Journal of Public Health Issues and Practices Volume 3 (2019), Article ID: JPHIP-132
https://doi.org/10.33790/jphip1100132Review Article
Effect of NSAIDs on Muscle Adaptations to Resistance Training: A Brief Review
Ahmad Salamah1, Junchul Shin2, Joon-Young Park2*
1Department of Kinesiology, College of Public Health, Temple University, 1800 N. Broad Street, Philadelphia, PA 19122, U.S.A
2Cardiovascular Research Center, Lewis Katz School of Medicine, Temple University, 3500 N. Broad Street, Philadelphia, PA 19140, U.S.A.
Corresponding Author Details: Joon-Young Park, Department of Kinesiology, College of Public Health; Cardiovascular Research Center, Lewis Katz School of Medicine, 1052 MERB, 3500 N. Broad St., Philadelphia, PA 19140, U.S.A. E-mail: parkjy@ temple.edu
Received date: 06th November, 2018
Accepted date: 21st January, 2019
Published date: 24th January, 2019
Citation: Salamah, A., Shin, J.C., & Park, J.Y. (2019). Effect of NSAIDs on Muscle Adaptations to Resistance Training: A Brief Review. J Pub Health Issue Pract 3: 132.
Copyright: ©2019, This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Non-steroidal anti-inflammatory drugs (NSAIDs) are popular amongst various populations to treat a myriad of ailments. One common use is to lessen delayed onset muscle soreness (DOMS) after intense exercise, generally prevalent in eccentric muscle contractions. To date, many studies have been conducted investigating the effects of NSAIDs on skeletal muscle hypertrophy and function. Existing literature reports very different results and conclusions with respect to the effects of these drugs on resistance training-induced adaptations in skeletal muscle. To best answer the question concerning NSAIDs and functional muscle hypertrophy, this review outlines literature and categorizes various factors which influence study outcomes. Although the literature examining the topic is conflicting, the results seem to vary heavily based on the subjects’ characteristics, dosages, and resistance training protocols. Specifically, the age can be a pivotal factor in determining the response seen from supplementing with NSAIDs. In conclusion, NSAIDs may be a smart approach to DOMS for older populations, while younger individuals may be limiting exercise-induced skeletal muscle adaptations.
Keywords: NSAID, resistance training, muscle hypertrophy
Abbreviations:
NSAIDs: non-steroidal anti-inflammatory drugs
DOMS: delayed onset muscle soreness
COX: cyclooxygenase
PGH2: prostaglandin H2
PGF2α: prostaglandin F2α
PGE2: prostaglandin E2
ROS: reactive oxygen species
FSR: fractional synthesis rate
Introduction
Delayed onset muscle soreness (DOMS) is described as the stiffness, pain, or tenderness that exists 1-7 days after a bout of intense exercise [1]. DOMS is believed to be caused by trauma, which damages the associated myofibrils. This microtrauma then leads to a local inflammatory response, and with it, a dull soreness. This inflammatory process is a naturally occurring muscle repair mechanism leading to the structural and functional adaptations of skeletal muscle to resistance training. However, when uncontrolled, it can cause excess tissue damage and muscle atrophy, and ultimately interfere with normal muscle growth. Generally, DOMS is looked at as a negative effect of resistance training, both subjectively and objectively. In fact, DOMS has been shown to significantly decrease strength/power output, which may be detrimental to athletic performance [2].
To mitigate the pain from the soreness (DOMS), many individuals choose to use pharmaceutical interventions, such as non-steroidal anti-inflammatory drugs (NSAIDs). Approximately 30 million people worldwide are believed to take some form of these drugs every day [3]. Popular over-the-counter options include ibuprofen, aspirin, and naproxen, with stronger options available by prescription. Because these drugs are fast acting and typically out of the system within 24 hours, many individuals rely on them for daily use. The underlying mechanisms by which NSAIDs work may, however, interfere with adaptations related to resistance training.
To date, there is a considerable number of experimental studies aiming to investigate whether the use of NSAIDs during following exercise interferes with or facilitates normal muscle hypertrophy. However, the results are somewhat mixed and largely inconclusive. This mini-review focuses on the major variables and other confounding factors affecting the association of NSAID use and muscle hypertrophy including age, dosage, exercise modality, and training history. In addition, principles of muscle damage, muscle inflammation and acting mechanisms of NSAIDs will be discussed.
