Journal of Public Health Issues and Practices Volume 6 (2022), Article ID: JPHIP-196
https://doi.org/10.33790/jphip1100196Research Article
Patient Preferences for Early Diagnosis of Endometriosis and Associated Determinants in the United States: A Discrete Choice Experiment
Carmen Lyttle-Nguessan, Ph.D., Vakaramoko Diaby, Ph.D.
Research Associate, Florida A&M University, Tallahassee, Florida 32307, United States.
University of Florida, 1225 Center Dr., Gainesville, Florida 32610, United States.
*Corresponding Author Details: Carmen Lyttle-Nguessan, Ph.D., Research Associate, Florida A&M University, Tallahassee, Florida 32307, United States. E-mail: carmen.lyttlenguessa@famu.edu
**Co-Author: Vakaramoko Diaby, Ph.D., University of Florida, 1225 Center Dr., Gainesville, Florida 32610, United States. E-mail: v.diaby@cop.ufl.edu
Received date: 10th January, 2022
Accepted date: 26th February, 2022
Published date: 28th February, 2022
Citation: Nguessan, C. L., Diaby, V., (2022). Patient Preferences for Early Diagnosis of Endometriosis and Associated Determinants in the United States: A Discrete Choice Experiment. J Pub Health Issue Pract 6(1): 196.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Background: Endometriosis is a chronic and incurable gynecological disease that mainly affects women of reproductive age worldwide. It imposes clinical and economic burdens on patients, families, and society. A better understanding of the determinants of preferences towards early diagnosis of endometriosis may help develop programs and interventions to reduce the risk of more severe illness. We quantified patient preferences for early endometriosis diagnosis and explored whether preferences vary on the patient characteristics and pre-established social determinants of health.
Methods: A discrete choice experiment (DCE) was designed to elicit women's preferences and willingness to pay for early diagnosis of endometriosis. Women ages 18 and older were eligible to participate in the study. The attributes (and levels) considered to describe hypothetical scenarios included diagnosis (immediate/postponed), the chance of advanced endometriosis and more severe illness (low/ high), time away from living, and professional activities (8 days, 15 days, 22 days and 30 days), and possible out-of pocket costs ($0, $15, $60 and $210). The effects of participants' characteristics and social determinants of health on the preference for early diagnosis were modeled using a Tobit model.
Results: A total of 66 women with (2) or at-risk (64) of endometriosis completed the experiment. The respondents' age and insurance statuses significantly influenced their preference or choice for early diagnosis. On average, respondents were willing to give up $61.55 out-of-pocket cost to have a low risk of advanced endometriosis and more severe disease. The Tobit model indicates only age and insurance variables significantly affected early diagnosis preference. The results suggest that older ages and not having insurance increase the likelihood of respondents choosing early diagnosis than the younger age group and having insurance.
Conclusions: This study indicates the importance of considering the patient characteristics and social determinants of health when designing and implementing health programs and interventions for endometriosis.
Key Words: Willingness to Pay, Tobit Model, Attributes, Decision-Making, Optimal Design, Health Outcomes, Individuals' Characteristics, Age and Insurance Status
Background
Endometriosis is a disease that can affect all women of reproductive age, regardless of race, ethnicity, or socioeconomic status [1,2]. The literature defines endometriosis as the presence of the tissues of the endometrial lining outside the uterus inflaming areas of the body such as the ovaries, pelvis, abdominal cavity, and even the thorax and skin [2-5]. The disease imposes both clinical and economic burdens and concerns individuals and society. In addition to the clinical effects, endometriosis can profoundly impact women's quality of life. In fact, in a study that assessed the quality-adjusted life years, women described their experience of endometriosis to be worse than death [6,7].
There is no cure for endometriosis, and the treatment depends on several factors, including the disease's severity. Delays in treatment may exacerbate the burden of the disease and reduce the quality of treatment outcome [6,8,9]. Delays in diagnosis, high hospital admission rates, surgical procedures, and incidences of comorbid conditions make endometriosis a more costly public health problem than other chronic conditions such as migraine and Crohn's disease [10].
Studies have shown that age and insurance access often influence decisions to seek early diagnosis and treatment for endometriosis [11,12]. Younger adults and individuals without insurance are less likely to get a routine medical checkup and seek medical attention before a critical health issue [13]. A deeper understanding of the determinants of early diagnosis and treatments for endometriosis may help develop targeted programs and interventions to reduce the risk of more severe illness or reduce the impact of the disease outcomes [14,15]. In the absence of such critical evidence, women of reproductive age may continue to suffer clinical, humanistic, and economic burdens associated with endometriosis.
In this study, we quantified patient preferences for early diagnosis of endometriosis and explored whether preferences vary on the patient characteristics and pre-established social determinants of health.
No other DCE, to our knowledge, has addressed the role of age and insurance status in influencing preferences on early diagnosis of endometriosis. This study highlights the importance of accounting for individual preferences in improving decision-making for diagnosing and treating endometriosis.
