infections [7,27,28], inhalational injury [7,29], chronic hypersensitivity pneumonitis [29], drugs [30,31] organ transplantation [32], and many others.
Rheumatoid arthritis–associated constrictive bronchiolitis occurs principally in women in their fifth to sixth decades of life [33-36]. Most have had long-standing rheumatoid arthritis, although in rare cases pulmonary abnormalities antedate rheumatologic manifestations. There is considerable heterogeneity in the rapidity of progression, with some patients deteriorating slowly [37]. Minor degrees of constrictive bronchiolitis are probably present and subclinical in many patients with rheumatoid arthritis [37]. Penicillamine therapy has been implicated as a potential etiologic factor in some of these patients [34].
Constrictive bronchiolitis with airflow obstruction has been known to complicate a variety ofpulmonary infections and inhalational injury [7,27,28]. Viral infections, particularly adenovirus, have been most frequently implicated [7,27,28].
One of the long-term complications of post infectious constrictive bronchiolitis occurring in childhood is the development of the Swyer– James (or MacLeod's) syndrome, that is, unilateral hyperlucent lung with evidence of air trapping and decreased vascularity [2,7,6,38].
Constrictive bronchiolitis after noxious inhalational injury, e.g., ammonia, presents as cough and progressive dyspnea beginning daysto weeks after recovery from acute exposure [39].
Patients with allogeneic or autologous bone marrow transplantation, heart–lung transplantation, or lung transplantation may develop constrictive bronchiolitis as a chronicrejection phenomenon [7,32,40-43]. This problem is a major threat to long-term survival in these transplant recipients and may affect t up to 65% of patients at 5 years after lung transplantation [43].
Constrictive bronchiolitis is the primary cause of late death after lung transplantation [32,43]. Confirming the diagnosis of constrictive bronchiolitis in transplant recipients by transbronchial lung biopsies is problematic because of the patchy distribution of lesions and difficulties in obtaining adequate samples of bronchioles [32,43].
Thus, the phenomenon of progressive airway obstruction in transplant recipients is termed “bronchiolitis obliterans syndrome,” a clinical diagnosis, and is defined physiologically by a decrement in FEVof 20% or more below a stable baseline [32,44] .
Other causes and associations with constrictive bronchiolitis have included neuroendocrine cellhyperplasia or multiple carcinoid tumorlets [45-48], paraneoplastic pemphigus [49], inflammatory bowel disease [50,51], exposure to incineratory ash [52], Sauropus androgynus ingestion [53], gold therapy [30], and penicillamine therapy [31]. It is likely that pathogenetic mechanisms vary according to the cause or underlying disease associated with constrictive bronchiolitis. However, various forms of insults can eventually lead to a similar histopathologic lesion.
For example, in bronchiolitis obliterans syndrome associated with lung transplantation, alloreactivity directed toward human leukocyte antigens, airway ischemia, viral infections, and airway inflammation are major factors that lead to accumulationof profibrotic cytokines and the development of bronchiolar fibrosis [43,54,55]. Constrictive bronchiolitis occurring in patients with paraneoplastic pemphigus, however, involves deposition of IgG autoantibodies on the surface of bronchial epithelial cells and acantholytic changes [49]. There are likely multiple mechanisms through which the lesion of constrictive bronchiolitis takes form. When constrictive bronchiolitis occurs with no identifiable cause, it is referred to as cryptogenic constrictive bronchiolitis [56]. It is rare and occurs mostly in women [56]. Patients with constrictive bronchiolitis present with persistent cough and worsening dyspnea.
Basilar inspiratory crackles may be heard on auscultation of the lungs in some patients [56] (20). Progressive airway obstruction, often associated with air trapping, is seen by pulmonary function testing in the majority of affected patients [5,7,9,56].
Diffusing capacity is commonly reduced, and there is no significant response to bronchodilators during pulmonary functiontesting [56,36].
Chest radiography in patients with constrictive bronchiolitis demonstrates normal findings ornonspecific abnormalities including variable degrees of hyperinflation, peripheral attenuation of the vascular markings, and, sometimes, nodular or reticulonodular opacities [14,56].
HRCT demonstrates mosaic (multilobular) areas of decreased attenuation and vascularity, evidence of air-trapping (accentuated on expiratory views), and peripheral cylindric bronchiectasis [14-17,29]. Marked heterogeneity of lung density (mosaic attenuation) may be seen due to decreased perfusion of areas with bronchiolar obstruction and blood flow redistribution to normal areas [57].
Although these HRCT findings are not specific for constrictive bronchiolitis, this constellation of features can be diagnostic in the appropriate clinical setting.
Radiologic finding of mosaic pattern of lung attenuation can be due to pulmonary vascular disease and diffuse parenchymal disease as well as small airway disease. Expiratory HRCT imaging andcontrast enhancement of vasculature can distinguish these deferential diagnostic possibilities [58]. Additional features may be seen on HRCT of constrictive bronchiolitis depending on the underlying cause.
In most clinical settings, constrictive bronchiolitis tends to be progressive and is poorly responsive to corticosteroid therapy [5,7,9,36,59]. Progressive airflow limitation may result in respiratory failure and death. Management of posttransplant constrictive bronchiolitis involves augmentation of immunosuppression that is often ineffective, thus requiring consideration of retransplantation in some patients [41,43]. Use of statin medications has been associated witha reduced incidence of bronchiolitis obliterans syndrome after lung transplantation.
Acute Bronchiolitis
Acute bronchiolitis is a term most often used to describe an illness in infants and children characterized by acute wheezing with concomitant signs of respiratory viral infection [60,61].
Acute bronchiolitis is the most common disease of the respiratory tract during the first year of life and occurs in annual epidemics during winter. Respiratory syncytial virus is the etiologic agent in the majority of patients, but other viruses (adenovirus, influenza, parainfluenza) and nonviral pathogens (mycoplasma, chlamydia) can cause a similar syndrome [28,60-63].
Other pathogens including fungal and mycobacterial infections also may cause infectious bronchiolitis [16]. Symptomatic acute bronchiolitis in adults is relatively rare but can be caused by infectious agents such as respiratory syncytial virus.
Infants often present with tachypnea, wheezing, and tachycardia, with more severely ill patients exhibiting chest retraction and nasal flaring. Lung hyperinflation is often found on x-ray. Some cases may show patchy ground glass opacities or small centrilobular nodules as well. High-resolution chest CT scan may show branching linear opacities or small centrilobular nodules. Focal areas of nodularity may be present, representing foci of bronchopneumonia. In obvious cases of acute bronchiolitis, chest radiology is more frequently being omitted [18]. Patients typically do not undergo open biopsy; however, biopsy may be obtained in some cases having unusual presentations.
Because small airways in adults contribute less to total pulmonary resistance, acute infectious bronchiolitis may spare adults the severesymptoms characteristic of bronchiolitis in infants. Acute bronchiolitis in adults may also be seen with aspiration, toxic inhalation, connective tissue diseases, lung and bone marrow transplantation, and Stevens–Johnson syndrome [63].
Histologically, acute bronchiolitis consists of bronchioles filled with necrotic debris and purulent exudative material, as well as sloughed bronchiolar mucosal cells. The residual bronchiolar mucosa and surrounding bronchiolar wall contain a neutrophilic infiltrate [5,9,13,64-68] (Figure 13).
Most cases are treated symptomatically, with generally excellent prognosis. Rare cases exhibit long-term morbidity or mortality. In a small number of patients, long-term disease results in constrictive bronchiolitis. Drug treatments have not shown convincing benefit; however, oxygen is generally administered in cases with significant hypoxia. Home oxygen therapy is being more frequently used to reduce hospital stay. Also, corticosteroids have reportedly shown benefit in some patients with croup, and nebulized saline has been reported to reduce morbidity and length of hospital stay in patients with acute bronchiolitis.
The major clinical findings in acute bronchiolitis include tachypnea, tachycardia, and prolonged expiration [60,63]. Wheezing and crackles are usually present and may be accompanied by nasal flaring and chest retractions in infants [63]. Pulmonary function testing demonstrates findings of airway obstruction [62]. The radiologic pattern of acute bronchiolitis is variable.
