Journal of Public Health Issues and Practices Volume 6 (2022), Article ID: JPHIP-200
https://doi.org/10.33790/jphip1100200Research Article
Single Leg Balance, Vestibular Input, Turns During Gait and Cognitive Components as Predictors of Gait Speed Alteration in Hispanic Latinx Participants Living with HIV
Martín G. Rosario PT, PhD, CSFI, ATRIC, Kennedy Kessler SPT, Taryn Myers SPT, & Leah Jamison PT, DPT
1Assistant Professor, School of Physical Therapy, Texas Woman’s University, Dallas Campus, 5500 Southwestern Medical Ave. Dallas, TX 75235-7299. United States.
Corresponding Author Details: Martín G. Rosario PT, PhD, CSFI, ATRIC, Assistant Professor, School of Physical Therapy, Texas Woman’s University, Dallas Campus, 5500 Southwestern Medical Ave. Dallas, TX 75235-7299. United States. E-mail: mro-sario1@twu.edu
Received date: 18nd March, 2022
Accepted date: 12th April, 2022
Published date: 15th April, 2022
Citation: Rosario, M.G., Kessler, K., Myers, T., & Jamison, L., (2022). Single Leg Balance, Vestibular Input, Turns During Gait and Cognitive Components as Predictors of Gait Speed Alteration in Hispanic Latinx Participants Living with HIV. J Pub Health Issue Pract 6(1): 200.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Those living with HIV experience a number of complications that negatively affect gait. Gait speed is an important predictor of overall health, and people living with the virus experience an accelerated decline as the disease progresses.
Purpose: To identify components associated with gait speed decreased.
Methods: Thirty individuals diagnosed with HIV, a stable immune system (cd4=878.5) and aged 60.31±7.8 years (19 males, and 11 females) were recruited for this study from a clinical fitness specialized in HIV. Participants completed the HIV dementia scale, vestibular fukuda test, 5 challenging single leg balance tests, 5 time sit to stand, and a 7-meter walk.
Results: A multiple linear regression analysis revealed the association of all components to the prediction of gait speed (R2 = [.963], F(df regression =15, df residual=6) = [F-10.5], p = [p-0.004]) except for the lower limb strength.
Conclusion: Exercise interventions that include vestibular stimulation, such as head rotations, single limb activities, and dynamics turning tasks, should be added to a 3 point (cardio, strengthening and stretching) exercise program. Physical therapists and exercise physiologists should assess and treat all those aforementioned components.
Introduction
According to UNAIDS, 37.7 million individuals worldwide live with human immunodeficiency virus (HIV) disorder in 2020, while approximately 1.5 million were newly infected in 2020. HIV eventually evolves into acquired immunodeficiency syndrome (AIDS), which progresses without treatment, causing many problems, including declining motor function [1]. Reductions in motor function begin when HIV invades CD4 cells, causing a decline in the immune system. Microglial cells serve as a link between the immune system and the central nervous system (CNS). Therefore, if the immune system is deteriorating, the prolonged activation of microglial cells leads to neurological deficits that directly impact motor function [2]. Additionally, the continued decline of the immune system can cause brain injuries, leading to impaired postural stability and psychomotor decline [3]. Therefore, further assessment of changes in motor function in those living with HIV is essential to preserve the quality of life.
Balance is an aspect of motor function commonly altered in those living with HIV [4]. Three sensory systems regulate balance: visual, vestibular, and proprioceptive systems [5]. Gait and balance deficits have been shown to exist in people living with HIV (PLHIV), including slower gait speed and increased center of pressure excursions [6,7]. In addition, challenging static balance tasks has shown increased postural sway [8,9] and decreased functional base of support (BoS) [10] in PLHIV. Evidence has also reported impaired limits of stability and sway strategies in PLHIV [11]. As stated before, visual, vestibular, and proprioceptive systems manage balance, which can all be negatively affected by the progression of HIV [8,12,13]. Based on the information above, it is apparent that balance deficits exist in PLHIV, influencing different aspects of gait.