Materials and Methods
DCEs have become a common technique in health economics research providing information on relevant characteristics (attributes) of services and programs [16]. Typically, a DCE elicits preferences that estimate individuals' value on a particular good or service [17,18]. In this, the researcher asked respondents to choose between two or more alternatives among several scenarios in which they described several attributes regarding the good or service at different levels. One step in DCE is to gather demographics and socioeconomic information to help explain the individuals' preferences or choices. We collected several demographic information, including age and insurance.
Target population and subgroups
Women 18 years and older at risk for or diagnosed with endometriosis were eligible to participate in the study. Non-English-speaking individuals cognitively impaired needing a caregiver to complete a survey were excluded. We targeted women with potential risk for endometriosis and rich information on endometriosis. The literature defines women at risk for endometriosis as women currently menstruating, using replacement estrogen, and with opportunity for diagnosis [19]. This definition suggests a broad age group and characteristics and provided guidance in determining eligible participants for our research. This study identified suitable women aged 18-64 years. Still, it did not exclude older women if they have a history of endometriosis since they may add richness to the research. While endometriosis predominantly affects younger women, the most severe cases are found in older and postmenopausal women. Moreover, older women have a greater risk for endometriosis-associated ovarian cancer [20,21].
We excluded non-English speaking individuals from taking the survey since it was written only in English and otherwise invalid. In addition, a non-English language survey instrument would be costly, time-consuming, and difficult to validate. Because of the potential cognitive demands of the DCE instrument, we excluded individuals with cognitive impairment and who needed caregiver assistance in completing the survey from the survey.
Setting and location
We conducted the study in Tallahassee, Florida. We identified the eligible participants from various group settings, mainly among students, the general community setting, and a local community health center.
Sample size
Sample size calculations are complicated for DCE, and the literature has several recommendations for what it should be [22-25]. For this study, we based the sample size primarily on convenience.
Data collection
A pre-tested DCE survey instrument was hand-delivered to individuals at health clinics and other group settings to collect individuals' preferences effectively. The questionnaire gathered choice and demographic data such as age, race, education, insurance status, employment status, knowledge, endometriosis status (diagnosed or not), and annual household income. The second section of the questionnaire comprised the choice task with a series of 16 paired scenarios, each requiring respondents to select one of two options. The questionnaire included only the English language.
Discrete choice experiment
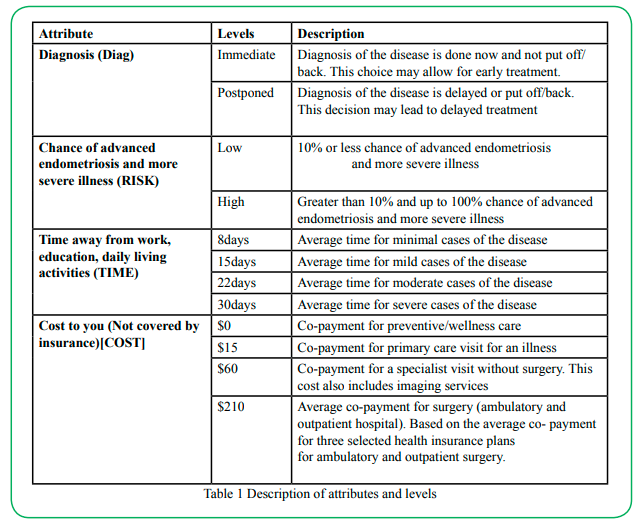
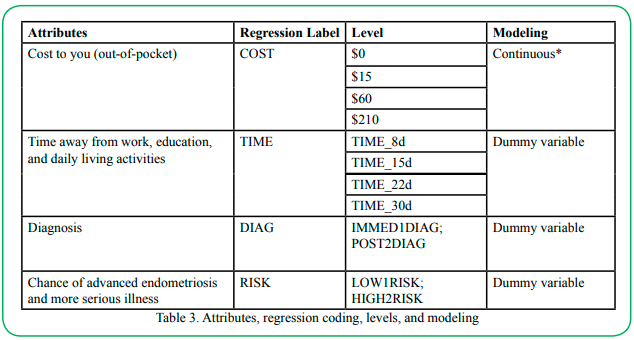
A discrete choice experiment instrument was created and administered to all participants (Table 1). The final attribute selection included diagnosis (immediate, postponed); chance of advanced endometriosis and more severe illness (low, high); time away from work, education, daily living activities (8 days, 15days, 22days, 30days), and cost to you not covered by insurance ($0, $15, $60, $210). We used literature search and expert opinion to define the attributes and levels and pre-test the survey with selected women willing to participate (n=10).