Chest radiography typically demonstrates only hyperinflation. Tiny nodules, linear opacities, patchy ground-glass opacities or consolidation and collapse (atelectasis) may sometimes be seen [55,63]. HRCT in acute infectious bronchiolitis demonstrates small, ill-defined centrilobular nodules representing bronchioles impacted with inflammatory material and peribronchiolar inflammation, branching linear opacities corresponding to inflamed airway walls, and focal areas of consolidation due to bronchopneumonia [63,69].
Most patients with acute bronchiolitis can be managed at home with supportive care because respiratory symptoms are generally mild. When the illness is more severe, hospitalization, supple mental oxygen, antiviral agents, corticosteroid therapy, and bronchodilators will need tobe considered [62,69].
Due to its clinically self-limited course, acute viral bronchiolitis is rarely encountered in biopsy material. Pathologic studies of infectious bronchiolitis have shown intense neutrophilic and mononuclear infiltration [70]. Necrosis of bronchiolar epithelium occurs in severe cases. By definition, there is no fibroblast proliferation or collagen deposition. In rare cases, though, healing of viral bronchiolitis can lead to fibrous obliteration of small airways, resulting in chronic airflow limitation (7). At least some of these patients are believed to evolve to the histologic finding of constrictive bronchiolitis, which is characterized by a number of causes and clinical associations.
Overall, the mortality rate of acute bronchiolitis is less than 1% [62,69]. In a small subset of patients, healing of acute bronchiolitis can lead to bronchiolitis obliteration of small airways resulting in chronic airflow limitation, i.e., constrictive bronchiolitis [69]. This phenomenon is seenmost often after adenovirus infection but also after measles, pertussis, mycoplasma, influenzaA, and other infections.
In some of these cases, unilateral hyperlucent lung and/or acombination of geographic hyperlucency, central bronchiectasis, and vascular attenuation maybe seen (Swyer–James syndrome) [62,69].
Acute and Chronic Bronchiolitis
Acute and chronic bronchiolitis is generally found in adults. Potential etiologies include infections, such as respiratory syncytial virus, other noninfectious etiologies exist. Some cases are idiopathic [5,9,64,71]. Patients often have mild symptoms but may present with shortness of breath of variable duration, with or without cough. Pulmonary function tests may show an obstructive pattern. Histologically, bronchiolar lumen contains purulent exudative material and sloughed bronchiolar mucosal cellular debris and variable amounts of mucus, and bronchiole mucosa and walls contain a mixed neutrophilic infiltrate and chronic inflammatory cell infiltrate made up predominantly of lymphocytes and plasma cells. The mixed acute and chronic inflammatory cell infiltrate extends into peribronchiolar tissue, and edema may be present [23,24,39,67,68,71,72]. In patients with disease caused by respiratory syncytial virus, various chemokines, including regulated upon activation, normal T cell expressed and secreted; IL-8; IL-1; and IL-6 have been implicated in recruitment and activation of inflammatory cells, including neutrophils, macrophages, lymphocytes, and eosinophils, to the infected area [65,69,70,72]. Most patients have a good prognosis with generally full.
Diffuse Panbronchiolitis
Diffuse panbronchiolitis is a rare form of bronchiolitis identifiedmainly in Asia, particularly in Japanese adults, and is characterized by bronchiolar inflammation and chronic sinusitis [56,72,73].
Few cases occurring in non-Asian patients have been described in the United States [73,74].
The cause is unknown, but an association with human leukocyte antigen Bw54 antigen has been reported in Japanese patients [72].
Pathogenesis is not well understood, but the racial tendency of this disease suggests genetic predisposition. Neutrophils and T-lymphocytes, particularly CD8 cells, together with cytokines IL-8 and macrophage inflammatory protein-1-αare believed to play key roles in the development of this disease [72,75]. Accumulation of activated neutrophils in the airways appears to be an important mechanism of injury in this disease [73]. Histopathologic findings in diffuse panbronchiolitis are characteristic and consist of bronchiolocentric infiltration of lymphocytes, plasma cells, and foamy macrophages at the levelof the respiratory bronchioles dense peribronchiolar infiltrate of mainly mononuclear cells is associated with astriking infiltrate of vacuolated histiocytes. Vacuolated, lipid-laden histiocytes extend and expand contiguous peribronchiolar alveolar septa [74]. Intraluminal neutrophils can be prominent.
Organization of intraluminal exudates may form polypoid plugs. In addition, marked increase in the number of dendritic cells has been found in both the bronchiolar epithelium and submucosal tissues of patients with diffuse panbronchiolitis [76]. Although this pattern of bronchiolitis is characteristic of diffuse panbronchiolitis, nearly identical changes have been described in a broad range of airway-centered disease processes including bronchiectasis and rheumatoid arthritis–related bronchiolitis [77,78]. Patients present with subacute onset of cough productive of purulent sputum, dyspnea, and evidence of airflow obstruction. The majority of patients also complain of chronic sinusitis, suggesting that there may be an underlying abnormality of ciliary function [72].
Chest examination may reveal decreased breath sounds with coarse crackles or wheezing present [74]. Digital clubbing is unusual [74]. The most characteristic laboratory feature associated with diffuse panbronchiolitis is the persistent elevation of cold agglutinins, but tests for Mycoplasma pneumoniae are negative [72]. Elevated leukocyte count and erythrocyte sedimentation rate are common [74].
In the early stages of the disease, the sputum generally contains normalflora or Hemophilus influenzae. Colonization with Pseudomonas aeruginosa eventually occurs, which appears to accelerate the destructive process [74].
Pulmonary function testing generally demonstrates marked obstructive impairment [72].
Insome patients, a superimposed mild to moderate restriction may also be seen [56,74].
On chest radiography, diffuse panbronchiolitis is characterized by diffuse small (up to 5 mm indiameter), ill-defined nodular opacities most prominent over the lung bases and symmetrically distributed.
Mild to moderate hyperinflation may be seen. In late stages, the radiographic features of cylindric bronchiectasis may become evident [72,56,74].
Findings on HRCT depend on the stage of the disease and include centrilobular nodules, thickened and ectatic bronchioles, and peripheral air trapping [56,72,74].
Centrilobular nodules are poorly defined and measure less than 5 mm in diameter some of these nodules are connected to distal branching structures that represent secretion filled bronchioles (tree-in-bud appearance). In later stages, cystic dilatation of the nodules that corresponds to bronchiolectasis is seen [15]. The natural history of diffuse panbronchiolitis is characterized by progressive respiratory days function with episodic bacterial superinfection, often with P. aeruginosa [72,74]. In advanced disease, patients succumb to chronic respiratory failure and cor pulmonale leading to death. Low-dose erythromycin therapy, 400 to 600 mg per day, is the preferred therapy and hasshown some efficacy [56,72]. Therapeutic efficacy of macrolides in the treatment of patients with diffuse panbronchiolitis may be based on the ability of these antibiotics to impair the production of proinflammatory cytokines, including IL-1β and IL-8, rather than the irantibacterial properties [73,79-81]. Decreased levels of chemokines reduce neutrophil influxand associated inflammatory mediators in the airways [73]. Macrolide therapy has also been demonstrated to inhibit mucus and water secretion from airway epithelial cells [73]. Lung transplantation has been used in some patients, and diffuse panbronchiolitis has recurred inthe allograft of one patient [82].
Respiratory Bronchiolitis
Exposure to cigarette smoke results in various alterations of bronchioles ranging from potentially reversible inflammatory reactions to fixed scarring [83,84]. Respiratory bronchiolitis is a specific form of smoking-related small airways disease and was first described by Niewoehner and colleagues [85] as an incidental finding at autopsy in young cigarette smokers.
Respiratory bronchiolitis has been rarely reported in nonsmokers with other inhalational exposures, especially asbestos and non-asbestos dusts as well as various fumes [7,86].
The most distinctive feature of respiratory bronchiolitis is the prominent accumulation of pigmented macrophages in the lumen of respiratory bronchioles and adjacent alveoli. Respiratory bronchiolitis is suspected when HRCT of the chest demonstrates parenchymal micronodules in a smoker who undergoes CT of the chest, usually for an unrelated indication.
Many such patients are asymptomatic except for a “smoker's cough” and do not need any specific treatment other than smoking cessation. The term RB-ILD is reserved for those patients who have additional findings of interstitial lung disease.
Respiratory bronchiolitis is suspected when HRCT of the chest demonstrates parenchymal micronodules in a smoker who undergoes CT of the chest, usually for an unrelated indication.
