Journal of Public Health Issues and Practices Volume 6 (2022), Article ID: JPHIP-199
https://doi.org/10.33790/jphip1100199Research Article
Association Between Duration of Diabetes Mellitus and Chronic Kidney Disease Staging Among Type II Diabetes Mellitus Patients in Padang Terap, Kedah, Malaysia
Abdul Hadi Mohd Zuki 1, & Mohamad Rodi Isa 1*
1Department of Public Health Medicine, Faculty of Medicine, Universiti Teknologi MARA(UiTM) Selangor, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia.
Corresponding Author Details: Mohamad Rodi Isa, MBBS, DAP&E, MPH, DrPH, Department of Public Health Medicine, Faculty of Medicine, Universiti Teknologi MARA, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia. E-mail: rodi@uitm.edu.my
Received date: 22nd March, 2022
Accepted date: 04th April, 2022
Published date: 06th April, 2022
Citation: Mohd Zuki, A.H., & Isa, M.R. (2022). Association Between Duration of Diabetes Mellitus and Chronic Kidney Disease Staging Among Type II Diabetes Mellitus Patients in Padang Terap, Kedah, Malaysia. J Pub Health Issue Pract 6(1): 199.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Background: Chronic kidney disease (CKD) is a global health burden with a high economic cost to health systems and diabetes mellitus (DM) is one of the significant predictors to the development of CKD. The study aimed to determine the association between the duration of DM and CKD staging among T2DM patients.
Methods: A cross-sectional study was conducted using secondary data from the National Diabetes Registry’s (NDR) audited patients for the year of 2018 to 2020 at Padang Terap, Kedah. The eGFR was calculated from serum creatinine value and classified into six categories based on the KDIGO classification. Multinominal logistic regression analysis was used to determine the association.
Results: A total of 963 patients were included in the study. The prevalence of CKD was 38.6%. The majority of T2DM patients were elderly Malay females. The prevalence of stage 1 was 6.95%, stage 2 was 5.4%, stage 3a was 12.35%, stage 3b was 8.10%, stage 4 was 3.86% and stage 5 was 1.93%. In the multivariable analysis, in model 1 (unadjusted) the p-value showed a significant association between duration of DM and CKD staging. By adjusting with other blocks of variables (Model 2 to Model 6) were also found to be in a significant association (p<0.05).
Conclusion: It is concluded that there was an association between the duration of DM and CKD staging. Early detection of CKD and glycemic control are essential to delay the onset of diabetic kidney disease. Diabetes control should be optimized to reduce diabetes-related complications and minimize adverse events.
Keywords: Chronic Kidney Disease, Type 2 Diabetes Mellitus, Prevalence of CKD, Progression factors, Primary Health Care Setting
Introduction
Chronic Kidney Disease (CKD) had emerged as one of the major burdens in the world. It has been reported that the global prevalence of CKD was 13.4% [1]. In 2018, the prevalence of CKD in Malaysia was 15.48% (95% CI: 12.30, 19.31). It was increased compared to the year 2011 where the prevalence of CKD was only 9.07% [2]. Based on the same study, an estimated prevalence according to staging was 3.85% (95%CI: 2.51, 5.87) had stage 1 CKD, 4.82% (95%CI: 3.14, 7.32) had stage 2 CKD, 6.48% (95%CI: 4.41, 9.43) had stage 3 CKD and 0.33% (95%CI: 0.14, 0.78) had stage 4 to 5 CKD. The same study also found that diabetes mellitus (DM) was one of the significant factors for CKD [adj. OR 3.32 (95%CI: 2.20, 5.03)] [2].
As the prevalence of DM in Malaysia is increasing in trend over the years, the prevalence of CKD is estimated to be higher because DM is the significant factor for CKD. In 2015, Singapore reported that the prevalence of CKD among patients with T2DM was 53% [3]. A similar study was carried out in Thailand primary care setting in 2020 showed that the prevalence of CKD among DM patients was 24.4% [4] with 11.4% was stage 3A, 6.8% was stage 3B, 4.6% was stage 4 and 1.6% was stage 5. However, a study by Mubarakali N Janmohamed et al. [5] found that the prevalence of CKD among DM patients was 83.7% with 46.0% was stage 1, 29.2% was stage 2, 12.0% was stage 3A, 8.9% was stage 3B, 3.3% was stage 4, and 0.5% was stage 5. The moderate to severe CKD is estimated to be found in 15% to 23% of patients with DM [6].
Diabetes mellitus (DM) and metabolic syndrome were identified as the most common cause of CKD worldwide that involve multiple mechanisms, including glomerular hemodynamic factors and advanced glycation end product [7]. With DM, the small blood vessels in the kidney are injured. Therefore, the kidneys cannot clean the blood properly causes water and salt retention in the body and the waste material build up in the blood [8]. There were around 45% of patients who received dialysis therapy is caused by kidney failure due to DM [9].
The duration of diabetes mellitus was found to be one of the significant factors for the progression of CKD staging. The longer the duration of diabetes mellitus, the higher the CKD stage. Several types of research similar findings highlight the duration of diabetes as a risk factor in predicting CKD [10,11]. It has been estimated that one-third of diabetics had early kidney disease, and approximately two-thirds of patients were classified with early CKD due to proteinuria [12]. Proteinuria is considered a valuable surrogate endpoint in patients with CKDs and a target for reno- and cardioprotective strategies [13].