Experimental design
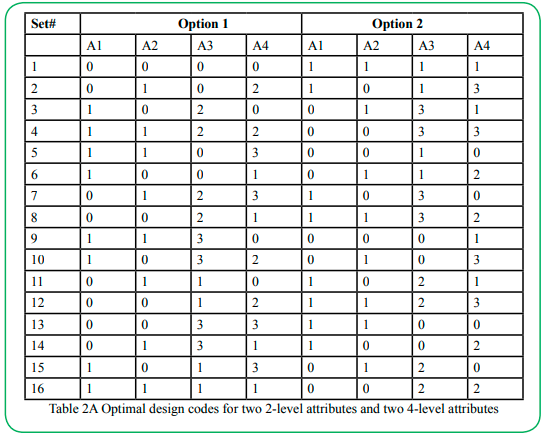
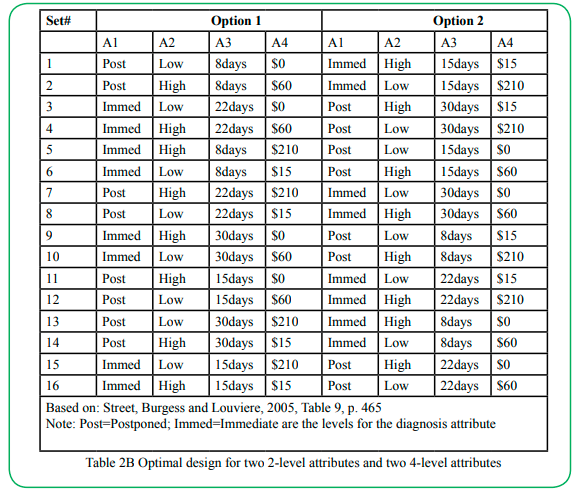
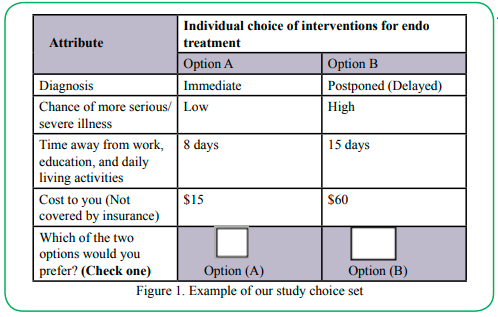
We designed the DCE using the optimal design approach from the Street, Burgess, and Louviere [26] table format. This approach systematically coded the levels starting with 0 and counting according to attribute levels. For instance, "0, 1, 2, and 3" was coded for a 4-level attribute, and 0 and 1 for a 2-level attribute. The design permits one to choose a format that matches the number of levels and attributes in one's research plan. We used the Table 9 version optimal design for two 2-level and 4-level attributes (Table 2A&B). Table 2B comprises the level names of the different attributes of the study. The optimal design approach we used automatically checked for orthogonality, level balance, and minimal levels overlapping [26]. We asked respondents to choose their preferred treatment effect scenario from the options labeled A and B (Figure 1).
Data collection
Women visiting at a local community health clinic, in the general public, and at students, gatherings were hand delivered the pre-tested survey instrument to complete independently. The cover letter of the questionnaire also served as informed consent. The letter informed participants that their participation was completely voluntary and of their right to withdraw at any time without penalty. Completed questionnaires were delivered directly to the research team/researcher for data inputting.
Data Analysis
The DCE experimental design and data analysis are directly linked. The response choice (Option A or Option B) is the dependent variable in the statistical model in which we estimated utility from observed choices. The data were organized and summarized with descriptive statistics (e.g., frequency, percentage and, standard deviation).
A mixed logit model with random effects was used to assess the impact of attribute levels on participants' preferences for early diagnosis of endometriosis. The variable cost was assumed log-normally distributed while the remaining variables were normally distributed. The model estimates the value or utility each respondent attaches to the different levels of the attributes and how the levels of the attributes impact individuals' choices. Using Hiligsmann et al. [27] as a guide, we specified the model in Equation 1.
Vij = β0 + (β1 + n1i) COSTj + (β2 + n2і) TIME_15dj + (β3 + n3i) TIME_22dj + (β41 + n4i ) TIME_30dj + (β5 + n5і) IMMED1DIAGj + (β6 + n6і)HIGH1RISKj + εij (1)
Where Vij indicates the utility that an individual i assigns to an intervention j. Vij is modeled as the sum of two parts: a systematic part based on the attributes in the DCE and an error or stochastic part (random component), Eij. The random component is a function of the unobserved attributes and variation in an individual's preference [27,25]. β0 is the constant reflecting the preferences for selected option or intervention relative to no option/intervention, (β1to β6) the mean attribute utility weights in the population, and n1ito n6i error terms for individual-specific unexplained variation in the utility weights.
We coded the categorical variables (TIME, DIAG, and RISK) to reflect the levels of the primary attributes. For example, the variable labeled HIGH1RISKj refers to the attribute with the level of risk considered high. Out-of-pocket cost is the COST attribute treated as with the level of risk considered high. Out-of-pocket cost is the COST attribute treated as calculating the WTP values [25]. We used Dummy codes to describe the categorical variables (Table 3). At any given moment, one level will take a value of 1, and 0 for all others [25]. The coefficient signs reflect whether the attribute has a positive or negative effect on the intervention utility.
We use the WTP to quantify an individual's tradeoff and utility by calculating the marginal substitution rate. WTP estimates for the categorical attributes were calculated as the ratio of the coefficients of the attributes (numerator) and cost attribute (denominator). A WTP value represents how much one is willing to pay (or give up) for a unit change in the attribute and is calculated by taking the ratio of the mean parameter for the attribute level to the mean parameter related to the cost (or other continuous quantitative variables). For example, what individuals are willing to pay, on average, to reduce the risk (chance) of advanced endometriosis and more serious (or for early [immediate] diagnosis). Likewise, they were willing to pay to spend less time away from work, education, and daily living activities. We use the mixed logit model (MXL) for the WTP estimates.