Follicular Bronchiolitis
Follicular bronchiolitis (FB) is defined by the presence of hyperplastic lymphoid follicles with reactive germinal centers distributed along the bronchovascular bundles [87,88].
Currently, FB is classified as one of the non-neoplastic (reactive) pulmonary lymphoid disorders in a group known as the lymphoproliferative pulmonary diseases (LPDs) It is mainly distinguished from other reactive pulmonary lymphoid diseases in its group such as lymphocytic interstitial pneumonia (LIP) and nodular lymphoid hyperplasia (NLH) by the pattern and extent of pulmonary parenchymal involvement [89].
FB can be generally classified based on its underlying etiology into a primary and secondary form. Secondary follicular bronchiolitis is a relatively common disease that occurs in association with many systemic and pulmonary diseases like with connective tissue diseases (particularly rheumatoid arthritis), immunodeficiency syndromes including AIDS, pulmonary infections, or ill-defined hypersensitivity reactions [90]. Idiopathic (Primary) form of follicular bronchiolitis is a rare disease that occurs without an associated primary immunodeficiency state, inflammatory, autoimmune, infectious, or connective tissue disease [88].
Patients with FB usually present with progressive dyspnea [15,91]. Variable pulmonary function abnormalities have been reported, including obstructive, restrictive, and mixed patterns [15,92-94]. The predominant finding on chest radiography is bilateral, small nodular, or reticulonodular infiltrates with intrathoracic adenopathy [15].
Chest radiographs may sometimes look normal [15]. The cardinal features of follicular bronchiolitis on HRCT consist of centrilobular nodules measuring 1 to 12 mm in diameter, variably associated with peribronchial nodules and patchy areas of ground-glass opacity [88].
Nodules and ground-glass opacities are generally bilateral and diffuse in distribution. Mild bronchial dilatation with wall thickening is seen in some cases. Mosaic perfusion, pleural effusions, or areas of honeycombing are not seen [88].
Diagnosing FB on pathological specimens requires demonstrating two fundamental features, first, the presence of well-formed lymphoid follicles in the walls of bronchioles and the second is narrowing or complete obliteration of the bronchiolar lumen [89,95]. Secondary features that can be associated with FB on pathologic specimens include foci of organizing pneumonia, foci of obstructive pneumonia and bronchiolar intraluminal neutrophilic infiltrate [95] (Figure14).
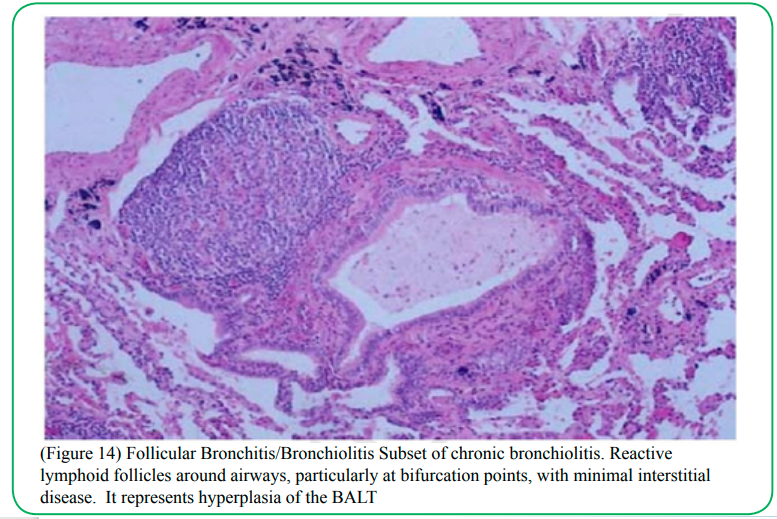
(Figure 14) Follicular Bronchitis/Bronchiolitis Subset of chronic bronchiolitis. Reactive lymphoid follicles around airways, particularly at bifurcation points, with minimal interstitial disease. It represents hyperplasia of the BALT
Prognostic implication of follicular bronchiolitis is unclear. Treatment is generally directed to the underlying disease when such association is recognized. Those patients with no identifiable underlying cause have generally been treated with bronchodilators and corticosteroids. More recently, erythromycin therapy has been reported to be of benefit [96].
Interstitial Lung Disease with a Prominent Bronchiolar Component:
These interstitial lung diseases include hypersensitivity pneumonitis, respiratory bronchiolitis -associated interstitial lung disease (RB-ILD), desquamative interstitial pneumonia (DIP), and cryptogenic organizing pneumonia [13].
Hypersensitivity pneumonitis is an immunologically induced inflammatory disease involving the lung parenchyma and terminal airways secondary to repeated inhalation of a variety of organic dusts and other agents in a sensitized host [97].
Hypersensitivity pneumonitis is one of the more common forms of interstitial lung disease. Aside from the obvious parenchymal involvement, histologic evidence of bronchiolitis is seen in virtually all cases [98-101].
Bronchiolitis in hypersensitivity pneumonitis is characterized by patchy peribronchiolar infiltrate of mainly lymphocytes with variable numbers of poorly formed granulomas or isolated multinucleated histiocytes [98]. In about half of patients, the bronchiolitis is accompanied by intraluminal fibrosis resembling that seen in organizing pneumonia. Smooth muscle hypertrophy associated with peribronchiolar fibrosis contributes to extrinsic bronchiolar narrowing [99]. This inflammatory reaction may produce scarring of the airway wall and may be associated with mild airflow abnormalities. Bronchiolar involvement is the basis of airflow obstruction that is observed in varying degrees in all stages of hypersensitivity pneumonitis. In general, the degree of bronchiolar involvement tends to be proportional to the severity of fibrosis seen in the lung parenchyma [99].
On HRCT, the most frequent features of hypersensitivity pneumonitis are areas of decreased attenuation in a mosaic pattern (caused by small airways obstruction), followed by ground glass opacities, poorly circumscribed centrilobular nodules (1–5 mm in diameter), and a reticular pattern. Areas of decreased attenuation and mosaic perfusion (mosaic pattern) suggesting air trapping are indirect signs of bronchiolar obstruction. The poorly defined nodules and micronodules seen in hypersensitivity pneumonitis have a predominantly centrilobular distribution and represent intraluminal granulation tissue in the bronchioles and adjacent alveoli. Parenchymal inflammation and fibrosis are manifest as areas of ground-glass opacities and reticular densities.
Management of hypersensitivity pneumonitis mainly involves avoidance of exposure to the inciting agent when the cause can be identified. If the cause is unknown or respiratory impairment is substantial, corticosteroid therapy may be needed.
RB-ILD and DIP As discussed earlier, respiratory bronchiolitis is common in cigarette smokers and usually occurs without significant accompanying interstitial lung disease. In a small portion of smokers, symptomatic interstitial lung disease may occur in association with respiratory bronchiolitis.
In 1987, Myers and colleagues described what was subsequently labeled RB-ILD. This is a clinicopathologic entity seen almost exclusively in current or former cigarette smokers and may be confused with other interstitial lung diseases, in particular, idiopathic pulmonary fibrosis [80-83,111-113].
Histologically, RB-ILD is characterized by the presence of pigmented macrophages and mild interstitial inflammatory changes centering on respiratory bronchioles (Figure 7) and neighboring alveoli (peribronchiolar air spaces) with sparing of more distal air spaces [1,2,81,82,111,112,114].
The changes are patchy and have a bronchiolocentric distribution. Alveolar septa in the peribronchiolar region may be mildly thickened.
DIP is highly related, differing mainly in that the airspace macrophages and interstitial thickening are more extensive and diffuse [1,2,81,86,111,112,114]. That is, respiratory bronchiolitis, RBILD, and DIP represent a continuous spectrum of smoking-related interstitial lung disease [83,86,115].
Because respiratory bronchiolitis and “DIP-like” changes are common incidental findings in cigarette smokers, attributing clinical significance requires careful correlation with the clinical and radiographic findings [116]. These histopathologic changes are presumably triggered by epithelial damage induced by cigarette smoke.
Initial events in the pathogenesis of these lesions have not been clarified. For most patients with RB-ILD and DIP, the onset of symptoms is usually in the fourth or fifth decades of life and is considerably earlier than that noted in patients with idiopathic pulmonary fibrosis/usual interstitial pneumonia [80-83,111,112].