Response to tissue damage caused by resistance training
Mechanism of muscle damage
Damage to muscle tissue following various types of physical exercise has been well-established [4]. There are three types of muscle contraction that occur during exercise: concentric, eccentric, and isometric. Concentric contractions occur when a muscle produces force and the length of muscle shortens. Eccentric contractions occur when a muscle produces force as it lengthens, due to external forces exceeding those produced by the muscle. Finally, isometric contractions involve a muscle producing force while maintaining a static length. Eccentrically contracting muscles can generally resist more force than they are able to concentrically produce. Indeed, eccentric contractions seem to produce the most damage when compared to concentric or isometric contractions [5]. This may be due to the fact that fewer motor units are activated during the eccentric muscle contraction, exerting more stress on the muscle fibers involved. This increased stress may cause damaged T-tubules, leading to inactivation of certain sarcomeres. Although there is newer conflicting evidence concerning whether eccentric muscle contractions lead to greater hypertrophy [6], there is a general agreement that muscle damage caused by loaded stretching is a potent stimulator of hypertrophy. Unsurprisingly, various strategies are utilized in almost every resistance training regimen to drive adaptations associated with such damage.
There are several structural indicators of muscle damage following exercise, which may help explain why damage is a distinct driver of hypertrophy. Fridén et al., was the first to show that exercise causes Z-line streaming, where they are broken down and have disrupted structure [7]. Later studies suggest that the Z-lines are the weak points of the myofibrillar chain, with the anchoring and connecting proteins (such as titin and desmin, respectively) being compromised. Such disruptions to the Z-lines can then cause overstretched full and half sarcomeres, which are no longer at optimal lengths for contraction. Such damage to the sarcomeres and contractile elements then leads to a local inflammatory response, which is the start of the healing process (Refer to an article by Proske & Morgan, 2001 [8] for more on structural damage). Moreover, damage to the sarcolemma has been shown to be accompanied by increased calcium concentration in the cell [9]. The increased intracellular calcium activates several proteolytic enzymes, which can further degrade the Z-discs and proteins within the muscle fiber.
Inflammatory response in skeletal muscle fibers
Arachidonic acid metabolites are crucial for regulating inflammation. Arachidonic acid is polyunsaturated fatty acid that resides abundantly in the cell plasma membrane. There are a wide range of arachidonic acid derivatives that are synthesized by specific synthase enzymes. Notably, prostaglandins are formed from arachidonic acid that are key mediators of inflammation through the cyclooxygenase (COX) pathway. The COX enzyme catalyzes the reaction in producing prostaglandin H2 (PGH2) The concentration of COX is increased significantly following exercise in the muscle. PGH2 is further processed to prostaglandin F2α (PGF2α) and prostaglandin E2 (PGE2) by PGF2α synthase and PGE2 synthase, respectively. Thus, COX enzyme can promote muscle production of these two prostaglandins following exercise.
In skeletal muscle, PGF2α instigates muscle protein synthesis via a phosphoinositide 3-kinase/extracellular receptor kinase/mammalian target of rapamycin-dependent (PI3K/ERK/mTOR) pathway in a PGF2α receptor-dependent manner [10]. A study showed that inhibition of PGF2α largely reduces protein synthesis and muscle hypertrophy, indicating that PGF2α is important in promoting muscle cell growth [11]. In contrast, PGE2 increases the presence of inflammatory signals, such as neutrophils, cytkines, and myokines. For instance, PGE2, has been shown to increase IL-6 transcription, which is a proinflammatory cytokine associated with increased muscle atrophy [12]. PGE2 also stimulates muscle RING finger protein -1 (MuRF- 1) production, which is a proteolytic ubiquitin ligase contributing to protein degradation in the atrophic muscle. Rodemann and Goldberg (1982) demonstrated that incubating muscles with varying concentration of arachidonic acid that increased prostaglandin concentration, which then led to increased muscle protein synthesis and protein degradation [13]. Together, it is important to note that arachidonic acid is important in both muscle growth and atrophy.
Neutrophil counts increase by over 30% immediately following exercise, with levels reaching 86% higher one hour following downhill running [14]. Leukocyte concentrations showed a similarly significant trend. In addition, myoglobin levels increased by 1800% and creatine kinase activity by 420% post-exercise. While it is clear that muscle damage occurred, perhaps an important conclusion of this particular study was that indicators of neutrophil activation, as well as their reactive oxygen species (ROS) production rate, were unchanged. Peake et al. (2005) suggests that this may be due to the subjects’ higher training experience and exercise protocol, as previous evidence has shown an increased oxidative tolerance in trained individuals. On the contrary, a large number of studies have produced opposing results. Neutrophil-derived ROS increases following eccentric resistance training [15]. Neutrophils that infiltrate the tissue can also further the physiological destruction through phagocytosis, degranulation, and reactive oxygen species production [16]. The ROS produced by the neutrophils can lead to the synthesis of additional cytokines, which continue the process. It should be noted that cytokine regulation is complex and involves several up and downstream processes that influence the level of local inflammation [17]. Regardless, there is a clear exercise-induced inflammatory pathway involving the elevation of specific biomarkers.