Treatment to prevent diabetic kidney disease should begin as early as possible before kidney damage develops [8]. Glycemic control is essential to delay or prevent the onset of diabetic kidney disease and diabetes control should be optimized for DM patients, with measures to reduce diabetes-related complications and minimize adverse events [14]. The “legacy effect” suggests that early intensive glycaemic control can prevent irreversible damage to the kidney [15]. Therefore, the assessment and management of other comorbid diseases, such as hypertension, hyperlipidaemia, hyperphosphatemia, hyperparathyroidism and anaemia is very important in the care of patients with diabetes and CKD [16].
The objective of this study was to determine the association between the duration of DM and CKD Staging Among Type II Diabetes Mellitus Patients in Padang Terap, Kedah and to explore the factor related to the progression of CKD, thus assisting them to have better decision-making when an encounter with CKD patients with multiple risk factors.
Materials and Methods
Underlying Data and Study Design
A cross-sectional study was conducted using secondary data from The National Diabetes Registry (NDR) of Malaysia. NDR was established in 2009 to monitor clinical outcomes of diabetes patients managed at the primary health clinics of the Ministry of Health, Malaysia (MOH). All diabetic patients who receive diabetes care at participating health clinics are required to be registered in this registry. A proportion of patient records are audited annually, and all information about the clinical and treatment is stored in the National Diabetes Registry [17].
Inclusion and Exclusion Criteria
All T2DM patients that have been registered in the NDR under District Health Office, Padang Terap, Kedah and audited in the year 2018 to 2020 were included in this study. Since CKD stages were used as the outcome variable, any patient without serum creatinine value was excluded from the study.
Diagnosis of CKD
The diagnosis of CKD is defined as when the estimated glomerular filtration rate (eGFR) is less than 60 ml/min/1.73 m2 and is present for more than three months with or without evidence of kidney damage or if there is evidence of kidney damage that is present for more than three months with or without eGFR <60 ml/min/1.73 m2 [18]. The presence of proteinuria is considered kidney damage in this study.
The eGFR value requires renal profile blood investigation with a creatinine value. The serum creatinine value from renal profile blood investigation was be converted into eGFR by using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation [18]:
eGFR = 141 x min (SCr/κ, 1)α x max(SCr/κ, 1)-1.209 x 0.993Age x 1.018 [if female] x 1.159 [if Black].
where:
SCr is serum creatinine (in mg/dl) κ is 0.7 for females and 0.9 for males. α is -0.329 for females and -0.411 for males. min indicates the minimum of SCr/κ or 1 max indicates the maximum of SCr/κ or 1
The SCr was conferred to the isotope dilution mass spectrophotometry method to minimise variations in results [19].
Then, the eGFR value was classified into six stages of CKD according to the Kidney Disease: Improving Global Outcomes (KDIGO) classification [20]. Stage 1 CKD is defined when the eGFR ≥ 90 ml/min/1.73m2; Stage 2 CKD is defined when the eGFR is within 60 – 89 ml/min/1.73m2; Stage 3a CKD is defined when the eGFR is within 45 – 59 ml/min/1.73m2; Stage 3b is defined when the eGFR is within 30 – 44 ml/min/1.73m2; Stage 4 CKD is defined when the eGFR is within 15 – 20 ml/min/1.73m2and Stage 5 CKD is defined when the eGFR is ≤ 15 ml/min/1.73m2.
Ethical consideration
Ethical approval for this research was obtained from UiTM Research Ethics Review Boards Committee (REC/04/2021 (MR/197)) and The Medical Research and Ethics Committee (MREC), Ministry of Health Malaysia (MOH) (NMRR-20-3082-57796 (IIR)).
Statistical Analysis
In the descriptive analysis, the categorical data were expressed by the frequency (percentage) while the continuous data were presented by mean ± standard deviation. In the univariate analysis, an unadjusted Odds ratio (OR) in the association between duration of DM and different stages of CKD severity was analyzed by using ANOVA for trend for the continuous data, and Linear by Linear association Chi-Square test for the categorical data.
In the multivariable analysis, the adjusted odds ratio (OR) in the association between duration of DM and different stages of CKD severity was analyzed using the multinomial logistic regression. Model 1 was unadjusted by the duration of diabetes mellitus. Model 2 was adjusted by the sociodemographic data. Model 3 was adjusted by chronic hypertension status. Model 4 was adjusted by anthropometry (BMI). Model 5 was adjusted by the medications and model 6 was adjusted by the biochemical readings to measure the trend of risk measurement across different CKD stages of severity. All the analyses were performed by using the SPSS version 27 software program and a two-sided p-value of less than 0.05 was the criteria for the statistical significance.