There is a slight male predominance, and these patients have average exposures of over 30 pack-years of cigarette smoking [80-82,112]. Patients with RB-ILD and DIP commonly present with gradual onset of cough and dyspnea [80-83]. Most patients with RB-ILD have relatively mild symptoms compared with those with DIP who commonly have significant dyspnea and hypoxemia [80-83,117].
Auscultation of the lungs reveals inspiratory crackles in about one-half of patients [80-83,118].
Digital clubbing is occasionally seen in patients with RB-ILD compared with nearly 50% of those with DIP. Pulmonary function results in patients with RB-ILD may be normal but more commonly show a mixed obstructive and restrictive pattern of mild to moderate degree. Most patients with DIP have a restrictive defect with reduced diffusing capacity.
Chest radiographs are usually abnormal in both RB-ILD and DIP. Diffuse, fine reticular, or reticulonodular opacities are the most common radiographic findings in patients with RB-ILD and DIP, but ground-glass pattern may be the predominant abnormality in some patients. Thickening of the walls of central and peripheral bronchi is also a common radiographic feature in RB-ILD.
Lung volumes are usually preserved in RB-ILD but tend to appear reduced in DIP unless there is coexisting obstructive lung disease such as emphysema. Normal chest radiographs have been reported in up to 20% of patients with RB-ILD and DIP [102].
Areas of ground-glass attenuation are the most common finding on HRCT for both RB-ILD and DIP (Figure 13), but fine centrilobular nodules may also be seen in RB-ILD. The extent of centrilobular nodules on CT correlates with the degree of macrophage accumulation and chronic inflammation in respiratory bronchioles, whereas ground-glass attenuation correlates with macrophage accumulation in the alveolar spaces and alveolar ducts [102].
Associated centrilobular emphysematous changes may be present, particularly in the upper lobes, but signs of idiopathic pulmonary fibrosis, such as subpleural honeycombing and traction bronchiectasis, are generally absent. Septal thickening and irregular linear opacities are infrequently reported. The mainstay of therapy for RB-ILD and DIP is smoking cessation after which the lesion appears to stabilize or to resolve slowly and generally does not show progression to fibrotic lung disease. However, some of these patients may experience continuing deterioration despite treatment. No death have been attributed to RB-ILD but have occurred with DIP [102]. Corticosteroid therapy has been employed in a few anecdotal cases of RB-ILD, and more widely in DIP, with beneficial results reported [102].
Cryptogenic Organizing Pneumonia (Also Known as Idiopathic BOOP) As already discussed, cases of “bronchiolitis obliterans” in the older literature include a heterogeneous mix of lesions.
Organizing pneumonia has been included in older reports under different names such as bronchiolitis obliterans, proliferative bronchiolitis, or more general terms such as interstitial pneumonias. Liebow and Carrington, included some examples of organizing pneumonia in their original classification of interstitial pneumonias as “bronchiolitis obliterans with classical interstitial pneumonia.” Davidson and coworkers coined the term “cryptogenic organizing pneumonitis” to describe a specific clinicopathologic syndrome with distinctive histopathologic findings. “BOOP” was the term proposed by Epler and colleagues for the same condition in 1985, and since then the term BOOP has become entrenched in popular usage in the United States.
Epler and colleagues separated BOOP from a group of 10 patients described as having “bronchiolitis obliterans without organizing pneumonia” (i.e., “pure” bronchiolitis obliterans), but this was a heterogenous group including three patients with probable constrictive bronchiolitis and five patients with biopsies that were “not a representative sample of tissue.” A recently published classification of the idiopathic interstitial pneumonias developed by an international panel has proposed the term “cryptogenic organizing pneumonia” [121].
The term cryptogenic organizing pneumonia has the advantages of being descriptively accurate, both clinically and pathologically, and of avoiding confusion with other entities traditionally included under the heading of bronchiolitis obliterans. The connotation fits the clinical and morphologic features of a mainly alveolar process, rather than an obstructive airway disease.
Organizing pneumonia is a histopathologic pattern characterized by polypoid intraluminal plugs of proliferating fibroblasts and myofibroblasts within alveolar ducts and spaces with varying degrees of bronchiolar involvement [1,2,121]. The fibroblastic plugs may form “casts” that outline the branching configuration of alveolar ducts and distal airways. The airway lumen appears to be occluded from within, as opposed to the concentric narrowing seen in constrictive bronchiolitis.
The lung parenchyma is involved in a patchy distribution, and the background architecture is generally preserved. Alveolar septal thickening due to an interstitial infiltrate of chronic inflammatory cells and hyperplasia of alveolar lining epithelium may be seen limited to areas of intraluminal fibrosis. Honeycombing is generally not seen [126].
Although the pathogenesis of the organizing pneumonia lesion is not fully understood, an animal model study suggests that Tlymphocytes and IFN- play important roles in the development of intraluminal fibrosis. Bronchoalveolar lavage studies in patients with cryptogenic organizing pneumonia have demonstrated lymphocytic alveolitis with expansion of CD8cells and increased levels of Th1- related cytokines including IFN-, IL-12, and IL-18. Newly formed intraluminal fibromyxoid tissue in cryptogenic organizing pneumonia demonstrates increased capillarization compared with fibroblastic foci of usual interstitial pneumonia. Angiogenesis appears to be mediated by growth factors, vascular endothelial growth factor and basic fibroblast growth factor [126].
The erythrocyte sedimentation rate and C-reactive protein level are increased in many patients.
The most common radiographic finding in patients with cryptogenic organizing pneumonia is patchy bilateral alveolar infiltrates that may have a “ground glass” appearance and normal lung volume. These infiltrates may come and go in different locations over a course of several weeks or longer. The distribution of these patchy infiltrates can be peripheral, resembling chronic eosinophilic pneumonia. Sometimes, these infiltrates may look denser and appear as multifocal mass-like regions of consolidation. Occasionally, infiltrates may be unilateral [126].
Diffuse interstitial infiltrates are the predominant radiographic abnormality in less than 20% of patients with cryptogenic organizing pneumonia [7]. This latter radiographic pattern may be more common in patients with associated connective tissue diseases and tends to be associated with a poorer prognosis. Other radiographic presentations include a unifocal region of consolidation or mass as in those patients with “focal” organizing pneumonia. Small rounded opacities are described in 18 to 30% of cases [121,123]. Pleural disease is seen in up to one-quarter of subjects. Hyperinflation is not a feature of organizing pneumonia.
On HRCT, nearly all patients with organizing pneumonia have patchy, unilateral or bilateral, areas of air-space consolidation or ground-glass attenuation (ranging from 2 cm to extensive bilateral disease), with nodules being less common [6,123]. Subpleural or peribronchial distribution is demonstrated in up to 50% of cases. Linear or reticular opacities may be seen in some subjects. A small number of patients with cryptogenic organizing pneumonia will progress to honeycombing. As is true in many inflammatory lung diseases, moderate mediastinal adenopathy may occur in organizing pneumonia. Bronchoalveolar lavage usually reveals an increase in lymphocytes count as the most common pattern.
Diagnosis of cryptogenic organizing pneumonia requires identification of the pathologic findings of organizing pneumonia in the appropriate clinical and radiographic context and exclusion of known causes or underlying disease [103,104]. Transbronchial lung biopsy may be sufficient if the specimen is large enough to contain all the elements of the lesion and if the clinical findings are appropriate [103,105,106]. If not, a surgical lung biopsy may be needed to rule out other diseases such as hypersensitivity pneumonitis, nonspecific interstitial pneumonia, usual interstitial pneumonia, diffuse alveolar damage, or chronic eosinophilic pneumonia [104].
A few other interstitial lung diseases deserve mention in this section with respect to their involvement of bronchioles. Pulmonary Langerhans’ cell histiocytosis is a smoking-related interstitial lung disease characterized by excessive proliferation of Langerhans’ cells and destructive granulomatous lesions in the lung. The earliest lesion in pulmonary Langerhans’ cell histiocytosis is accumulation of Langerhans’ cells with varying numbers of eosinophils in the submucosa and adjacent interstitium of bronchioles and alveolar ducts [107]. Recruitment and activation of Langerhans’ cells to the lung may result directly from cigarette smoke or may be mediated by cytokines released from other cells including alveolar macrophages and airway neuroendocrine cells [107].