Specific balance deficits can be related to alterations to the lower leg neuromuscular musculature [14], decreased function of the vestibular system [13], and proprioceptive input [8]. Balance deficits seen in PLHIV can be compared to balance deficits of fall-associated parameters in the elderly [6]. Some of these parameters are related to reduced functional BoS [10], a significant decrease in stability limits, and a reduction in standing balance [10]. The obstacle is that even in the asymptomatic stage before fall events, those with HIV showed a reduction in balance and postural sway [14]. These postural alterations cause significant difficulties due to significant balance modifications and complications that are not readily seen until it is too late and the quality of life of PLHIV is impacted [15].
Commonly, the quality of life in PLHIV is influenced when experiencing falls associated with postural instability [16]. These above-mentioned postural variations also influence dynamic balance, such as gait speed [11,17]. PLHIV experience a prompt decline in gait speed than those HIV negative [17], which affects their quality of life. Further, research has established that PLHIV shows increased gait difficulty among challenging conditions, such as dual-task activities [18] and demanding conditions [6] involving the interplay of the balance sensory systems.
The conflict with attempting to single out proper intervention strategies for PLHIV is that these gait and balance issues are related to multifactorial components [19]. In order to reduce or slow down the progression of HIV complications that influence gait and balance, it is imperative to identify which components are directly involved. Therefore, the present investigation aims to determine the various components that negatively impact gait. Identifying these components can facilitate the development of tailored interventions, enhancing balance and thus the quality of life.
Methods
Procedures
IRB and the HIV institution approved this study (protocol # FY2020-32). Participants were recruited from La Perla de Gran Precio (LDGP) in San Juan, Puerto Rico. All participants signed the consent documents before data collection. In addition, all demographic information and baseline vitals were collected during the screening process prior to data collection, including SaO2, heart rate, and the use of glasses and contact lenses. Then, participant data collection started, which included completion of the International HIV Dementia Scale (IHDS), five times sit-to-stand test (5xSTS), and the Fukuda stepping test (FST), 5 balance activities and gait tasks.
International HIV Dementia Scale
IHDS, an assessment to determine a possible diagnosis of dementia (https://www.hiv.uw.edu/page/mental-health-screening/ihds). The IHDS contains four components: memory registration, motor speed, psychomotor speed, and memory recall. The memory registration component instructs the participants to say four words: "dog, hat, bean, red." If the participants preferred Spanish, the four words were spoken in Spanish as "perro, sombrero, habichuela, rojo." Each component was scored from 0-4, with zero indicating "unable to perform" the task. If the summed score was ≤ 10, more testing should be performed due to the possibility of dementia.
Fukuda Stepping Test
The FST assesses alterations to the vestibular system. Specifically, the test examines labyrinthine function via vestibulospinal reflexes. Instructions were given to the participants to stand on a grid to grade displacement and rotation while performing the tasks, marching in place with eyes close. For these participants' ages, the normal rotation range should be between 26.1-42.1 degrees (https://www.sralab.org/ sites/default/files/2017-06/Fukuda_Stepping_Test_SAMPLE.pdf).
Five Time Sit to Stand
The 5xSTS examines lower-extremity functional mobility and strength. A free-standing chair was used during the assessment for all participants. Participants cross their arms over their chest, stand up, and sit down five times as quickly as possible. The time started when the tester stated "go" and stopped when the participant touched the chair on the fifth repetition.
Balance Assessment
After the above assessment, single-limb stance balance tests for medial-lateral (ML) and anterior-posterior (AP) sway direction, velocity, and distance were performed with the MobilityLab APDM MobilityLabTM (APDM, Inc., http://apdm.com).