Result
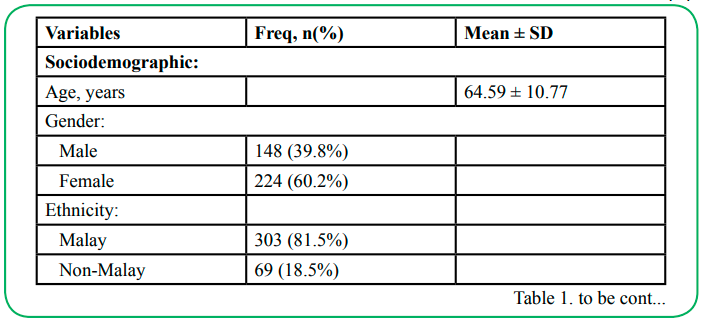
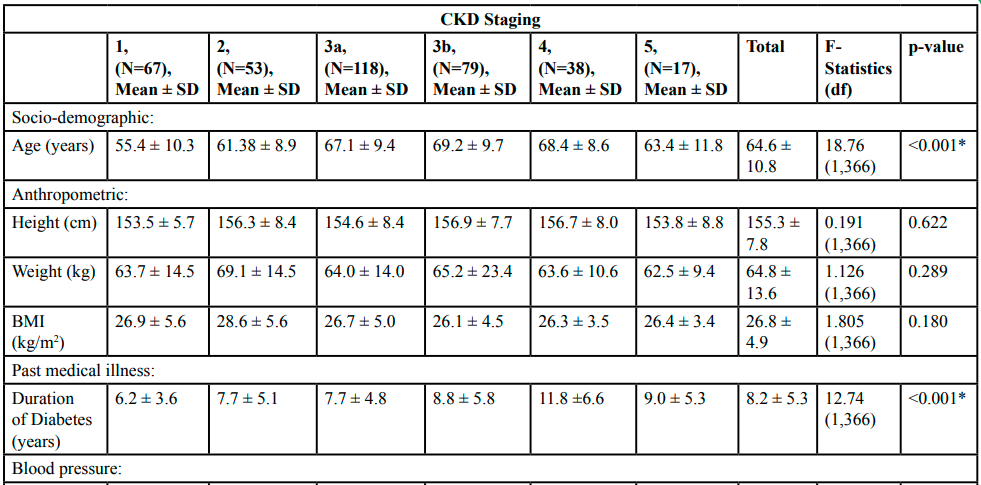
The characteristics of the T2DM patients in PKD Padang Terap, Kedah are shown in Table 1. The majority of T2DM patients with CKD were elderly and Malay females. Most of them were overweight and the mean for the duration of diabetes mellitus among the patients were 8.21 ± 5.38 years. The diastolic blood pressure (DBP) was in good control but the systolic blood pressure (SBP) and the overall HBA1c level were higher than the normal range. The biochemical markers, such as fasting serum lipid was higher than the recommended range for T2DM patients. However, the mean value for high-density lipoprotein (HDL) was within the recommended range which was more than 1.2 mmol/L.
Base on KDIGO Classification, the overall prevalence of CKD among T2DM patients was 38.6% (95% CI: 35.5, 41.70). The prevalence of Stage 1 CKD was 6.95% (95%CI: 3.81, 10.09); stage 2 CKD was 5.40% (995%CI: 2.26, 8.54); stage 3a CKD was 12.35% (95%CI: 9.21, 15.49); stage 3b CKD was 8.1% (95%CI: 4.96, 11.24); stage 4 CKD was 3.86% (95%CI: 0.73, 6.99) and stage 5 CKD was 1.93 (95%CI: 0.10, 5.10).
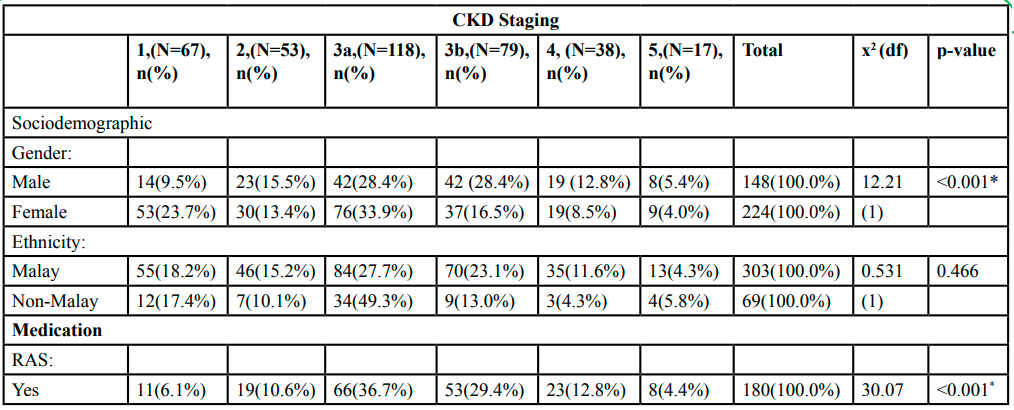
Table 2 and Table 3 shows the distribution of categorical and numerical variables stratified by the CKD staging (stage 1 CKD to stage 5 CKD). For categorical variables, there were significant trends in the severity of CKD for gender (p < 0.001), RAS (p < 0.001), CCB (p = 0.007) and proteinuria. For numerical variables, there were significant trends of the severity for age (p < 0.001), duration of DM (p < 0.001), HbA1c (p < 0.001) and LDL (p = 0.013). The other variables were found no significant trend.
Table 4 shows the unadjusted model (Model 1) (duration of DM) shows a significant trend across the staging of the severity of CKD (p = 0.001). It can be concluded that the longer the duration of DM, the higher the stage of the CKD. There was a significant difference between stages 3a, 3b, 4 and 5 compared to stage 1. The trend of odds is increasing from stage 3a to 4 but is reduced when it reaches stage 5.