Mechanism of action for NSAIDs
NSAIDs work by inhibiting the COX enzymes, which are responsible for the formation of prostaglandins and thromboxone from arachidonic acid. More specifically, the COX-1 isoform seems to be more implicated in maintaining various normal physiological functions, such as maintaining the stomach lining, and can be found readily in the body. The COX-2 isoform, however, is associated with the inflammatory response following damage [18]. Furthermore, COX-2 has been implicated as a crucial component for satellite cell proliferation, suggesting its importance to myogenesis. It is unclear, however, if COX-2 is induced by the damage related to resistance training in humans [19]. Different types of NSAIDs will affect these isoforms differently, potentially producing varying responses. The first category of NSAIDs are selective COX-1 inhibitors, which includes aspirin. The second category are non-selective inhibitors, which includes ibuprofen. NSAIDs that selectively inhibit COX-2 enzymatic activity are the least common, with Celebrex being the only one available in the United States. Most studies examine the effects of non-specific COX inhibitors (e.g. ibuprofen) and such will be the focus of this review paper. Interestingly, there is evidence that the dosage of NSAIDs can be manipulated to produce an analgesic or anti-inflammatory response. This introduces yet another variable that may impact the action of NSAIDs, in response to pain or injury. As mentioned above, inhibiting COX enzymatic activity can then decrease the level of prostaglandins and associated inflammatory response. In addition to finding the important role of prostaglandins in muscle synthesis/degradation, Rodemann and Goldberg (1982) also noted that the introduction of a non-selective COX inhibitor to the experimental setup mitigated the upregulating effects of the arachidonic acid [13].
An important note is the inclusion of acetaminophen (e.g. Tylenol) in this category of drugs. Although it is sometimes thought of as an NSAID due to its analgesic properties, acetaminophen is technically not classified as one, with earlier research suggesting that acetaminophen works at the neural level. Thus, it does not impact many of the local or systemic inflammatory biomarkers associated with the response. These pharmacodynamics are being challenged, however, with evidence growing that acetaminophen does impact local inflammatory markers. For instance, acetaminophen has been proposed to have an inhibitory effect on peripheral COX-2 through a different, unknown, mechanism of action [20]. Thus, many studies examining the effect of NSAIDs on training induced adaptations use acetaminophen as well as ibuprofen.
Factors affecting NSAIDs effects on skeletal muscle adaptations
Age
Age seems to be a key variable in determining the effects brought on by NSAIDs, with different results occurring in different populations. The NSAIDs are thought to inhibit production of PGE2, which is involved in muscle atrophy and muscle inflammation as described in the previous section. NSAIDs seem to have widely different effects amongst different age groups yet the underlying mechanisms are unknown. Trappe's group reported that daily ibuprofen or acetaminophen consumption increased muscle volume and strength in older adults [21]. In this study, thirty-six older subjects (mean: 61 ± 1 yr; range: 60 - 78 yr of age) were split into ibuprofen (1,200 mg daily over three dosages), acetaminophen (4,000 mg daily over three dosages), or placebo groups. The subjects performed three sessions of lower body resistance training, consisting of leg extensions, for 12 weeks. Surprisingly, muscle volume increased to a greater extent than the placebo group by ~35%. Upon further analysis of muscle biopsies, levels of the COX enzymes were not affected by these drugs in the worked muscle, with COX-1 levels increasing across all groups.
The same group followed these findings, specifically implicating acetaminophen as a potential enhancer of hypertrophy in older adults [22]. The researchers wanted to focus on the effects of daily acetaminophen intake on type I and type II fibers, separately. The experimental procedure was identical to the previous study, with dosage, resistance training protocol, and testing procedure being the same. It is important to note that the subjects were the same that Trappe used in previous studies that resulted in positive effects from NSAIDs [12,21]. Type I fibers did not change their sizes in the placebo group, while increasing by 28% in the acetaminophen group.