The assessment aimed to maintain balance until the signaling tone ended the trial. Verbal instructions were given prior to each trial, while the participants rested in a standing position on a firm surface. The participant balanced on their dominant lower extremity 10 feet away from a blank white wall that displayed two crossed pieces of black tape at eye level for participants to gaze towards. The trial lasted 15s, followed by the completion of a tone signaling trial. If necessary, the participant could regain balance during the trial by utilizing the contralateral limb and continuing until the trial was complete. During each trial, one researcher recorded the number of times foot touchdowns occurred on the contralateral limb, and another researcher stood behind the participant to protect against falls. The specific balance activity was conducted as follows.
Balance 1: The trial was considered a baseline assessment. The participants were instructed to maintain a double-leg stance (DL) on a firm surface with their eyes open.
Balance 2: The participants performed a single-leg stance (SL firm) on a firm surface with eyes open. This trial aimed to reduce the BoS, which challenges balance.
Balance 3: Prior to starting tone, the participants stood on their dominant lower extremity (SL) and performed rhythmic cervical flexion and extension, also known as head up and down (HUD), to a 60 bpm metronome while maintaining their gaze on the black tape. At the start of the tone, the participants closed their eyes (SL firm ECHUD). This trial aimed to cancel visual input, challenge the vestibular input, and conserve proprioceptive input.
Balance 4: The participants balanced in a single-leg stance (SL) on a foam surface with eyes open prior to starting tone. At the start of the tone, the participants performed head up and down (HUD) to a 60 bpm metronome (SL foam EOHUD). This trial aimed to challenge vestibular and proprioceptive inputs while preserving visual input.
Balance 5: The participants maintained a single-leg stance (SL) on the foam surface before starting the tone. The subjects closed their eyes (SL foam EC). This trial aimed to cancel the visual input, challenge proprioceptive input, and conserve the vestibular system.
Gait Assessment
Gait speed was collected using APDM’s Mobility Lab (APDM Inc, http://apdm.com). Rosario et al., [20], published the standardized gait protocol in detail.
Data Analysis and Results
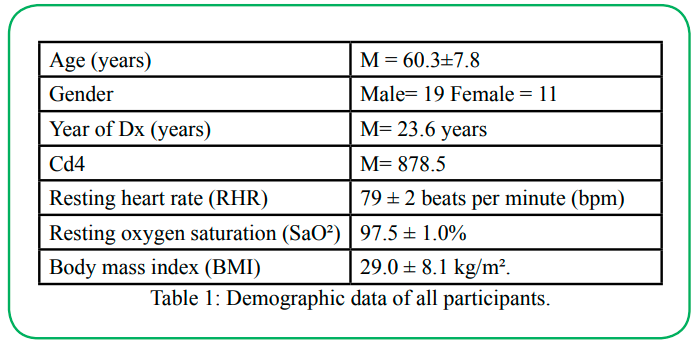
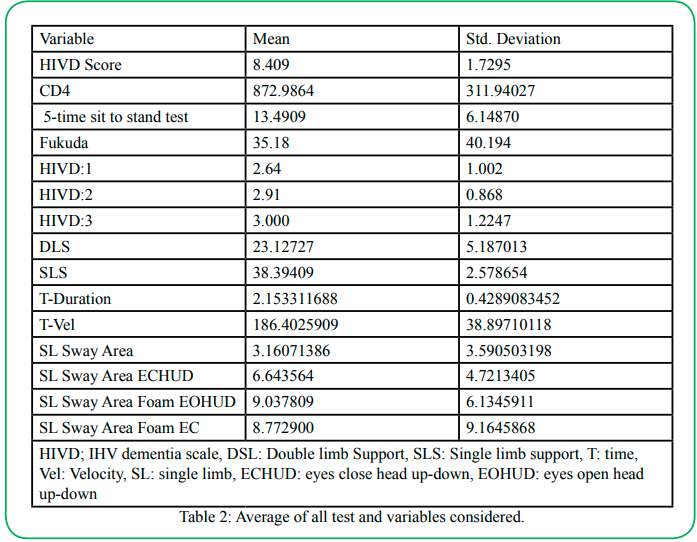
The current study recruited 30 individuals living with HIV aged 60.31±7.8 years, 19 males, and 11 females. Participants had a stable immune system (cd4=878.5), and an average of years from diagnosis was 23.6 years. Additionally, participant information is illustrated in table 1. Table 2 shows the average of all data points collected prior to the analysis.