We used a Tobit model to investigate the effects of respondents' characteristics on their choice for immediate diagnosis. The Tobit model estimates a linear relationship between variables when either left – or right- censoring the dependent variable [28]. The dependent variable was the proportion of selected profiles that contained the attribute "immediate diagnosis" for each respondent. Because the Tobit model requires 15 observations per variable (participant-level characteristics) included in the model, a forward stepwise regression was conducted to identify statistically significant variables in predicting our dependent variable at a 5% significance level. We coded the respondents' background information (Section 3 of the survey), assigning a single identifier name for each question. For instance, questions 17 to 26 were named 'knowledge', 'information', 'diagnosed', 'stage', 'insurance', 'race', 'age', 'education', 'employment', and 'income' respectively. We used these as the independent variables in the analysis.
We tested the goodness of fit of the Tobit models using a log-likelihood ratio (LR) and Wald Chi-square tests. The data were analyzed using SAS version 9.4 and Stata version 12.0.
Assumptions
This study has several assumptions relating to the participants and the variables used. We assumed that the participants have the cognitive ability to make a rational choice independent of a caregiver and have some knowledge about endometriosis. This ability helped them make the tradeoffs in the decision-making process. We also assumed that the payment vehicle (cost attribute) represented the typical out-of-pocket healthcare cost for an individual seeking healthcare. We also took that the out-of-pocket cost means the actual co-payment insurance for preventive/wellness care, diagnostic and surgical procedures, and specialist visits. These assumptions imply that an individual has sought medical assistance (medical visit) at one time or the other.
We based the cost attribute on co-payments from selected HMO (Health Maintenance Organization) insurance providers such as Blue Cross/Blue Shield (Blue), Capital Health Plan (CHP), and Humana. We based cost attribute calculations on potential healthcare visits or care such as surgery (ambulatory and outpatient hospital), specialist, imaging, preventive care/ wellness, and primary care. These determined the four payment (cost attribute) levels. We averaged ambulatory and inpatient hospital copayment amounts for the three insurance plans to combine as one payment level ($210). Likewise, the specialist and imaging costs were combined to form another group ($60). Overall, the cost attribute included only network or referred provider cost based on a single visit for the specific care and the average of health plans co-payments per visit. Preventive care, also known as wellness care, costs is $0 for all health insurance plans. Co-payment for primary care visit for an illness is $15.
Results
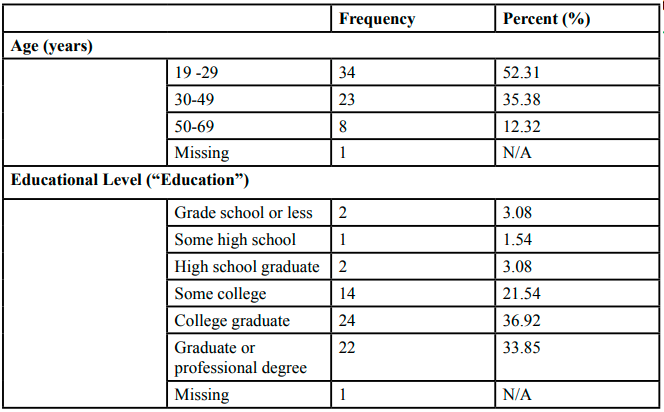
We distributed 72 questionnaires to individuals, received 67 in return, representing a response rate of 93%. We excluded one questionnaire because the individual, per Hiligsmann and others' [27] recommendation, did not complete at least five of the choice sets in the DCE task. We included the remaining 66 (92%) questionnaires for data analysis. Respondents' socio-demographics and health characteristics are in Table 3. There was no restriction on participation based on individuals' race and ethnicity, but individuals were mainly black (about 72%). The other 28% comprises whites, Asians, Hispanics, and mixed races. Individuals were primarily in 19-29 and 30-49 age groups, and none of the respondents fell in the extreme upper (70+) or lower (18 or less) age groups. Of those responding, two (3.13%) were diagnosed with endometriosis but did not know the stage of their disease. The percent of those insured to some extent was 86.
We did a pre-test with 10 participants as face validity to test the entire survey instrument's clarity, ease (or difficulty), and comprehension. We gave the participants follow-up questions to determine their understanding of the DCE choice task and the length and ease of the instrument. Almost all participants indicated the choice task was straightforward, generally not tricky, and understandable. We also asked an endometriosis expert to evaluate the instrument's noteworthiness based on the contents. We revised and updated the survey instrument based on any comments or discourses.