Type II fibers also followed this trend, with the placebo group showing 26% growth and the acetaminophen group growing by 37%. This suggests that the COX pathway that regulates PGE2 production may be more active for a variety of reasons, including enzymatic activity, receptor density, and downstream signaling. The authors also report an increase in the metabolic enzyme, citrate synthase, for the acetaminophen group compared to pre-training values. This suggests that prostaglandins are involved in the regulation of protein turnover for sarcoplasmic, mitochondrial, and myofibrillar proteins.
Other studies have also shown the similar positive effects of NSAIDs, in terms of adaptations to resistance training. Baldwin, Stevenson, and Dudley (2001) reported that daily naproxen sodium ingestion by older subjects (60 yrs of age) resulted in decreased soreness, impairment on strength, and muscle damage determined by MRI [23]. The participants took either 660 mg of naproxen or a placebo for 10 days following unilateral eccentric leg extension training. After allowing for a 3-week washout period, the same procedure was repeated with the contralateral leg. Results showed that the drug caused significant improvements in muscle soreness, and consequently, less strength loss three days following the exercise session. Muscle damage index was 35% higher for the placebo group ten days following the exercise. This may suggest that the participants using naproxen had a decreased inflammatory response, which may lead to decreased secondary damage to the muscle fibers. Although the single exercise session was not enough to produce significant changes in muscle cross-sectional area, these results further implicate NSAIDs as a useful aid in older populations.
In another study, a resistance training protocol did not show any advantage in taking NSAIDs in postmenopausal women [24]. The consumption of 400 mg ibuprofen did not produce significant changes in muscle size, when taken immediately following whole-body resistance training, compared to placebo. Subjects with an average age of 57.8 yr were asked to complete the resistance training protocol 3 days per week for 9 weeks. There were essentially no differences between the ibuprofen and placebo groups, when looking at lean tissue, muscle thickness, and strength. It should be noted that the dosage in this study was lower compared to that which has been shown to have positive effects in older populations. A study by Petersen et al. (2011) showed similar results [25]. In the study, 36 participants (Mean age: 62.3 yr; range: 50-70 yr of age) were split into either an ibuprofen, glucosamine, or placebo group, and all performed three sessions per week of resistance training (leg press and leg extension) for 36 weeks. The study showed that 1,200 mg of ibuprofen daily did not alter quadriceps hypertrophy, as cross-sectional area increased similarly for all three groups. Somewhat counterintuitively, dynamic and isometric strength was shown to increase more in the ibuprofen group compared to the placebo. In addition, results unique to the ibuprofen group included decreased c-reactive protein levels (unchanged in other groups) and unchanged levels of cholesterol (decreased in other groups). It can be concluded, then, that ibuprofen intake may influence certain inflammatory markers but does not necessarily manifest through increased hypertrophy.
In younger muscles, NSAIDs tend to show decreased hypertrophic adaptations to resistance training. For example, Dudley et al. (1997) showed in young male adults (No age data was provided) that 660 mg of naproxen sodium hindered the acute hypertrophy associated with eccentric leg extensions [26]. In this study, each participant performed 10 sets of 7-10 repetitions at 85% of their measured 1-repetition maximum. In this cross-over design, they were given the naproxen drug or placebo to take every day for 10 days and a 2-week washout period ensued, before the other leg was given the remaining treatment. Although the participants in the drug group displayed diminished hypertrophy, they did show less of a strength decrement in 4 days after the exercise protocol. At day 4, strength levels were only 24% below baseline for the naproxen sodium group, while the same measurements were 45% below the baseline for the placebo group. Similarly, ratings of soreness were increased at day 4 for the placebo group, while soreness continually decreased for those in the experimental group. These may be important details, since certain populations, like athletes, may be more concerned with subjective feelings of discomfort and the subsequent strength levels rather than hypertrophy.
In addition, Trappe et al. (2001) provided a more in-depth look at the physiology underlying such differences. Twenty-four young males (mean age was 25 ± 3 yr) were divided into a placebo, ibuprofen, or acetaminophen and underwent 10 sets of 10-14 eccentric leg extensions at 120% of their concentric 1 repetition maximum. In the drug groups, maximum recommended daily dosages were consumed immediately following exercise. In muscle biopsy samples, PGF2α levels for the ibuprofen and acetaminophen groups remained unchanged, while increasing in the placebo. PGE2 levels actually decreased from baseline levels in the acetaminophen group, although they were still non-significantly less for the ibuprofen group compared to placebo. These results are partly expected, as COX inhibition should decrease the production of these prostaglandins. Unfortunately, it is hard to explain the unique effects of acetaminophen and why ibuprofen did not result in the same decreases. The physiology behind the extent to which each prostaglandin is reduced in skeletal muscle is largely unknown.