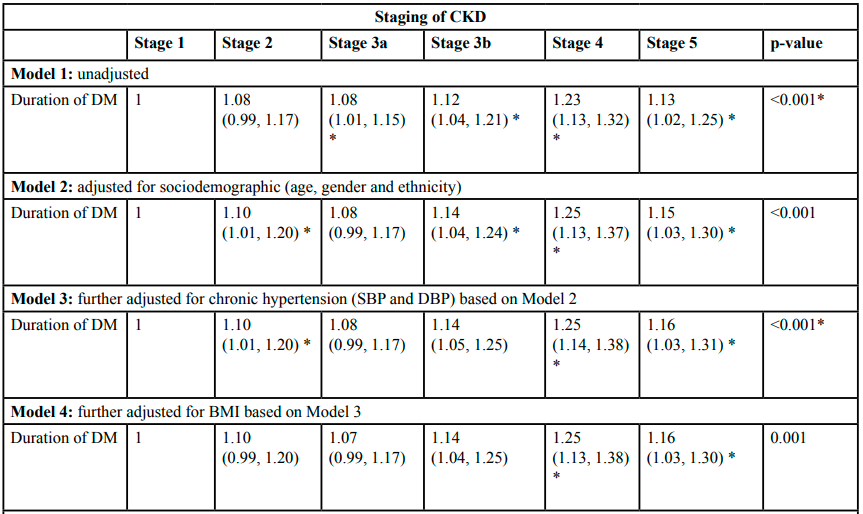
Table 4: Odds and Adjusted Odds Ratios of the duration of diabetes mellitus according to the CKD staging
In model 2, after being adjusted by sociodemographic data (age, gender, and ethnicity), there was a significant trend in the duration of DM across the staging of CKD (p < 0.001). There was a significant difference between stages 2, 3b, 4 and 5 compared to stage 1. The trend of odds is increasing from stage 2 to 4 but is reduced when it reaches stage 5.
In model 3, after being adjusted for chronic hypertension (systolic and diastolic blood pressures) based on Model 2, there was a significant trend across the staging of the severity of CKD (p < 0.001). There was a significant difference between stages 2, 3b, 4 and 5 compared to stage 1. The trend of odds is increasing from stage 2 to 4 but is reduced when it reaches stage 5.
In model 4, after being adjusted by body mass index based on Model 3, there was a significant trend across the staging of the severity of CKD (p < 0.001). There was a significant difference between stages 3b, 4 and 5 compared to stage 1. The trend of odds is increasing from stage 3b to 4 but reduced when it reaches stage 5.
In model 5, after being adjusted by the medications (RAS and CCB) based on Model 4, there was a significant trend across the staging of the severity of CKD (p = 0.001). There was a significant difference between stages 3b and 4 compared to stage 1. The trend of odds is increasing from stage 3b to 4.
In model 6, after being adjusted by biochemical readings based on Model 5, there was a significant trend across the staging of the severity of CKD (p = 0.001). There was a significant difference between stages 3b, 4 and 5 compared to stage 1. The trend of odds is increasing from stage 3b to 4. The trend of odds is increasing from stage 3b to 4 but reduced when it reaches stage 5.
In Stage 2 CKD, after being adjusted by sociodemographic data (Model 2), the adjusted OR increases from 1.08 to 1.10. It could be due to when the age increases, the adjusted OR will also increase. There was no difference in the adjusted OR between Model 2, Model 3 (adjusted for chronic hypertension) and Model 4 (adjusted for BMI). However, in Model 5 (adjusted by medication (RAS and CCB), the adjusted OR has a slight decrease from 1.10 to 1.09. It could be that the medication has improved the condition of severity in stage 2 CKD. In Model 6 (adjusted by biochemical readings), the OR has increased from 1.09 to 1.12. It could be that the biochemical readings were not in a good condition (based on the descriptive statistics), as the value of adjusted OR became higher.
Discussion
The distribution of the diabetes patients with CKD was almost the same with other studies [4,21,22]. Females and elders were the majority of the patients [4,21,22]. The BMI of the patients in this study was overweight and consistent with a study by Bramlage P et al. [21]. However, a study by Jitraknatee et al. [4] found the BMI was at the normal range. The duration of diabetes in this study was longer compared to other studies [4,21]. The level of Hba1c was slightly lower compared to a study by Bramlage P et al. [21]. Malaysia has adopted a yearly audited program by National Diabetes Registries with a target of more than 30% from the randomly selected patient to have HbA1c of less than 6.5% [17].
The SBP and DBP in this study were almost the same as with other studies [4,21,22]. The biochemical markers, such as fasting serum lipid was higher than the recommended range for T2DM patients except for high-density lipoprotein (HDL). However, the biochemical profile in this study was almost the same as in another study [4,21]. 55.9% of the patients in this study were having proteinuria. A study by Marshall S.M [23] suggested that 85% to 100% of diabetic patients with microalbuminuria progressed to proteinuria.
Chronic kidney disease (CKD) is common and can be found in 23% of patients with diabetes [6]. The prevalence of CKD among T2DM patients in this study was 38.6% (95% CI: 35.5, 41.7). This finding was higher compared to the majority of a similar study done in other countries which found the prevalence of CKD of less than 30% [24]. The finding for the prevalence in this study according to different CKD stages found that stages 3a CKD and 3b CKD have higher prevalence as compared to the other stages and it was similar in other researches done in Japan, Canada and Spain [25,26,27]. However, studies done in Finland and China found that most of the CKD patients were presented at stage 1 CKD and stage 2 CKD followed by stage 3 CKD [28,29]. The prevalence of stage 4 CKD and 5 CKD was expected to have the lowest percentage compared to the other stages [24]. However, the low percentage of stages 4 and 5 may be underestimated due to the majority of the study done in the primary health centres while patients with higher stages are required to refer to the nephrologist for further investigations and treatments [18].