Willingness to pay analysis
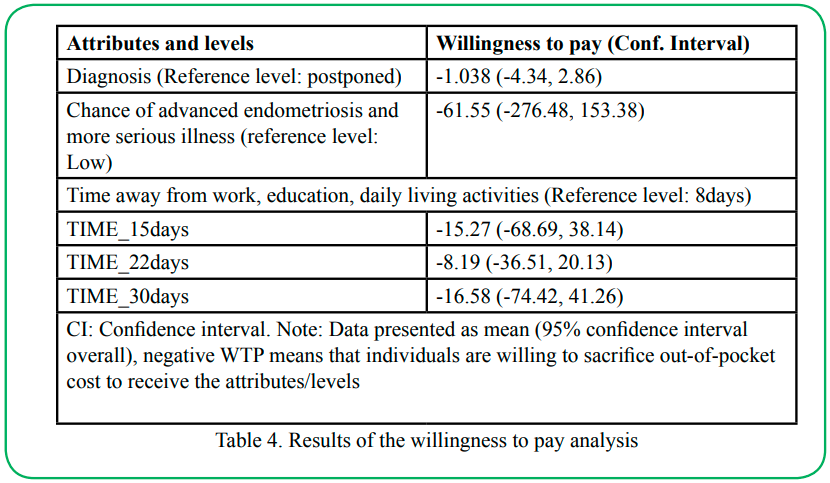
The WTP values for attributes levels are in Table 4. WTP results were not statistically significant, but the values are noteworthy economically. Though the results are not statistically significant, the values indicate that respondents would be willing to give up money 1) to put off their diagnosis for a later time, 2) to have an intervention that would lower their risk of endometriosis, and 3) to spend less time away from work, education and other daily living activities. For instance, respondents were willing (on average) to give up $61.55 out-of-pocket cost to have a low risk of advanced endometriosis and more severe disease.
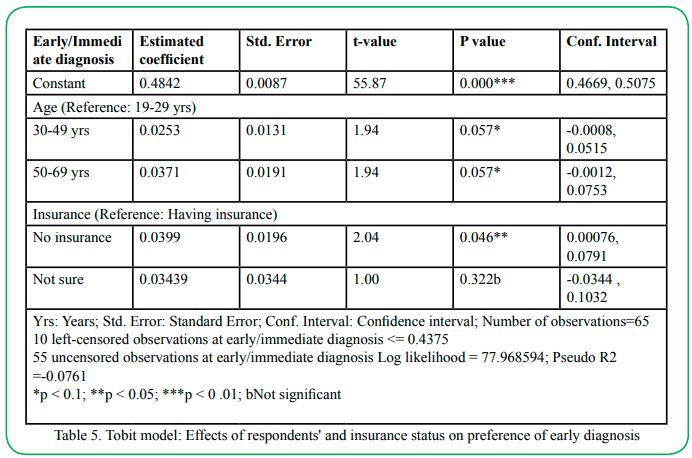
Tobit model: Stepwise model selection results
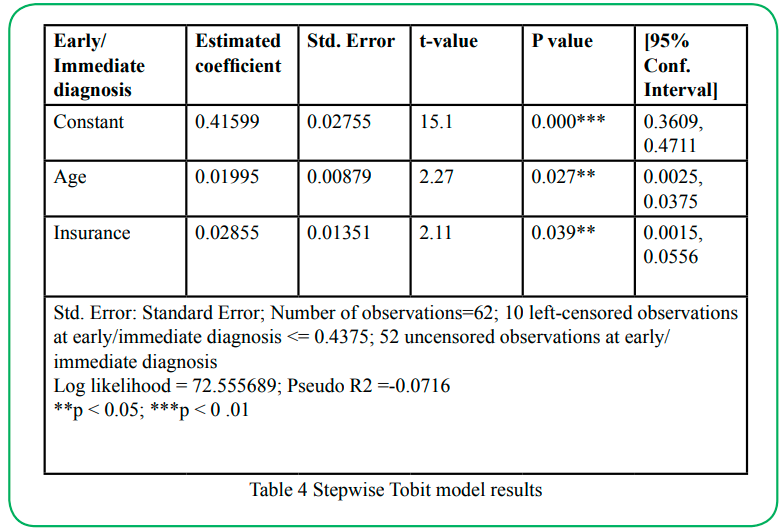
The results of the stepwise Tobit model are in Table 5. The final model was statistically significant (p < 0.01) compared to an empty model. Of the variables introduced into the model, only age and insurance significantly affected early diagnosis preference.
A. Tobit model: Effects of respondents' characteristics on preferences
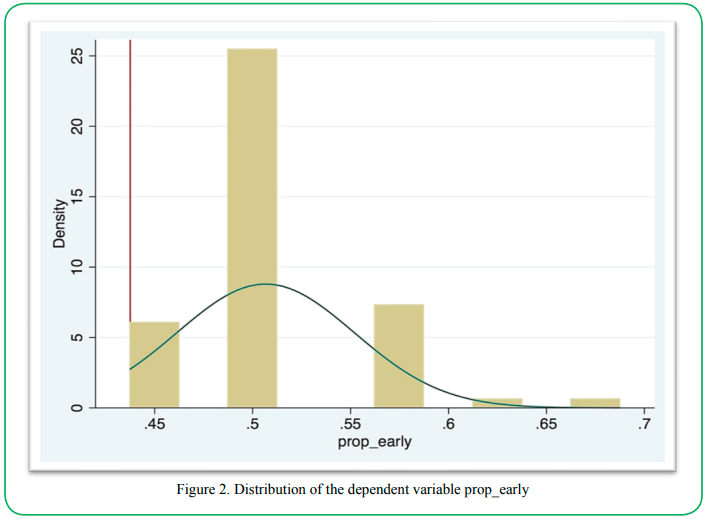
Figure 2 shows the distribution of our dependent variable "proportion of selected profiles that contained the attribute immediate diagnosis" (prop_early). The data is left-censored, with our dependent variable being observable only between the values 0.4375 (lower bound) and 0.6875 (upper bound). The visual inspection of the data supports using a Tobit model to analyze the data. The results of the Tobit model are in Table 5. The overall model was statistically significant (p < 0.05).