Dosage
Most studies, especially those that examine the relatively acute effects, tend to use dosages closer to the maximum recommended daily dose. For ibuprofen, that is 1,200 mg per day, while it is 4,000 mg for acetaminophen. Generally, studies show that ibuprofen consumption does decrease markers of muscle damage following a resistance training protocol. Pizza et al. (1999) showed that acute consumption of ibuprofen decreased creatine kinase levels, following one-arm eccentric training [27]. In this study, ten untrained subjects (no age data provided) were given 2,400 mg of ibuprofen per day for 5 days prior to exercise and during a 10-day recovery period following 25 forced eccentric biceps contractions. Interestingly, there was no effect on neutrophil response. This is contrary to other findings and the intuitive conclusion that neutrophil response may be impaired while using ibuprofen.
Building off these preliminary results, Trappe et al. (2002) found that ibuprofen blunted the fractional synthesis rate (FSR) by 41% in eccentrically trained muscle, when compared to placebo [28]. Twenty-four untrained subjects (25 ± 3 yr of age) were randomly assigned into three groups: ibuprofen, acetaminophen, and placebo. Each subject performed 10-14 sets of 10 reps of supramaximal singleleg eccentric leg extensions. Those in the ibuprofen group took 1,200 mg (the maximal recommended daily dosage) for 10 days following the resistance exercise training, while those in the acetaminophen group took 4,000 mg. Surprisingly, creatine kinase levels amongst the groups were similar to the exercise group, although it appears that the levels in the ibuprofen group may show a non-significant decrease. This may be due, at least in part, to the subjects not taking the drug prior to performing the exercise. The main takeaway, however, is that the FSR was unchanged in the acetaminophen and ibuprofen groups, while increasing significantly in the placebo group. Upon further examination, this response is in accordance with PGF2α levels. This shows that short-term ibuprofen consumption following resistance training decreases protein synthesis at the cellular level, which may lead to decreased hypertrophy in the longer-term.
One of the few studies using a submaximal dosage showed little effects of NSAIDs on muscle hypertrophy. Krentz et al. (2008) studied eighteen young resistance training subjects (24.1 ± 0.6 yr of age) who ingested 400 mg of ibuprofen after training one arm and a placebo following training the other arm on the next day [29]. Each training session took place for 5 days per week for 6 weeks. Although there were time effects present for muscle thickness and strength, there were no significant differences found between the placebo and ibuprofen arms. The dosage seems low since the drug would be out of the body by the time of the next dosage. There could also be systemic reactions that NSAIDs affect, which may explain why no significant results were found using this experimental design. Moreover, this may not depict a realistic model of NSAID consumption, as chronic usage of NSAIDs are more prevalent in older populations.
In summary, although there is a scarcity of research using smaller dosages, it seems that any blunting effects of NSAIDs are not present in such circumstances. Near-maximal dosages may hinder shortterm protein synthesis, however there is still conflicting evidence regarding prolonged hypertrophy and so dosage may not be the most critical factor, assuming it is at or near the maximum recommended amount.
Other factors influencing NSAIDs effects on muscle hypertrophy
Intuitively, subjects with previous training experience may be less prone to extreme muscle damage. In the literature, there seems to be a lack of results stemming from individuals with resistance training experience. However, one example of this can be seen in the study by Krentz's group, in which all subjects had previous resistance training experience [29]. This is based off the thought that they will be able to neurally adapt quicker, and consequently provide more reliable results concerning hypertrophy. This previous training experience may have influenced the inflammation associated with the exercise protocol since data showed that more experienced athletes tend to show fewer signs of muscle damage.
Although there lacks research regarding whole-body resistance training, it is possible that an exercise protocol rich in compound movements can induce higher levels of inflammation. Compound movements inherently induce a higher number of muscles undergoing contractions, potentially leading to an elevated inflammatory response. Most exercise organizations recommend such progressively loaded compound exercises to be a key aspect of resistance training programs. The American College of Sports Medicine (2009) recommends the inclusion of both isolation and compound exercises, with compound movements being prioritized in exercise bouts [30]. Programs rich in compound movements are commonly utilized in real-world applications, such as rehabilitation and strengthening settings. As such, many of the aforementioned findings may not be clinically relevant.