This study found that the duration of diabetes is significantly associated with CKD staging. The association is independent of sociodemographic (age, gender and ethnicity), blood pressure (systolic and diastolic), body mass index, medication (RAS and CCB) and biochemical readings (HbA1c%, Total cholesterol, TG, HDL, LDL and proteinuria). The trend was increased in the duration of diabetes mellitus although the prevalence in stage 5 CKD was slightly lower compared to stage 4 CKD (P<0.001) (model 1). Several potential mechanisms may account for the link between the duration of diabetes and CKD staging. The longer duration of diabetes may cause filtering units of the kidney to become narrow and clogged. The kidney is damaged due to this process [8]. The duration of diabetes was also significant even after adjusting with age (model 2). Increasing age leads to increased risk for CKD progression in most studies in similar fields [5,30,31]. The kidneys are affected by the ageing process which resulted from several morphological and functional process alterations along the course; thus the kidney disease has also been found to be more dominant in those over the age of 60 when compared to the rest of the general population [32].
There was a significant trend between gender and CKD staging (model 2). A study done in Spain reported that only the male sex was associated with CKD [33] while another study done in Ghana found the female sex as a predictor for CKD ccurrence [34]. A review report in 2006 found that the incidence of CKD among males was higher because they were more susceptible to age-related glomerulosclerosis than females [35]. The Odds ratio of the duration of diabetes mellitus increases after being adjusted by hypertension across the CKD staging (model 3). It was found that diabetes mellitus, hypertension or a combination of both factors may progress the CKD toward the end-stage of renal failure (ESRF). The incidence of ESRD is up to 10 times higher in those with diabetes mellitus compared to adults with no diabetes [24].
The Odds ratio of the duration of diabetes mellitus increases after being adjusted by body mass index (BMI) in model 4 (p < 0.001). Most studies found that obesity is the predictor for CKD instead of being overweight [35]. Hormones and cytokines secreted from adipose tissue contributes to CKD besides the renal hemodynamic changes that contribute to the development of obesity-related glomerulopathy [36].
The number of CKD patients receiving Renal Angiotensin System blocker (RAS) and Calcium Channel Blocker (CCB) were lower in stage 1 CKD and stage 2 CKD, while the usage of the medications was higher in stage 3a CKD until stage 5 CKD. However, based on the guideline in the management of CKD in 2018 suggest optimizing the use of RAS for the renoprotective effect in CKD patients for stages 1 and 2 CKDs [18]. The Odds ratio of the duration of diabetes mellitus decreases significantly after being adjusted by medication (RAS and CCB) (model 5). It can be concluded that treating patients with these two drugs can reduce the odds of progression of CKD. Dose adjustment may require in medication therapy for diabetes patients with CKD. The assessment and management of comorbid diseases, including hypertension, hyperlipidaemia, anaemia, hyperphosphatemia, and hyperparathyroidism, is important in the care of patients with diabetes and CKD [6].
In Model 6, the odds ratio of the duration of diabetes was significantly increased after being adjusted by the biochemical profile and proteinuria status in stage 3b CKD until stage 5 CKD. The findings can be explained by the distribution of biochemical markers with poor control. The lipid profile is significantly associated with the development of CKD. Total cholesterol, LDL, and triglyceride were found to be CKD risk factors while HDL was a protective factor [37].
Previous research on T2DM patients found that CKD was associated with a higher value of Hba1c [3,4]. However, a study by Shipman et al., [38] found the mean Hba1c was higher in stages 3, stage 4 and stage 5 CKD as compared to stages 1 and 2 CDs. There are two explanations for the lower Hba1c level in a higher stage of CKD. First, all patients with stages 4 and 5 CKDs need to be referred to a Family Medicine Specialist based on the current guideline in primary care settings [18]. Sometimes, the patients need to be referred to the nephrologist if indicated such as requiring dialysis [18]. Hence, the management of the patients will be optimized by the physician reflecting in lower value of HbA1c in stages 4 and stage 5 CKDs. Second, anaemia is recognized as one of the CKD complications while the Hba1c parameter is highly dependent on the concentration of haemoglobin in the patient's blood. The occurrence of severe anaemia is commonly present among higher stages of CKD especially in ESRD patients, thus reflecting in the reduction value of Hba1c across different stages of severity [39].
The presence of proteinuria proportion was 100% in stages 1 and stage 2 CKDs. It is due to the definition of CKD in patients with eGFR of less than 60 requiring evidence of kidney damage [40]. The presence of proteinuria is one of the evidence of kidney damage, thus the result proportion of 100% reflects the definition for the diagnosis of CKD in stages 1 and 2 CKD patients [20]. In this study, proteinuria was also a significant factor in CKD progression across the stages of severity (p < 0.05). Proteinuria is not only can be used as a diagnostic value for CKD but it can also be used as a prognostic value in CKD. It is accepted as the marker for kidney damage, usually associated with lower eGFR value and increased end stage of renal disease (ESRD) occurrence risk [41]. The presence of proteinuria was the major risk factor for CKD in diabetic patients and also among the diagnosed hypertension, undiagnosed hypertension, and pre-hypertension patients [42]. Thus, medications such as RAS blocker is recommended to reduce the incidence of proteinuria subsequently reduce the risk of CKD progression among the patients.
Limitation of the study
The research findings should be interpreted in the context of limitations. This study was conducted by using secondary data which may not provide all of the interesting information. Based on the theoretical framework, several variables were not presented in the National Diabetes Registry (NDR) because the system has been developed to answer different objectives [43]. The secondary data has a lack of control over the quality of data because the researcher did not participate in the data collection processes [44]. Next, the cross-sectional observational study has limitations in terms of the analyses of causal inference as the study design was unable to measure temporality based on Bradford Hill's criteria [45].