As mentioned before, we modeled only age and insurance to explain the effect of preference on early diagnosis. The estimated coefficients of the variables had positive signs, which suggest positive effects (increase) on the dependent variable prop_early. The results indicate that older age and not having insurance increased the likelihood of respondents choosing immediate or early diagnosis compared to the younger age group and having insurance, respectively. For example, if the dependent variable prop_early were not censored, the estimated coefficient for age category 30-49 years (0.0253) would mean that prop_early is 0.025 points higher for respondents in 30- 49 to respondents in the age group 19-29. Likewise, the estimated coefficient for no insurance (0.0399) would mean that prop_early is 0.04 points higher for respondents in the category of no insurance than respondents with insurance. Since the data are censored, then it is latent censored variable y that is linearly related with the independent variables age and insurance. As a result, a marginal effect analysis of the effect of the variable age and insurance is needed to draw more accurate conclusions.
B. Tobit model: Marginal effects of respondents' characteristics on preferences
The results for the Tobit marginal effect analysis are in Table 6. The marginal effects calculated the exact change on the truncated expected value of our dependent variable prop_early. For example, being aged 19-29 would cause the truncated expected value of prop_ early to increase by 0.854 points.
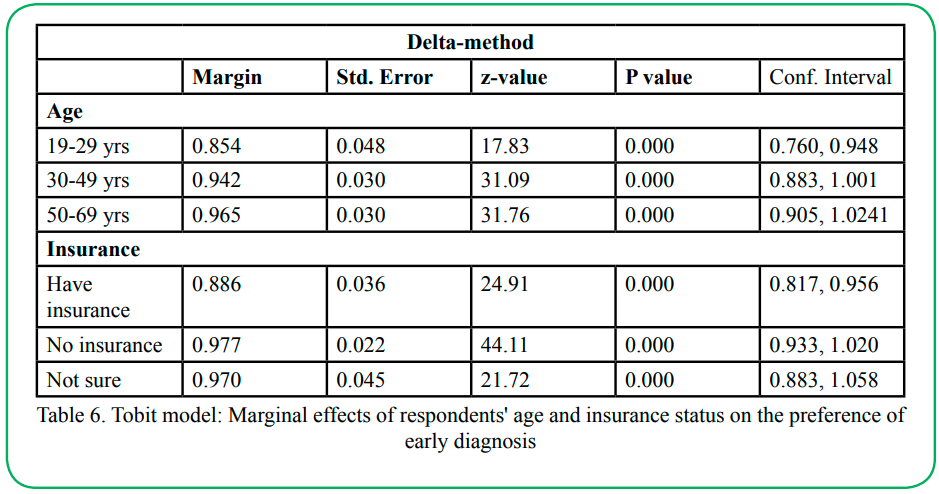
Figure 7. Tobit model: Marginal effects of respondents' age and insurance status on the preference of early diagnosis
Discussion
Study findings
This study, to our knowledge, is the first to use DCE to examine the role of age and insurance status in influencing preferences on early diagnosis of endometriosis. Our mixed logit model results suggest that respondents prefer to put off diagnosis later. Given that our participants are generally younger individuals (ages 19-29) may shed some light regarding why women do not get diagnosed early. Furthermore, our results indicate that individuals without insurance are more likely to prefer immediate/early diagnosis of endometriosis, which is not generally what we would expect. However, studies suggest that being insured does not guarantee an early diagnosis of a condition, and non-Hispanic blacks (compared to non-Hispanic whites) are less likely (even insured) to get an early diagnosis [29]. Our respondents were mainly non- Hispanic blacks, which might explain the differences.
A Tobit model was estimated to explain the impact of the respondents' characteristics on the choice for early diagnosis. This evaluation attempted to answer what factors prompt women toward earlier diagnosis and why they do not get diagnosed early. The significant explanatory variables were age and insurance. The results suggest that older individuals and those without insurance prefer early diagnosis. Studies showed that younger individuals delay seeking medical attention than their older counterparts.
Limitations
While the Tobit model seemed to be the best fit for our data, given a censored distribution of our dependent variable, there were limitations to its use. For example, the Tobit model generally requires at least 15 observations per variable included, while our data only contained 66 observations with several patient-level variables. Therefore, we had to remove some variables before modeling the data, impacting the results' accuracy. The mean WTP results were not statistically significant, but the values are noteworthy economically. The negative signs of the values reveal that individuals are willing to give up the out-of-pocket cost to avoid difficult or uncomfortable situations. At the same time, ordinarily, they would prefer low out-of-pocket costs. This concept is consistent with common sense logic and other DCE studies [30].