Some studies used a rodent resistance exercise model to elucidate potential underlying mechanisms regarding how NSAIDs affect muscle inflammation and muscle damage following exaggerated muscle contraction. Soltow and colleagues (2006) examined the effect of ibuprofen on muscle enlargement in Sprague-Dawley rats. This study utilized an overload model for the plantaris muscle constructed by surgically removing the soleus and gastrocnemius muscles. Rats in the experimental group were given ~20 mg/kg/ day dose of ibuprofen in drinking water, which is comparable to a high dosage in human. After 14 days of regular cage activity, muscle hypertrophy was blunted by 30% in the ibuprofen treatment group [31]. Bryant et al. (2017) noted a direct negative effect of ketorolac tromethamine on muscle repair following electrically stimulated contractions in mice [32]. This prescription NSAID was selected, as it has a similar mechanism of action as ibuprofen and is prescribed to adults with moderate to severe muscle pain. Nine female mice were used, where tibialis anterior was electrically stimulated (10-12 volts, 100-150 Hz for 400 ms every 30 seconds for 25 minutes per session). Two days after the single contraction session, the mice were injected with 7.5 mg/kg of ketorolac and the muscles were examined 7 hours later. The authors bring up an intriguing point concerning the methods, in that the timing of the dosage mimics a real-life scenario where an individual takes an NSAID when soreness is greatest (e.g. 2 days post resistance training). As has been suggested by others, COX-1 and COX-2 levels increased following this exercise model. The administration of ketorolac significantly decreased COX- 2 levels, with COX-1 levels being unaffected. The researchers also found decreased biomarkers of metabolism and transport, myogensis, and sarcolemma/myocytoskeletal repair after the NSAID was introduced. Along with this, several indicators of anti-apoptosis were downregulated by the ketorolac. This supports the notion that COX inhibitors can influence myotubular response and hypertrophy in a negative manner, assuming that the drug does actually interrupt the enzymatic activity to the same degree in humans.
Conclusion
Currently, there seems to be a fascinating divergence in the direction of the effects seen with NSAID consumption on skeletal muscle adaptations. Age presents as the most influential factor in this equation, with older individuals appearing to benefit from NSAID supplementation during a resistance training regimen. (Table 1) The older population may show benefits from taking NSAIDs due to their increased levels of chronic inflammation and increased rates of skeletal muscle protein degradation. In fact, this area of research is shifting to specifically examine NSAID usage as a pharmaceutical intervention for preventing age-related sarcopenia and atrophy. Dosage may also prove to be an important factor, with maximum daily dosages leading to the most substantial effects. The practicality of these findings must be considered, since consistent high dosages of NSAIDs can lead to other health complications, such as those in the stomach and digestive tract. Unfortunately, most studies examining the topic seem to focus on the more acute, rather than long-term, changes. In addition, the resistance training protocol can also influence responses. Many animal and human studies did not implement realistic progressive overload programs, as the main goal of the exercise interventions was generally to produce substantial muscle damage.
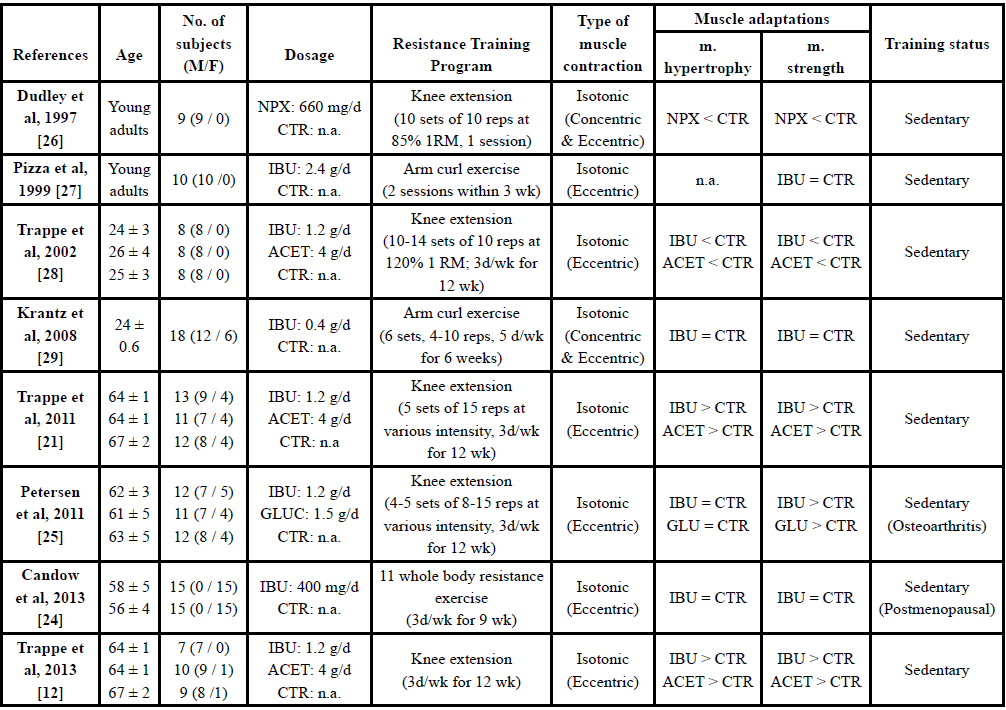
Table: 1Factors affecting NSAIDs effects on skeletal muscle adaptations to resistance training.