Conclusion
There was an association between the duration of DM and CKD staging even after adjusting by several blocks of confounders. Early detection of CKD and glycemic control are essential to delay or prevent the onset of diabetic kidney disease. Diabetes control should be optimized to reduce diabetes-related complications and minimize adverse events.
Abbreviations:
BMI: Body mass index; CCB: Calcium Channel Blocker; CKD: Chronic Kidney Disease; CKD-EPI: Chronic Epidemiology Collaboration Equation; eGFR: estimated glomerular filtration rate; HDL: high-density lipoprotein; KDIGO: Kidney Disease Improving Global Outcome; LDL: Low-density lipoprotein; NDR: National Diabetes Registry; OR: Odds ratio RAS: Renin-Angiotensin System; T2DM: Type II Diabetes Mellitus;
Acknowledgement:
We would like to acknowledge the immense contribution of study participants for agreeing to be part of the study.
Author’s contributions:
The authors were responsible for the research proposal, questionnaire design, collecting data, data analysis and interpretation and manuscript writing. The authors read and approved the final manuscript.
Funding
This is self-funding study
Availability of data and materials
The datasets used and/or analyzed are available from the corresponding author on reasonable request.
Declarations
Ethics approval and consent to participate
This study was conducted in accordance with the Institutional Research Ethics and the declaration of Helsinki. Ethical approval for this research was obtained from UiTM Research Ethics Review Boards Committee (REC/04/2021 (MR/197)) in January 2021 and The Medical Research and Ethics Committee (MREC), Ministry of Health Malaysia (MOH) (NMRR-20-3082-57796 (IIR)) on March 2021. The anonymity of the participants is guaranteed. Informed consent was not needed because this study was using secondary data from NDR Malaysia.
Consent for publication
Not applicable.
Competing interests:
The author declares that he has no competing interests.
References
Hill, N. R., Fatoba, S. T., Oke, J. L., Hirst, J. A., O'Callaghan, C. A., Lasserson, D. S., & Hobbs, F. D. (2016). Global Prevalence of Chronic Kidney Disease - A Systematic Review and Meta-Analysis. PLoS One, 11(7), e0158765. https://doi.org/10.1371/ journal.pone.0158765View
Saminathan, T. A., Hooi, L. S., Mohd Yusoff, M. F., Ong, L. M., Bavanandan, S., Rodzlan Hasani, W. S., Tan, E. Z. Z., Wong, I., Rifin, H. M., Robert, T. G., Ismail, H., Wong, N. I., Ahmad, G., Ambak, R., Othman, F., Abd Hamid, H. A., & Aris, T. (2020). Prevalence of chronic kidney disease and its associated factors in Malaysia; findings from a nationwide population-based cross-sectional study. BMC Nephrol, 21(1), 344. https://doi. org/10.1186/s12882-020-01966-8 View
Low, S.K.M, Sum, C.F, Yeoh L.Y, Tavintharan, S., Ng, X.W, Lee, S.B.M, Tang, W.E.e, & Lim, S.C. (2015). Prevalence of Chronic Kidney Disease in Adults with Type 2 Diabetes Mellitus. Ann Acad Med Singap, 44(5), 164-171. View
Jitraknatee, J., Ruengorn, C., & Nochaiwong, S. (2020). Prevalence and Risk Factors of Chronic Kidney Disease among Type 2 Diabetes Patients: A Cross-Sectional Study in Primary Care Practice. Sci Rep, 10(1), 6205. https://doi.org/10.1038/ s41598-020-63443-4 View
Mubarakali N Janmohamed, Samuel E Kalluvya, Andreas Mueller, Rodrick Kabangila, Luke R Smart, Jennifer A Downs, & Robert N Peck. (2013). Prevalence of chronic kidney disease in diabetic adult out-patients in Tanzania. BMC Nephrology, 14, 183. View
Coresh, J., Astor, B.C., Greene, T., Eknoyan, G., & Levey, A.S. (2003). Prevalence of chronic kidney disease and decreased kidney function in the adult US population: Third national health and nutrition examination survey. American Journal of Kidney Diseases, 41(1), 1-12. https://doi.org/10.1053/ajkd.2003.50007View
Amin, R., Turner, C., van Aken, S., Bahu, T. K., Watts, A., Lindsell, D. R., Dalton, R. N., & Dunger, D. B. (2005). The relationship between microalbuminuria and glomerular filtration rate in young type 1 diabetic subjects: The Oxford Regional Prospective Study. Kidney Int, 68(4), 1740-1749. https://doi. org/10.1111/j.1523-1755.2005.00590.xView
National Kidney Foundation. (2020). Diabetes - A Major Risk Factor for Kidney Disease. https://www.kidney.org/atoz/ content/diabetesView
Bethesda M.D. (2006). U.S. Renal Data System, USRDS 2006 Annual Data Report: Atlas of End-Stage Renal Disease in the United States. http://www.usrds.org.