This process affects predominantly respiratory bronchioles and to a lesser extent distal terminal bronchioles [107,108]. Some of the cystic lesions seen in pulmonary Langerhans’ cell histiocytosis result from this bronchiolocentric process, which results in destruction of the bronchiolar wall [108]. Pulmonary function testing in patients with pulmonary Langerhans’ cell histiocytosis reveals both obstructive and restrictive changes [109]. The effects from cigarette smoking may be superimposed and can be difficult to distinguish from the effects of pulmonary Langerhans’ cell histiocytosis itself.
By virtue of their perilymphatic distribution, sarcoid granulomas are concentrated around the airways [110]. Small and large airways are frequently involved by granulomas, and measurable airflow obstruction occurs in a small percentage of cases [110]. Functional studies using sophisticated tests have suggested airflow obstruction located at the level of small airways to be an early feature of sarcoidosis [15,111]. Supportive evidence may be seen as patchy air trapping on expiratory CT [112],[113].
In some cases, air trapping, believed to reflect bronchiolar obstruction, foreshadows more typical parenchymal manifestations of sarcoidosis [113]. However, the exact prevalence of this phenomenon and its clinical significance, if any, are as yet unknown. Inflammation and fibrosis involving bronchioles may also be seen in idiopathic pulmonary fibrosis [99,114].
However, the degree of this reaction is not as severe as that seen in chronic hypersensitivity pneumonitis [99] (106). Bronchiolectasis accounts for some of the cystic changes seen by HRCT in idiopathic pulmonary fibrosis [115]. Recently, Yousem and Dacic [111] described another interstitial lung disease with a prominent bronchiolar component that they named idiopathic bronchiolocentric interstitial pneumonia. Histopathologic features are characterized by chronic inflammatory cell infiltrates with a centrilobular and bronchiolocentric distribution [111] . These findings may be similar to hypersensitivity pneumonitis, but there are no interstitial granulomas. Chest radiography demonstrates bibasilar interstitial infiltrates, and restrictive defect is seen on pulmonary function testing [111] . Most patients are middle aged, and there appears to be a predilection for women [111] . Idiopathic bronchiolocentric interstitial pneumonia is associated with a relatively poor prognosis, including 33% mortality at 4 years follow-up [111] .
Bronchiolar Involvement in Large Airway Diseases:
Bronchiectasis, chronic bronchitis/emphysema, cystic fibrosis, and similar conditions of the bronchi typically manifest pathologic changes at the level of the bronchioles that reflect the full spectrum of bronchiolar pathology [13] . Bronchiolar abnormalities found in these large airway diseases include variable degrees of inflammation in the wall and lumen of bronchioles, smooth muscle hypertrophy, and mucostasis [116,117] . Additional histologic findings in asthma may include mucus plugging, bronchiolar epithelial sloughing, luminal and mural eosinophils, and luminal eosinophil debris [117,118] . Correlation was found between the severity of this inflammatory response and the degree of airflow limitation, suggesting a contributory role of small airway inflammation in the development of progressive chronic obstructive pulmonary disease [119] .
Obstruction of bronchioles by inflammatory exudates and bronchiolar wall thickening from edema and smooth muscle hyperplasia occurring in these large airway diseases produce the HRCT scan features ofatelectasis, air trapping, centrilobular nodules, and bronchiolar wall thickening [120] . Thus, air trapping suggestive of bronchiolar disease is not uncommon in patients with bronchiectasis [121] . In some cases of severe asthma and air trapping, HRCT features may be difficult to distinguish from that of constrictive bronchiolitis [122] . Branching or centrilobular nodules (reflecting the presence of dilated bronchioles with mucus impaction, infection, or peribronchiolar inflammation) and mosaic perfusion are seen in patients with cystic fibrosis [123,124]. Similar signs of bronchiolar disease are also seen in patients with allergic bronchopulmonary aspergillosis [125] .
Conclusions
Bronchiolar abnormalities are commonly encountered on lung biopsy specimens and HRCT of the chest. Various histopathologic patterns of bronchiolar injury have been described and have led to confusing nomenclature with redundancies and overlapping terms. There is a need to standardize the terminology and to recognize that bronchiolar disease may not have clinical significance in some clinico–radiologic contexts. Herein, we have presented a scheme to classify those disorders in which the bronchiolar disease can be the predominant (primary bronchiolar disorders) or contributing (i.e., in parenchymal and large airway diseases) pathologic component. When faced with a bronchiolar abnormality the clinician needs to ask three questions: (1) is the bronchiolar abnormality the predominant lesion causing disease? (2) what is the histopathologic pattern of the bronchiolar lesion? (3) what is the clinico–physiologic–radiologic context? The clinical and prognostic significance of a bronchiolar lesion is best determined by identifying the underlying histopathologic pattern and assessing the correlative clinico–physiologic– radiologic context.
Competing interests:
The author(s) declare that they have no competing interests.
References
Mead, J. (1970). The lung's "quiet zone". The New England journal of medicine , 282:1318-9.View
Burgel, P.R., Bergeron, A., de Blic J, Bonniaud P, Bourdin A, Chanez P, Chinet T, Dalphin JC, Devillier P, Deschildre A, Didier A, Kambouchner M, Knoop C, Laurent F, Nunes H, Perez T, Roche N, Tillie-Leblond I, & Dusser D. (2013). Small airways diseases, excluding asthma and COPD: an overview. European respiratory review : an official journal of the European Respiratory Society, 22:131-47.View
Lynch, J.P., 3rd, Weigt, S.S., DerHovanessian, A., Fishbein, M.C., Gutierrez, A., & Belperio, J.A. (2012). Obliterative (constrictive) bronchiolitis. Seminars in respiratory and critical care medicine, 33:509-32.
Schlesinger, C., Meyer, C.A., Veeraraghavan, S., & Koss, M.N. (1998). Constrictive (obliterative) bronchiolitis: diagnosis, etiology, and a critical review of the literature. Annals of diagnostic pathology, 2:321-34.View
Colby, T.V. (1998). Bronchiolitis. Pathologic considerations. American journal of clinical pathology , 109:101-9.View
Muller, N.L., & Miller, R.R. (1995). Diseases of the bronchioles: CT and histopathologic findings. Radiology, 196:3-12.View
Wright, J.L., Cagle, P., Churg, A., Colby, T.V., & Myers, J. (1992). Diseases of the small airways. The American review of respiratory disease, 146:240-62.View
Garg, K., Lynch, D.A., Newell, J.D., King, & T.E., Jr. (1994). Proliferative and constrictive bronchiolitis: classification and radiologic features. AJR American journal of roentgenology, 162:803-8.View
Myers, J.L., & Colby, T.V. (1993). Pathologic manifestations of bronchiolitis, constrictive bronchiolitis, cryptogenic organizing pneumonia, and diffuse panbronchiolitis. Clinics in chest medicine , 14:611-22.View
Colby, T.V., Leslie, K.O., & Samuel, A.Y. (2007). Histology for Pathologists. Mills, Stacey E.(Eds.). Thorax and Serous Membranes: Lungs. Lippincott Williams and Wilkins.View
Bienenstock, J., & McDermott, M.R. (2005). Bronchus- and nasal-associated lymphoid tissues. Immunological reviews, 206:22-31.View
Visscher, D.W., & Myers, J.L. (2006). Bronchiolitis: the pathologist's perspective. Proceedings of the American Thoracic Society, 3:41-7.View
Ryu, J.H., Myers, J.L., & Swensen, S.J. (2003). Bronchiolar disorders. American journal of respiratory and critical care medicine, 168:1277-92.View
Muller, N.L. (2001). Advances in imaging. The European respiratory journal, 18:867-71.
Hansell, D.M. (2001). HRCT of obliterative bronchiolitis and other small airways diseases. Seminars in roentgenology, 36:51-65.View
Franquet, T., & Stern, E.J, (1999). Bronchiolar inflammatory diseases: high-resolution CT findings with histologic correlation. European radiology, 9:1290-303.View
Collins, J., Blankenbaker, D., & Stern, E.J. (1998). CT patterns of bronchiolar disease: what is "tree-in-bud"? AJR American journal of roentgenology ,171:365-70.View
Allen, T.C. (2010). Pathology of small airways disease. Arch Pathol Lab Med , 134:702-18.View
Gunn, M.L., Godwin, J.D., Kanne, J.P., Flowers, M.E., & Chien, J.W. (2008). High-resolution CT findings of bronchiolitis obliterans syndrome after hematopoietic stem cell transplantation. Journal of thoracic imaging, 23:244-50.View
A-LA, K. (2006). Katzenstein A-LA. Miscellaneous II: nonspecific inflammatory and destructive diseases. Katzenstein and Askin’s Surgical Pathology of NonNeoplastic Lung Disease 4th ed Philadelphia, PA: Elsevier; 2006:445–476.:445–76.