Generalizability and Current knowledge
This study could benefit health professionals and decision-makers, especially given the long delays diagnosed with endometriosis. This study provides opportunities to explore programs and products to improve treatment outcomes of endometriosis by considering individuals' preferences. For instance, the results indicate that younger individuals are more likely to postpone diagnosis, suggesting the need for programs targeting youths and younger adults, perhaps highlighting the long-term benefit of early diagnosis. In addition, the wide variation in the individuals' preferences highlights the importance of incorporating individuals' preferences and informed and shared decision-making processes in improving endometriosis treatment and outcomes.
Conclusion
No other DCE addressed the role of age and insurance status in influencing preferences on early diagnosis of endometriosis. This study highlights the importance of accounting for individual preferences and demography in improving decision-making for diagnosing and treating endometriosis. The respondents' age and insurance status significantly influence their choice for early diagnosis. The respondents' preferences' results provide opportunities to examine current practices. Since this was exploratory research, we recommend further investigation about the influence of age and insurance status on the decision for early diagnosis of endometriosis. Future studies may investigate more diverse demographics and spatial impacts.
Additional/Supplemental file
Additional file:
data on the selection of attributes used in DCE?
Funding
This research received no funding
Conflicts of interest:
The authors declare that there is no conflict of interest
List of abbreviations
ACOG American College of Obstetricians and Gynecologists
ASRM American Society of Reproductive Medicine
CHP Capital Health Plan
Diag Diagnosis
DCE Discrete choice experiment
HMO Health Maintenance Organization
Immed Immediate
Post Postponed
Prop_early Proportion of preferences on immediate diagnosis
SAS Statistical Analysis Systems
Stata Data analysis and statistics
WERF World Endometriosis Research Foundation
WHO World Health Organization
WTP Willingness to pay
Acknowledgments:
I want to express special thanks to co-chair Dr. Ellen Campbell and committee members Drs. C. Perry Brown, Michael Thomas, and Hong Xiao for supporting research during my dissertation. This article is a subpart of my dissertation.
References
Simoens, S., Hummelshoj, L., & D’Hooghe, T. (2007). Endometriosis: Cost estimates and methodological perspective. Human Reproduction Update, 13(4), 395–404.View
Agarwal, A., & Subramanian, A. (2010). Endometriosis - morphology, clinical presentations and molecular pathology. Journal of Laboratory Physicians, 2(1), 1-9. DOI: 10.4103/0974- 2727.66699.View
Abbott, J., Hawe, J., Hunter, D., Holmes, M., Paul Finn, P., & Garry, R. (2004). Laparoscopic excision of endometriosis: A randomized, placebo-controlled trial. Fertility and Sterility, 82(4), October 2004. doi:10.1016/j.fertnstert.2004.03.046. View
Hediger, M. L., Hartnett, H. J., & Buck Louis, G. M. (2005). Association of endometriosis with body size and figure. Fertility and Sterility, 84(5), November 2005. doi:10.1016/j. fertnstert.2005.05.029. View
Nezhat, C., Nezhat, F., & Nezhat, Ceana. (2012). Endometriosis: ancient disease, ancient treatments. Fertility and Sterility, 98(65). http://dx.doi.org/10.1016/j.fertnstert.2012.08.001.View
Gao, X., Outley, J., Botteman, M., Spalding, J., Simon, J. A., & Pashos, C. L. (2006). Economic burden of endometriosis. Fertility and Sterility, 86, 1561-1572.
Simoens, S., Dunselman, G., Dirksen, C., Hummelshoj, L., Bokor, A., Brandes, I., … Brodszky, V. (2012). The burden of endometriosis: Costs and quality of life of women with endometriosis and treated in referral centres. Human Reproduction, 27, 1292-2012.View
American Society for Reproductive Medicine [ASRM]. (2012). Endometriosis and infertility: A committee opinion. Fertility and Sterility, 98(3). http://dx.doi.org/10.1016/j. fertnstert.2012.05.031 Practice Committee, American Society for Reproductive Medicine, 1209 Montgomery Hwy., Birmingham, AL 35216. View
What are the treatments for endometriosis? (n.d). In National Institute of Child Health and Human Development (NICHD) /National Institute of Health. Retrieved at: https://www.nichd. nih.gov/health/topics/endometri/conditioninfo/treatment#top.View
Schindler, A. E. (2011). Dienogest in long-term treatment of endometriosis. International Journal of Women's Health, 3, 175-184. http://dx.doi.org/10.2147/IJWH.S5633.View
Kirzinger, W. K., Cohen, R. A., &. Gindi, R.M. (2012). Health care access and utilization among young adults aged 19–25: Early release of estimates from the National Health Interview Survey, January–September 2011. Division of Health Interview Statistics, National Center for Health Statistics, May 2012. Available from: http://www.cdc.gov/nchs/nhis/releases.htm.View
Ponce, N., Glenn, B., Shimkhada, R., Scheitler, A.J.,& Ko, M. (2017). Barriers to Breast Cancer Care in California: A report to the California Breast Cancer Research Program. UCLA Center For Health Policy Research, 10960 Wilshire Blvd. Suite 1550, Los Angeles, CA 90024.View
Taber, J. M., Leyva, B., & Persoskie, A. (2015). Why do People Avoid Medical Care? A Qualitative Study Using National Data. Journal of General Internal Medicine, 30 (3), 290-297. doi: 10.1007/s11606-014-3089-1View
van Dijk, L. J., Nelen, W. L., D’Hooghe, T. M., Dunselman, G. A., Hermens, R. P., Bergh, C…., Kremer, J. A. (2011). The European Society of Human Reproduction and Embryology guideline for the diagnosis and treatment of endometriosis: an electronic guideline implementability appraisal. BioMed Central Implementation Science, 6(7). http://www. implementationscience.com/content/6/1/7 View
Shah,D.K., Moravek, M.B., Vahratian, A., Dalton, V.K., & Lebovic, D.L. (2010). Public Perceptions of Endometriosis: Perspectives from both genders. Acta Obstetricia et Gynecologica, 89, 646-650.