Alemu, H., Hailu, W., & Adane, A. (2020). Prevalence of Chronic Kidney Disease and Associated Factors among Patients with Diabetes in Northwest Ethiopia: A Hospital-Based Cross-Sectional Study. Curr Ther Res Clin Exp, 92, 100578. https:// doi.org/10.1016/j.curtheres.2020.100578 View
Metsarinne, K., Broijersen, A., Kantola, I., Niskanen, L., Rissanen, A., Appelroth, T., Pontynen, N., Poussa, T., Koivisto, V., Virkamaki, A., & Investigators, S. T. o. N. i. D. S. (2015). High prevalence of chronic kidney disease in Finnish patients with type 2 diabetes treated in primary care. Prim Care Diabetes, 9(1), 31-38. https://doi.org/10.1016/j.pcd.2014.06.001View
Wen, C.P, Chang, C.H, Tsai, M.K., Lee, J.H., Lu, P.J., Tsai, S.P., Wen, C., Chen, C.H., Kao, C.H., Tsao, C.K., & Wu, X. (2017). Diabetes with early kidney involvement mayshorten life expectancy by 16 years. Basic Research, 92(2), 388-396. https://doi. org/10.1016/j.kint.2017.01.030View
Cravedi, P., & Remuzzi, G. (2013). Pathophysiology of proteinuria and its value as an outcome measure in chronic kidney disease. British Journal of Clinical Pharmacology, 76(4), 516-523. View
Hahr, A.J., & Molitch, M.E. (2015). Management of diabetes mellitus in patients with chronic kidney disease. Clinical Diabetes and Endocrinology, 1, 2. https://doi.org/10.1186/ s40842-015-0001-9View
Tonna, S., El-Osta, A., Cooper, M.E., & Tikelli, C. (2010). Metabolic memory and diabetic nephropathy: potential role for epigenetic mechanisms. Nat Rev Nephrol, 6(6), 332-341. https://doi. org/10.1038/nrneph.2010.55. Epub View
Cavanaugh, K.L. (2007). Diabetes Management Issues for Patients with Chronic Kidney Disease. Clinical Diabetes, 25(3), 90-97.View
Ministry of Health Malaysia. (2020). National Diabetes Registry (NDR) Report 2013-2019. View
Ministry of Health Malaysia. (2018). CPG Management of Chronic Kidney Disease (Second Edition). Medical Development Division, Ministry of Health Malaysia.
Ministry of Health Malaysia. (2011). Management of Chronic Kidney Disease in Adults. Medical Development Division, Ministry of Health Malaysia.View
KDIGO. (2012). Kidney International Supplements. KDIGO Clinical Practice Guideline for the Management of Blood Pressure in Chronic Kidney Disease (Vol. 2). http://www. kidney-international.orgView
Bramlage, P., Lanzinger, S., van-Mark, G., Hess, E., Fahrner, S., Heyer, C.H.J., Friebe, M., Seufert, J., Danne, T., & Holl, R.W. (2019). Patient and disease characteristics of type-2 diabetes patients with or without chronic kidney disease: an analysis of the German DPV and DIVE databases. Cardiovasc Diabetol, 18, 33. View
Tuttle, K.R., Alicic, R.Z., Duru, O.K., Jones, C.R., Daratha, K.B., Nichola, S.B., McPherson, S.M., Neumiller, J.J., Bell, D.S., Mangione, C.M., & Norris, K.C. (2019). Clinical Characteristics of and Risk Factors for Chronic Kidney Disease Among Adults and Children An Analysis of the CURE-CKD Registry. JAMA Network Open, 2(12), e1918169. https://doi.org/10.1001/ jamanetworkopen.2019.18169View
Marshall, S.M. (2014). Natural History and Clinical Characteristics of CKD in Type 1 and Type 2 Diabetes Mellitus. Advances in Chronic Kidney Disease, 21(3), 267-272. https:// doi.org/10.1053/j.ackd.2014.03.007 View
Koye, D. N., Magliano, D. J., Nelson, R. G., & Pavkov, M. E. (2018). The Global Epidemiology of Diabetes and Kidney Disease. Adv Chronic Kidney Dis, 25(2), 121-132. https://doi. org/10.1053/j.ackd.2017.10.011View
Ohta, M., Babazono, T., Uchigata, Y., & Iwamoto. (2010). Comparison of the prevalence of chronic kidney disease in Japanese patients with Type 1 and Type 2 diabetes. 27(9), 1017- 1023.View
Rodriguez-Poncelas, A., Garre-Olmo, J., Franch-Nadal, J., Diez-Espino, J., Mundet-Tuduri, X., Barrot-De la Puente J, Coll-de Tuero, G., & RedGDPS Study Group. (2013). Prevalence of chronic kidney disease in patients with type 2 diabetes in Spain: PERCEDIME2 study. BMC Nephrology, 4, 46.View
Tonelli, M., Wiebe, N., Richard, J. F., Klarenbach, S. W., & Hemmelgarn, B. R. (2019). Characteristics of Adults With Type 2 Diabetes Mellitus by Category of Chronic Kidney Disease and Presence of Cardiovascular Disease in Alberta Canada: A Cross-Sectional Study. Can J Kidney Health Dis, 6, 2054358119854113. https://doi.org/10.1177/2054358119854113View
Duan, J.-Y., Duan, G.-C., Wang, C.-J., Liu, D.-W., Qiao, Y.-J., Pan, S.-K., Jiang, D.-K., Liu, Y., Zhao, Z.-H., & Liang, L.-L. J. B. n. (2020). Prevalence and risk factors of chronic kidney disease and diabetic kidney disease in a central Chinese urban population: a cross-sectional survey. 21, 1-13.View