Allen, T.C. (2010). Pathology of Small Airways Disease. Archives of Pathology & Laboratory Medicine, 134:702-18.View
Paradis, I., Yousem, S., & Griffith, B. (1993). Airway obstruction and bronchiolitis obliterans after lung transplantation. Clinics in chest medicine, 14:751-63.View
Yousem, S.A., Berry, G.J., Cagle, P.T., Chamberlain, D., Husain, A.N., Hruban, R.H., Marchevsky, A., Ohori, N.P., Ritter, J., Stewart, S., & Tazelaar, H.D. (1996). Revision of the 1990 working formulation for the classification of pulmonary allograft rejection: Lung Rejection Study Group. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation, 15:1-15.View
Bando, K., Paradis, I.L., Similo, S., Konishi, H., Komatsu, K., Zullo, T.G., Yousem, S.A., Close, J.M., Zeevi, A., & Duquesnoy, R.J., et al. (1995). Obliterative bronchiolitis after lung and heart-lung transplantation. An analysis of risk factors and management. The Journal of thoracic and cardiovascular surgery, 110:4-13; discussion -4.View
Berry, G.J., Brunt, E.M., Chamberlain, D., Hruban, R.H., Sibley, R.K., Stewart, S., & Tazelaar, H.D. (1990). A working formulation for the standardization of nomenclature in the diagnosis of heart and lung rejection: Lung Rejection Study Group. The International Society for Heart Transplantation. The Journal of heart transplantation, 9:593-601.View
Todd, J.L., & Palmer, S.M. (2011). Bronchiolitis obliterans syndrome: the final frontier for lung transplantation. Chest, 140:502-8.View
Chang, A.B., Masel, J.P., & Masters, B. (1998). Post-infectious bronchiolitis obliterans: clinical, radiological and pulmonary function sequelae. Pediatric radiology, 28:23-9.View
Penn, C.C., & Liu, C. (1993). Bronchiolitis following infection in adults and children. Clinics in chest medicine, 14:645-54.View
Markopoulo, K.D., Cool, C.D., Elliot, T.L., Lync, D.A., Newell, JD Jr., Hale V.A., Brown, K.K., Schwarz, M.I., & Tuder, R.M. (2002). Obliterative bronchiolitis: varying presentations and clinicopathological correlation. The European respiratory journal, 19:20-30.
Schwartzman, K.J., Bowie, D.M., Yeadon, C., Fraser, R., Sutton, E.D., & Levy, R.D. (1995). Constrictive bronchiolitis obliterans following gold therapy for psoriatic arthritis. The European respiratory journal, 8:2191-3.
Boehler, A., Vogt, P., Speich, R., Weder, W., & Russi, E.W. (1996). Bronchiolitis obliterans in a patient with localized scleroderma treated with D-penicillamine. The European respiratory journal, 9:1317-9.View
Estenne, M., Maurer, J.R., Boehler, A., Egan, J.J., Frost, A., Hertz, M., Mallory, G.B., Snell, G.I., & Yousem, S. (2002). Bronchiolitis obliterans syndrome 2001: an update of the diagnostic criteria. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation, 21:297-310.View
Geddes, D.M., Corrin, B., Brewerton, D.A., Davies, R.J., & Turner-Warwick, M. (1977). Progressive airway obliteration in adults and its association with rheumatoid disease. Q J Med, 46:427-44.View
Epler, G.R., Snider, G.L., Gaensler, E.A., Cathcart, E.S., FitzGerald, M.X., & Carrington, C.B. (1979). Bronchiolitis and bronchitis in connective tissue disease. A possible relationship to the use of penicillamine. Jama, 242:528-32.View
Herzog, C.A., Miller, R.R., & Hoidal, J.R. (1981). Bronchiolitis and rheumatoid arthritis. The American review of respiratory disease, 124:636-9.View
Wells, A.U., & du Bois, R.M. (1993). Bronchiolitis in association with connective tissue disorders. Clinics in chest medicine, 14:655-66.View
Perez, T., Remy-Jardin, M., & Cortet, B. (1998). Airways involvement in rheumatoid arthritis: clinical, functional, and HRCT findings. American journal of respiratory and critical care medicine, 157:1658-65.View
Lucaya, J., Gartner, S., Garcia-Pena, P., Cobos, N., Roca, I., & Linan, S. (1998). Spectrum of manifestations of Swyer-James-MacLeod syndrome. Journal of computer assisted tomography, 22:592-7.View
Respiratory health hazards in agriculture. (1998). American journal of respiratory and critical care medicine, 158:S1-s76.View
Yousem, S.A., Paradis, I.L., Dauber, J.H., & Griffith, B.P. (1989). Efficacy of transbronchial lung biopsy in the diagnosis of bronchiolitis obliterans in heart-lung transplant recipients. Transplantation, 47:893-5.View
Arcasoy, S.M., & Kotloff, R.M. (1999). Lung transplantation. The New England journal of medicine, 340:1081-91.View
Kelly, K., & Hertz, M.I. (1997). Obliterative bronchiolitis. Clinics in chest medicine, 18:319-38.View
Estenne, M., & Hertz, M.I. (2002). Bronchiolitis obliterans after human lung transplantation. American journal of respiratory and critical care medicine, 166:440-4.View
Cooper, J., Billingham, M., Egan, T., Hertz, M., Higenbottam, T., Lynch, J., Mauer, J., Paradis, I., Patterson, G., & Smith, C. (1993). A working formulation for the standardization of nomenclature and for clinical staging of chronic dysfunction in lung allografts. International Society for Heart and Lung Transplantation. The Journal of heart and lung transplantation: the official publication of the International Society for Heart Transplantation , 12:713-6.View
Brown, M.J., English, J., & Muller, N.L. (1997). Bronchiolitis obliterans due to neuroendocrine hyperplasia: high-resolution CT--pathologic correlation. AJR American journal of roentgenology, 168:1561-2.View
Sheerin, N., Harrison, N.K., Sheppard, M.N., Hansell, D.M., Yacoub, M., & Clark, T.J. (1995). Obliterative bronchiolitis caused by multiple tumourlets and microcarcinoids successfully treated by single lung transplantation. Thorax, 50:207-9.
Miller, R.R., & Muller, N.L. (1995). Neuroendocrine cell hyperplasia and obliterative bronchiolitis in patients with peripheral carcinoid tumors. The American journal of surgical pathology, 19:653-8.View
Aguayo, S.M., Miller, Y.E., Waldron, JA Jr., Bogin, R.M., Sunday, M.E., Staton G.W Jr., Beam, W.R., & King TE Jr. (1992). Brief report: idiopathic diffuse hyperplasia of pulmonary neuroendocrine cells and airways disease. The New England journal of medicine, 327:1285-8.
Nousari, H.C., Deterding, R., Wojtczack, H., Aho, S., Uitto, J., Hashimoto, T., & Anhalt, G.J. (1999). The mechanism of respiratory failure in paraneoplastic pemphigus. The New England journal of medicine, 340:1406-10.View
Mahadeva, R., Walsh, G., Flower, C.D., & Shneerson, J.M. (2000). Clinical and radiological characteristics of lung disease in inflammatory bowel disease. The European respiratory journal, 15:41-8.
Wilcox, P., Miller, R., Miller, G., Heath, J., Nelems, B., Muller, N., & Ostrow, D. (1987). Airway involvement in ulcerative colitis. Chest, 92:18-22.View
Boswell, R.T., & McCunney, R.J. (1995). Bronchiolitis obliterans from exposure to incinerator fly ash. Journal of occupational and environmental medicine, 37:850-5.View
Wang, J.S., Tseng, H.H., Lai, R.S., Hsu, H.K., & Ger, L.P. (2000). Sauropus androgynus-constrictive obliterative bronchitis/ bronchiolitis--histopathological study of pneumonectomy and biopsy specimens with emphasis on the inflammatory process and disease progression. Histopathology, 37:402-10.View
Reynaud-Gaubert, M., Marin, V., Thirion, X., Farnarier, C., Thomas, P., Badier, M., Bongrand, P., Giudicelli, R., & Fuentes, P. (2002). Upregulation of chemokines in bronchoalveolar lavage fluid as a predictive marker of post-transplant airway obliteration. The Journal of heart and lung transplantation : the official publication of the International Society for Heart Transplantation, 21:721-30.