IBU: ibuprofen; ACET: acetaminophen; GLUC: glucosamine; NPX: naproxen sodium; CTR: placebo; 1 RM: 1 repetition maximum
A suitable progressive resistance training program should gradually accumulate volume over time, which should attenuate such responses. Neither American College of Sports Medicine nor The National Strength and Conditioning Association, recommend practicing a training modality that stresses a single muscle group [30]. However, heavily loaded isolation exercise was investigated in most of the studies, with little influence of compound exercise. This is an important limitation.
Although these exercise modalities may provide a mechanism to determine physiologic responses, they are not generalizable to a normal bout of resistance exercise, which either consists of multiple upper body, lower body, or whole-body exercises. Moreover, there are different methods to induce skeletal muscle hypertrophy, such as metabolite training (i.e. blood flow restriction), that do not rely on muscle damage. These alternative drivers of muscle adaptations may not be influenced by such drugs. Finally, short-term NSAID use may be a valuable recovery tool for athletes. NSAIDs taken following exercise leads to decreased perception of soreness, which can lead to less of a decrement in athletic performance. More research is needed to examine the long-term effects of NSAID supplementation in various populations, particularly those of older age and trained athletes.
References
Lewis PB, Ruby D, Bush-Joseph CA, Muscle soreness and delayed-onset muscle soreness (2012). Clin Sports Med 31: 255-62.View
Smith DA, Perry PJ (1992) The efficacy of ergogenic agents in athletic competition. Part II: Other performance-enhancing agents. Ann Pharmacother 26: 653-659.View
Singh G (2000) Gastrointestinal complications of prescription and over-the-counter nonsteroidal anti-inflammatory drugs: a view from the ARAMIS database. Arthritis, Rheumatism, and Aging Medical Information System. Am J Ther 7: 115-121.View
Clarkson PM, Hubal MJ (2002) Exercise-induced muscle damage in humans. Am J Phys Med Rehabil 81: S52-69.View
Clarkson PM, Byrnes WC, McCormick KM, Turcotte LP, White JS et al. (1986) Muscle soreness and serum creatine kinase activity following isometric, eccentric, and concentric exercise. Int J Sports Med 7: 152-155.View
Schoenfeld BJ (2012) Does exercise-induced muscle damage play a role in skeletal muscle hypertrophy? J Strength Cond Res 26: 1441-1453.View
Friden J, Sfakianos PN, Hargens AR (1986) Muscle soreness and intramuscular fluid pressure: comparison between eccentric and concentric load. J Appl Physiol 61: 2175-2179.View
Proske U, Morgan DL (2001) Muscle damage from eccentric exercise: mechanism, mechanical signs, adaptation and clinical applications. J Physiol 537: 333-345.View
Armstrong RB (1984) Mechanisms of exercise-induced delayed onset muscular soreness: a brief review. Med Sci Sports Exerc 16: 529-538.View
Markworth JF, Cameron-Smith D (2011) Prostaglandin F2α stimulates PI3K/ERK/mTOR signaling and skeletal myotube hypertrophy. Am J Physiol Cell Physiol 300: 671-682.View
Trappe TA, Liu SZ (2013) Effects of prostaglandins and COXinhibiting drugs on skeletal muscle adaptations to exercise. J Appl Physiol (1985) 115: 909-919.View
Trappe TA, Standley RA, Jemiolo B, Carroll CC, Trappe SW et al. (2013) Prostaglandin and myokine involvement in the cyclooxygenase-inhibiting drug enhancement of skeletal muscle adaptations to resistance exercise in older adults. Am J Physiol Regul Integr Comp Physiol, 304: 198-205.View
Rodemann HP, Goldberg AL (1982) Arachidonic acid, prostaglandin E2 and F2 alpha influence rates of protein turnover in skeletal and cardiac muscle. J Biol Chem 257: 1632-1638.View