Metsärinne, K., Bröijersen, A., Kantola, I., Niskanen, L., Rissanen, A., Appelroth, T., Pöntynen, N., Poussa, T., Koivisto, V., & Virkamäki, A. J. P. c. d. (2015). High prevalence of chronic kidney disease in Finnish patients with type 2 diabetes treated in primary care. 9(1), 31-38. View
Lu, B., Song, X., Dong, X., Yang, Y., Zhang, Z., Wen, J., Li, Y., Zhou, L., Zhao, N., Zhu, X., & Hu, R. (2008). High prevalence of chronic kidney disease in population-based patients diagnosed with type 2 diabetes in downtown Shanghai. J Diabetes Complications, 22(2), 96-103. https://doi.org/10.1016/j. jdiacomp.2007.08.001 View
Nazzal, Z., Hamdan, Z., Masri, D., Abu-Kaf, O., & Hamad, M. (2020). Prevalence and risk factors of chronic kidney disease among Palestinian type 2 diabetic patients: a cross-sectional study. BMC Nephrol, 21(1), 484. https://doi.org/10.1186/ s12882-020-02138-4View
Nitta, K., Okada, K., Yanai, M., & Takahashi, S. (2013). Aging and chronic kidney disease. Kidney Blood Press Res, 38(1), 109-120. https://doi.org/10.1159/000355760View
De Cosmo, S., Viazzi, F., Pacilli, A., Giorda, C., Ceriello, A., Gentile, S., Russo, G., Rossi, M. C., Nicolucci, A., Guida, P., Pontremoli, R., & Group, A. M.-A. S. (2016). Predictors of chronic kidney disease in type 2 diabetes: A longitudinal study from the AMD Annals initiative. Medicine (Baltimore), 95(27), e4007. https://doi.org/10.1097/MD.0000000000004007 View
Tannor, E. K., Sarfo, F. S., Mobula, L. M., Sarfo-Kantanka, O., Adu-Gyamfi, R., & Plange-Rhule, J. (2019). Prevalence and predictors of chronic kidney disease among Ghanaian patients with hypertension and diabetes mellitus: A multicenter cross-sectional study. J Clin Hypertens (Greenwich), 21(10), 1542- 1550. https://doi.org/10.1111/jch.13672View
Taal, M. W., & Brenner, B. M. (2006). Predicting initiation and progression of chronic kidney disease: Developing renal risk scores. Kidney Int, 70(10), 1694-1705. https://doi.org/10.1038/ sj.ki.5001794View
Garland JS. (2014). Elevated body mass index as a risk factor for chronic kidney disease: current perspectives. Diabetes, Metabolic Syndrome and Obesity: Targets and Therapy, 7, 347-355.View
Zhang, Y.-B., Sheng, L.-T., Wei, W., Guo, H., Yang, H., Min, X., Guo, K., Yang, K., Zhang, X., He, M., Wu, T., & Pan, A. (2020). Association of blood lipid profile with incident chronic kidney disease: A Mendelian randomization study. Atherosclerosis, 300,19-25.https://doi.org/https://doi.org/10.1016/j. atherosclerosis.2020.03.020View
Shipman, K. E., Jawad, M., Sullivan, K. M., Ford, C., & Gama, R. (2015). Effect of chronic kidney disease on A1C in individuals being screened for diabetes. Primary Care Diabetes, 9(2), 142- 146. https://doi.org/https://doi.org/10.1016/j.pcd.2014.05.001View
Sinh, N., Mishra T.K, Singh T, & Gupta N. (2012). Effect of Iron Deficiency Anemia on Hemoglobin A1c Levels. Ann Lab Med, 32(1), 17-22. https://doi.org/10.3343/alm.2012.32.1.17View
Perico, N., Codreanu, I., Schieppati, A., & Remuzzi, G. (2005). Pathophysiology of disease progression in proteinuric nephropathies. Kidney Int Suppl(94), S79-82. https://doi. org/10.1111/j.1523-1755.2005.09420.xView
Obi, Y., Kimura, T., Nagasawa, Y., Yamamoto, R., Yasuda, K., Sasaki, K., Kitamura, H., Imai, E., Rakugi, H., Isaka, Y., & Hayashi, T. (2010). Impact of age and overt proteinuria on outcomes of stage 3 to 5 chronic kidney disease in a referred cohort. CLin J Am Soc Nephrol, 5(9), 1558-1565. https://doi. org/10.2215/CJN.08061109View
Crews, D. C., Plantinga, L. C., Miller, E. R., 3rd, Saran, R., Hedgeman, E., Saydah, S. H., Williams, D. E., Powe, N. R., Centers for Disease, C., & Prevention Chronic Kidney Disease Surveillance, T. (2010). Prevalence of chronic kidney disease in persons with undiagnosed or prehypertension in the United States. Hypertension, 55(5), 1102-1109. https://doi.org/10.1161/ HYPERTENSIONAHA.110.150722View
Pederson, L., Vingilis, E., Wickens, C., Koval, J., & Re, M. (2020). Use of secondary data analyses in research: Pros and Cons. Journal of Addiction Medicine and Therapeutic Science, 058-060. https://doi.org/10.17352/2455-3484.000039View
Johnston Melissa P. (2014). Secondary Data Analysis: A Method of which the Time Has Come. Qualitative and Quantitative Methods in Libraries (QQML) 3, 619-626.View
Fedak, K. M., Bernal, A., Capshaw, Z. A., & Gross, S. (2015). Applying the Bradford Hill criteria in the 21st century: how data integration has changed causal inference in molecular epidemiology. Emerg Themes Epidemiol, 12, 14. https://doi. org/10.1186/s12982-015-0037-4View