de Bekker-Grob, E. W., Ryan, M., & Gerard, K. (2010). Discrete choice experiments in health economics: A review of the literature. Health Economics. Published online in Wiley Online Library (wileyonlinelibrary.com). DOI: 10.1002/hec.1697View
Viney, R., Lancsar, E., & Louviere, J. Discrete choice experiments to measure consumer preferences for health and healthcare. Expert Review of Pharmacoeconomics & Outcomes Research, 2(4), August 2002. DOI: 10.1586/14737167.2.4.319. View
Rubin, G., Bate, A., George, A., Shackley, P., & Hall, N.( 2006). Preferences for access to the GP: A discrete choice experiment. The British Journal of General Practice 56(531): 743–748.View
American College of Obstetricians and Gynecologists [ACOG]. (2010). Management of Endometriosis. Washington DC: American College of Obstetricians and Gynecologists (ACOG); 2010 July 14 p. (ACOG practice bulletin; no. 114). [129 references]. View
Wei, J., William, J., & Bulun, S. (2011). Endometriosis and ovarian cancer: A review of clinical, pathologic, and molecular aspects. International Journal of Gynecological Pathology, 30(6), 553-568. doi:10.1097/PGP.0b013e31821f4b85.View
Pavone, M. E., & Lyttle, B.M. (2015). Endometriosis and ovarian cancer: links, risks, and challenges faced. International Journal of Women's Health, 5(7), 663-672.View
Bridges, J. F .P., Hauber, A. B., Marshall, D., Lloyd, A., Prosser, L. A., Regier, D. A.,…, Mauskopf, J. (2011). Conjoint analysis applications in health—a Checklist: A report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value In Health, 14(2011), 403-413. doi:10.1016/j.jval.2010.11.013. View
de Bekker-Grob , E. W. , Donkers, B., Jonker, M.F., & Stolk, E.A. (2015). Sample size requirements for discrete choice experiments in healthcare: A practical guide. Patient, 8, 373- 384. DOI 10.1007/s40271-015-0118-z. View
Louviere, J. J., Islam T., Wasi, N., Street, D. & Burgess, L. (2008). Designing discrete choice experiments: do optimal designs come at a price? Journal of Consumer Research, 35 360-375.
World Health Organization. (2012). How to Conduct a Discrete Choice Experiment for Health Workforce Recruitment and Retention in Remote and Rural Areas: A User Guide with Case Studies: WHO Library Cataloguing-in-Publication Data. ISBN 978 92 4 150480 5 (NLM classification: WA 390, www.who.int. View
Street, D.J., Burgess, L., & Louviere, J.J. (2005). Quick and easy choice sets: Constructing optimal and nearly optimal stated choice experiments. International Journal of Research in Marketing 22(4): 459–470. View
Hiligsmann, M., Dellaert, B.G., Dirksen, C.D., Weijden, T., Goemaere, S., Reginster, J., …Watson, V. (2014). Patients' preferences for osteoporosis drug treatment: A discrete choice experiment. Arthritis Research and Therapy, 16, R36; http:// arthritisresearch.com/content/16/1/R36.View
Tobit analysis. (n.d.) In Institute for Digital Research and Education, UCLA. Retrieve April 11, 2017 from http://stats. idre.ucla.edu/stata/dae/tobit-analysis/. View
Hoffman, H. J., LaVerda, N. L., Levine, P. H., Young, H. A., Alexander, L. M., Patierno, S. R., & DC-PNRP Research Group. (2011). Having health insurance does not eliminate race/ ethnicity-Associated delays in breast cancer diagnosis in the District of Columbia. Cancer, 117(16), 3824-3832. doi:10.1002/ cncr.25970. View
Wanders, J. O.P., Veldwijk, J., de Wits, G. A., Hart, H. E., van Gils, P. F., & Lambooij, M. (2014). The effect of out-of-pocket costs and financial rewards in a discrete choice experiment: an application to lifestyle programs. BMC Public Health, 14:870. http://www.biomedcentral.com/1471-2458/14/870 View