Peake JM, Suzuki K, Wilson G, Hordern M, Nosaka K et al. (2005) Exercise-induced muscle damage, plasma cytokines, and markers of neutrophil activation. Med Sci Sports Exerc, 37: 737-745.View
Pizza FX, Hernandez IJ, Tidball JG (1998) Nitric oxide synthase inhibition reduces muscle inflammation and necrosis in modified muscle use. J Leukoc Biol 64: 427-433.View
Feasson L, Stockholm D, Freyssenet D, Richard I, Duguez S et al. (2002) Molecular adaptations of neuromuscular diseaseassociated proteins in response to eccentric exercise in human skeletal muscle. J Physiol 543: 297-306.View
Calle MC, Fernandez ML (2010) Effects of resistance training on the inflammatory response. Nutr Res Pract 4: 259-269.View
Vane JR, Botting RM (1998) Mechanism of action of nonsteroidal anti-inflammatory drugs. Am J Med 10: 2S-8S; discussion 21S-22S.View
Trappe TA, Fluckey JD, White F, Lambert CP, Evans WJ. et al. (2001) Skeletal muscle PGF(2)(alpha) and PGE(2) in response to eccentric resistance exercise: influence of ibuprofen acetaminophen. J Clin Endocrinol Metab 8: 5067-5070.View
Graham GG, Scott KF (2005) Mechanism of action of paracetamol. Am J Ther 12: 46-55.View
Trappe TA, Carroll CC, Dickinson JM, LeMoine JK, Haus JM et al. (2011) Influence of acetaminophen and ibuprofen on skeletal muscle adaptations to resistance exercise in older adults. Am J Physiol Regul Integr Comp Physiol 300: 655-662.View
Trappe TA, Ratchford SM, Brower BE, Liu SZ, Lavin KM et al. (2016) COX Inhibitor Influence on Skeletal Muscle Fiber Size and Metabolic Adaptations to Resistance Exercise in Older Adults. J Gerontol A Biol Sci Med Sci 71: 1289-1324.View
Baldwin AC, Stevenson SW, Dudley DA (2001) Nonsteroidal anti-inflammatory therapy after eccentric exercise in healthy older individuals. J Gerontol A Biol Sci Med Sci 56: 510-513.View
Candow DG, Chilibeck PD, Weisgarber K, Vogt E, Baxter-Jones AD et al. (2013) Ingestion of low-dose ibuprofen following resistance exercise in postmenopausal women. J Cachexia Sarcopenia Muscle 4: 41-46.View
Petersen SG, Beyer N, Hansen M, Holm L, Aagaard P et al. (2011) Nonsteroidal anti-inflammatory drug or glucosamine reduced pain and improved muscle strength with resistance training in a randomized controlled trial of knee osteoarthritis patients. Arch Phys Med Rehabil 92: 1185-1193.View
Dudley GA, Czerkawski J, Meinrod A, Gillis G, Baldwin A et al. (1997) Efficacy of naproxen sodium for exercise-induced dysfunction muscle injury and soreness. Clin J Sport Med 7: 3-10.View
Pizza FX, Cavender D, Stockard A, Baylies H, Beighle A et al. (1999) Anti-inflammatory doses of ibuprofen: effect on neutrophils and exercise-induced muscle injury. Int J Sports Med 20: 98-102.View
Trappe TA, White F, Lambert CP, Cesar D, Hellerstein M et al. (2002) Effect of ibuprofen and acetaminophen on postexercise muscle protein synthesis. Am J Physiol Endocrinol Metab, 282: 551-556.View
Krentz JR, Quest B, Farthing JP, Quest DW, Chilibeck PD et al. (2008) The effects of ibuprofen on muscle hypertrophy, strength, and soreness during resistance training. Appl Physiol Nutr Metab 33: 470-475.View
American College of Sports, M (2009) American College of Sports Medicine position stand. Progression models in resistance training for healthy adults. Med Sci Sports Exerc 41: 687-708.View
Soltow QA, Betters JL, Sellman JE, Lira VA, Long JH et al. (2006) Ibuprofen inhibits skeletal muscle hypertrophy in rats. Med Sci Sports Exerc 38: 840-846.View
Bryant AE et al. (2017) Effects of delayed NSAID administration after experimental eccentric contraction injury - A cellular and proteomics study. PLoS One 12: 0172486.View