Belperio, J.A., Keane, M.P., Burdick, M.D., Lynch, J.P. 3rd, Xue, Y.Y., Li, K., Ross, D.J., & Strieter, R.M. (2002). Critical role for CXCR3 chemokine biology in the pathogenesis of bronchiolitis obliterans syndrome. Journal of immunology (Baltimore, Md : 1950), 169:1037-49.View
Kudoh, S., Azuma, A., Yamamoto, M., Izumi, T., & Ando, M. (1998). Improvement of survival in patients with diffuse panbronchiolitis treated with low-dose erythromycin. Am J Respir Crit Care Med, 157:1829-32.View
Desai, S.R., & Hansell, D.M. (1197). Small airways disease: expiratory computed tomography comes of age. Clin Radiol, 52:332-7.
Stern, E.J., Muller, N.L., Swensen, S.J., & Hartman, T.E. (1995). CT mosaic pattern of lung attenuation: etiologies and terminology. Journal of thoracic imaging, 10:294-7.View
Kindt, G.C., Weiland, J.E., Davis, W.B., Gadek, J.E., & Dorinsky, P.M. (1989). Bronchiolitis in adults. A reversible cause of airway obstruction associated with airway neutrophils and neutrophil products. The American review of respiratory disease, 140:483-92.View
Hall, C.B. (2001). Respiratory syncytial virus and parainfluenza virus. The New England journal of medicine, 344:1917-28.View
Andersen, P. (1998). Pathogenesis of lower respiratory tract infections due to Chlamydia, Mycoplasma, Legionella and viruses. Thorax, 53:302-7.View
Schlesinger, C., & Koss, M.N. (2002). Bronchiolitis: update 2001. Current opinion in pulmonary medicine, 8:112-6.View
Kim, C.K., Kim, S.W., Kim, J.S., Koh, Y.Y., Cohen, A.H., Deterding, R.R., & White, C.W. (2001). Bronchiolitis obliterans in the 1990s in Korea and the United States. Chest, 120:1101-6.
Cagle, P.T., Allen, T.C., Dacic, S., Beasley, M.B., Borczuk, A.C., Chirieac, L.R., Laucirica, R., Ro, J.Y., & Kerr, K.M. (2011). Revolution in Lung Cancer: New Challenges for the Surgical Pathologist. Archives of Pathology & Laboratory Medicine, 135:110-6.View
Hansell, D.M. (2001). Small airways diseases: detection and insights with computed tomography. European Respiratory Journal, 17:1294-313.View
Visscher, D.W., & Myers, J.L. (2006). Bronchiolitis. Proceedings of the American Thoracic Society, 3:41-7.View
Raphael, J., Collins, S.R., Wang, X-Q., Scalzo, D.C., Singla, P., Lau, C.L., Kozower, B.D., Durieux, M.E., & Blank, R.S. (2017). Perioperative statin use is associated with decreased incidence of primary graft dysfunction after lung transplantation. The Journal of Heart and Lung Transplantation, 36:948-56.View
Ryu, J.H. (2006). Classification and approach to bronchiolar diseases. Current opinion in pulmonary medicine, 12:145-51.View
Panitch, H.B. (2001). Bronchiolitis in infants. Current opinion in pediatrics, 13:256-60.View
Aherne, W., Bird, T., Court, S.D., Gardner, P.S., & McQuillin, J. (1970). Pathological changes in virus infections of the lower respiratory tract in children. Journal of clinical pathology, 23:7- 18.View
Harrison, A.M., Bonville, C.A., Rosenberg, H.F., & Domachowske, J.B. (1999). Respiratory syncytical virus-induced chemokine expression in the lower airways: eosinophil recruitment and degranulation. American journal of respiratory and critical care medicine, 159:1918-24.View
Sugiyama, Y. (1993). Diffuse panbronchiolitis. Clinics in chest medicine, 14:765-72.View
Kudoh, S. (1998). Erythromycin treatment in diffuse panbronchiolitis. Current opinion in pulmonary medicine, 4:116-21.View
Fitzgerald, J.E., King, T.E. Jr., Lynch, D.A., Tuder, R.M., & Schwarz, M.I. (1996). Diffuse panbronchiolitis in the United States. Am J Respir Crit Care Med, 154:497-503.View
Kadota, J., Mukae, H., Tomono, K., & Kohno, S. (2001). High concentrations of beta-chemokines in BAL fluid of patients with diffuse panbronchiolitis. Chest, 120:602-7.View
Todate, A., Chida, K., Suda, T., Imokawa, S., Sato, J., Ide, K., Tsuchiya, T., Inui, N., Nakamura, Y., Asada, K., Hayakawa, H., & Nakamura, H. (2000). Increased numbers of dendritic cells in the bronchiolar tissues of diffuse panbronchiolitis. American journal of respiratory and critical care medicine, 162:148-53.View
Iwata, M., Colby, T.V., & Kitaichi, M. (1994). Diffuse panbronchiolitis: diagnosis and distinction from various pulmonary diseases with centrilobular interstitial foam cell accumulations. Human pathology, 25:357-63.
Homma, S., Kawabata, M., Kishi, K., Tsuboi, E., Narui, K., Nakatani, T., Uekusa, T., Saiki, S., & Nakata, K. (1998). Diffuse panbronchiolitis in rheumatoid arthritis. The European respiratory journal, 12:444-52.View
Takizawa, H., Desaki, M., Ohtoshi, T., Kawasaki, S., Kohyama, T., Sato, M., Tanaka, M., Kasama, T., Kobayashi, K., Nakajima, J., & Ito, K. (1997). Erythromycin modulates IL-8 expression in normal and inflamed human bronchial epithelial cells. American journal of respiratory and critical care medicine, 156:266-71.View
Yanagihara, K., Kadoto, J., & Kohno, S. (2001). Diffuse panbronchiolitis--pathophysiology and treatment mechanisms. International journal of antimicrobial agents, 18 Suppl 1:S83- 7.View
Fujii, T., Kadota, J., Kawakami, K., Iida, K., Shirai, R., Kaseda, M., Kawamoto, S., & Kohno, S. (1995). Long term effect of erythromycin therapy in patients with chronic Pseudomonas aeruginosa infection. Thorax, 50:1246-52.View
Baz, M.A., Kussin, P.S., Van Trigt, P., Davis, R.D., Roggli, V.L., & Tapson, V.F. (1995). Recurrence of diffuse panbronchiolitis after lung transplantation. American journal of respiratory and critical care medicine, 151:895-8.View
Thurlbeck, W.M. (1980). Smoking, airflow limitation, and the pulmonary circulation. American Lung Association.View
Nagai, A., West, W.W., Paul, J.L., & Thurlbeck, W.M. (1985). The National Institutes of Health Intermittent Positive-Pressure Breathing trial: pathology studies. I. Interrelationship between morphologic lesions. The American review of respiratory disease, 132:937-45.View
Niewoehner, D.E., Kleinerman, J., & Rice, D.B. (1974). Pathologic changes in the peripheral airways of young cigarette smokers. The New England journal of medicine, 291:755-8.View
Moon, J., du Bois, R.M., Colby, T.V., Hansell, D.M., & Nicholson, A.G. (1999). Clinical significance of respiratory bronchiolitis on open lung biopsy and its relationship to smoking related interstitial lung disease. Thorax, 54:1009-14. View
Gardiner, K., Trethowan, N.W., Harrington, J.M., Rossiter, C.E., & Calvert, I.A. (1993). Respiratory health effects of carbon black: a survey of European carbon black workers. Occupational and Environmental Medicine, 50:1082-96.View
Howling, S.J., Hansell, D.M., Wells, A.U., Nicholson, A.G., Flint, J.D., & Muller, N.L. (1999). Follicular bronchiolitis: thin-section CT and histologic findings. Radiology, 212:637-42. View
Tashtoush, B., Okafor, N.C., Ramirez, J.F., & Smolley, L. (2015). Follicular Bronchiolitis: A Literature Review. Journal of clinical and diagnostic research : JCDR, 9:Oe01-5. View
Koss, M.N. (1995). Pulmonary lymphoid disorders. Seminars in diagnostic pathology, 12:158-71.View
Oh, Y.W., Effmann, E.L., Redding, G.J., & Godwin, J.D. (1999). Follicular hyperplasia of bronchus-associated lymphoid tissue causing severe air trapping. AJR American journal of roentgenology, 172:745-7.View
Fortoul, T.I., Cano-Valle, F., Oliva, E., & Barrios, R. (1985). Follicular bronchiolitis in association with connective tissue diseases. Lung, 163:305-14.View
Kinoshita, M., Higashi, T., Tanaka, C., Tokunaga, N., Ichikawa, Y., & Oizumi, K. (1992). Follicular bronchiolitis associated with rheumatoid arthritis. Internal medicine (Tokyo, Japan), 31:674-7.View
Kinane, B.T., Mansell, A.L., Zwerdling, R.G., Lapey, A., & Shannon, D.C. (1993). Follicular bronchitis in the pediatric population. Chest, 104:1183-6.View
Couture, C., & Colby, T.V. (2003). Histopathology of bronchiolar disorders. Seminars in respiratory and critical care medicine, 24:489-98.View
Hayakawa, H., Sato, A., Imokawa, S., Toyoshima, M., Chida, K., & Iwata, M. (1996). Bronchiolar disease in rheumatoid arthritis. American journal of respiratory and critical care medicine, 154:1531-6.View
Patel, A.M., Ryu, J.H., & Reed, C.E. (2001). Hypersensitivity pneumonitis: current concepts and future questions. The Journal of allergy and clinical immunology, 108:661-70. View
Wright, J.L. (1993). Inhalational lung injury causing bronchiolitis. Clinics in chest medicine, 14:635-44.View
Perez-Padilla, R., Gaxiola, M., Salas, J., Mejia, M., Ramos, C., & Selman, M. (1996). Bronchiolitis in chronic pigeon breeder's disease. Morphologic evidence of a spectrum of small airway lesions in hypersensitivity pneumonitis induced by avian antigens. Chest, 110:371-7.View
Reyes, C.N., Wenzel, F.J., Lawton, B.R., & Emanuel, D.A. (1982). The pulmonary pathology of farmer's lung disease. Chest, 81:142-6.View
Selman, M., & Vargas, M.H. (1998). Airway involvement in hypersensitivity pneumonitis. Current opinion in pulmonary medicine, 4:9-15.View
Ryu, J.H., Colby, T.V., Hartman, T.E., & Vassallo, R. (2001). Smoking-related interstitial lung diseases: a concise review. The European respiratory journal, 17:122-32.View
Wells, A.U. (2001). Cryptogenic organizing pneumonia. Seminars in respiratory and critical care medicine, 22:449-60.View
Cordier, J.F. (2000). Organising pneumonia. Thorax, 55:318-28.View
American Thoracic Society/European Respiratory Society International Multidisciplinary Consensus Classification of the Idiopathic Interstitial Pneumonias (2002). This joint statement of the American Thoracic Society (ATS), and the European Respiratory Society (ERS) was adopted by the ATS board of directors, June 2001 and by the ERS Executive Committee, June 2001. American journal of respiratory and critical care medicine, 165:277-304.View
Lohr, R.H., Boland, B.J., Douglas, W.W., Dockrell, D.H., Colby, T.V., Swensen, S.J., Wollan, P.C., & Silverstein, M.D. (1997). Organizing pneumonia. Features and prognosis of cryptogenic, secondary, and focal variants. Archives of internal medicine, 157:1323-9.View
Myers, J.L., & Aubry, M.C. (2002). Pulmonary langerhans cell histiocytosis: what was the question? American journal of respiratory and critical care medicine, 166:1419-21.View
Kambouchner, M., Basset, F., Marchal, J., Uhl, J.F., Hance, A.J., & Soler, P. (2002). Three-dimensional characterization of pathologic lesions in pulmonary langerhans cell histiocytosis. American journal of respiratory and critical care medicine, 166:1483-90.View
Vassallo, R., Ryu, J.H., Colby, T.V., Hartman, T., & Limper, A.H. (2000). Pulmonary Langerhans'-cell histiocytosis. The New England journal of medicine, 342:1969-78.View
Statement on sarcoidosis (1999). Joint Statement of the American Thoracic Society (ATS), the European Respiratory Society (ERS) and the World Association of Sarcoidosis and Other Granulomatous Disorders (WASOG) adopted by the ATS Board of Directors and by the ERS Executive Committee, February 1999. American journal of respiratory and critical care medicine, 160:736-55.View
Yousem, S.A., & Dacic, S. (2002). Idiopathic bronchiolocentric interstitial pneumonia. Modern pathology : an official journal of the United States and Canadian Academy of Pathology, Inc, 15:1148-53.View
Hansell, D.M., Milne, D.G., Wilsher, M.L., & Wells, A.U. (1998). Pulmonary sarcoidosis: morphologic associations of airflow obstruction at thin-section CT. Radiology, 209:697-704.View
Davies, C.W., Tasker, A.D., Padley, S.P., Davies, R.J., & Gleeson, F.V. (2000). Air trapping in sarcoidosis on computed tomography: correlation with lung function. Clin Radiol, 55:217-21.View
Fulmer, J.D., Roberts, W.C., von Gal, E.R., & Grystal, R.G. (1977). Small airways in idiopathic pulmonary fibrosis. Comparison of morphologic and physiologic observations. The Journal of clinical investigation, 60:595-610.View
Johkoh, T., Muller, N.L., Ichikado, K., Yoshida, S., Honda, O., Mihara, N., Higashi, M., Tomiyama, N., Nakamura, H., & Nagareda, T. (1999). Respiratory change in size of honeycombing: inspiratory and expiratory spiral volumetric CT analysis of 97 cases. Journal of computer assisted tomography, 23:174-80.View
Aubry, M.C., Wright, J.L., & Myers, J.L. (2000). The pathology of smoking-related lung diseases. Clinics in chest medicine, 21:11-35, vii.View
Hogg, J.C. (1993). Bronchiolitis in asthma and chronic obstructive pulmonary disease. Clinics in chest medicine, 14:733-40.View
Shaw, R.J., Djukanovic, R., Tashkin, D.P., Millar, A.B., du Bois, R.M., & Orr, P.A. (2002). The role of small airways in lung disease. Respiratory medicine, 96:67-80.View
Turato, G., Zuin, R., Miniati, M., Baraldo, S., Rea, F., Beghe, B., Monti, S., Formichi, B., Boschetto, P., Harari, S., Papi, A., Maestrelli, P., Fabbri, L.M., & Saetta, M. (2002). Airway inflammation in severe chronic obstructive pulmonary disease: relationship with lung function and radiologic emphysema. American journal of respiratory and critical care medicine, 166:105-10.View
Bousquet, J., Jeffery, P.K., Busse, W.W., Johnson, M., & Vignola, A.M. (2000). Asthma: from bronchoconstriction to airways inflammation and remodeling. American journal of respiratory and critical care medicine, 161:1720-45. View
Hansell, D.M., Wells, A.U., Rubens, M.B., & Cole, P.J. (1994). Bronchiectasis: functional significance of areas of decreased attenuation at expiratory CT. Radiology, 193:369-74.View
Jensen, S., Lynch, D., Brown, K., Wenzel, S., & Newell, J. (2002). High-resolution CT features of severe asthma and bronchiolitis obliterans. Clinical radiology, 57:1078-85. View
Lynch, D.A., Brasch, R.C., Hardy, K.A., & Webb, W.R. (1990). Pediatric pulmonary disease: assessment with high-resolution ultrafast CT. Radiology, 176:243-8.View
Kuhn, J.P., & Brody, A.S. (2002). High-resolution CT of pediatric lung disease. Radiologic clinics of North America, 40:89-110.View
McGuinness, G., & Naidich, D.P. (1985). CT of airways disease and bronchiectasis. Radiologic clinics of North America 2002, 40:1-19.[126] Epler GR, Colby TV, McCloud TC, Carrington CB, Gaensler EA. Bronchiolitis obliterans organizing pneumonia. N Engl J Med;312:152–158.View
Lappi-Blanco, E., Soini, Y., Kinnula, V., & Paakko P. (2002). VEGF and bFGF are highly expressed in intraluminal fibromyxoid lesions in bronchiolitis obliternas organizing pneumonia. J Pathol;196:220–227.