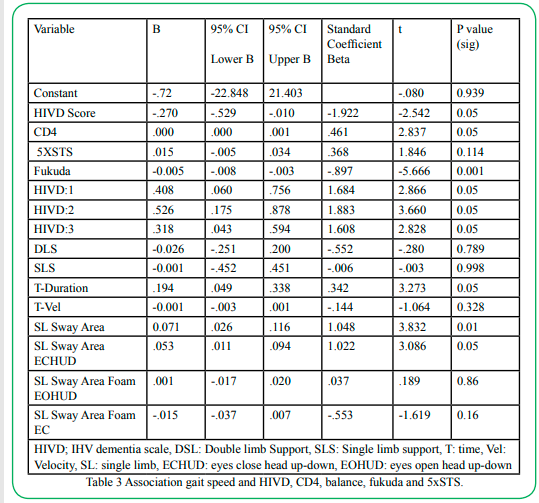
A secondary analysis employing multiple linear regression with SPSS version 28 was used to test if balance (single-leg tests), cognitive (HIVD Scale), vestibular (Fukuda), dynamic gait (turns), and lower strength (5xSTS) components significantly predicted or affected gait speed in those living with HIV. A P value of 0.05 A secondary analysis employing multiple linear regression with SPSS version 28 was used to test if balance (single-leg tests), cognitive (HIVD Scale), vestibular (Fukuda), dynamic gait (turns), and lower strength (5xSTS) components significantly predicted or affected gait speed in those living with HIV. A P value of 0.05.
Balance component: Single-leg balance on firm surface EO and ECHUD significantly predicted gait speed (β =0.071, p =0.001 and β =0.053, p =0.05). It was also found that single-leg balance on foam surface EOHUD and EC did not predict gait speed (β =0.001, p =0.86, and β =-0.015, p =0.16). Cognitive component: Results showed that all three HVID, HIVD1 (β =0.408, p =0.05), HIVD2 (β =0.526, p =0.05) and HIVD3 (β =0.318, p =0.05) parts of the scale significantly predicted gait speed. Vestibular component: The Fukuda test, which tests for peripheral problems, significantly predicted gait speed (β =10.005, p =0.001). Finally, dynamic gait variables time during turns significantly predicted gait speed (β =0.194, p =0.005).
Discussion
This study examined the relationship between diverse components and their impact on gait parameters, such as gait speed. The objective was to discern which components are affected in those with HIV. Therefore, a more appropriate intervention can be formulated to enhance the quality of life by improving gait through working on the affected components. This study identifies multiple components related to gait speed, such as standing balance, cognitive variables, vestibular inputs.
Gait speed is one of the most critical parameters of gait, as it is commonly referred to as the sixth vital sign [21]. Gait speed is correlated with functional abilities, meaning a decline in gait speed indicates deterioration in functional mobility [22]. A reduction in gait speed is associated with increased difficulties in maintaining instrumental activities of daily living, mild cognitive decline, and risk of cardiovascular death [22]. Furthermore, gait speed is used to predict future health status and disease occurrences, which could provide information regarding hospitalization, discharge location, and mortality [21]. Those living with HIV experience a faster decline in gait speed than those without HIV infection [17]. Based on the above, gait speed is an important parameter that should be considered when examining interventions for the HIV population. Therefore, the current work focused on this variable.
Balance component: The first main finding of this study is that a single leg balance on firm surface EO and ECHUD sway significantly affects gait speed. The results show that postural instability affecting gait can be observed by increased sway with EO or without visual input while engaging the vestibular system, which reduces gait speed. In a systematic review, Berner highlighted reductions in SLS time, increased COP sway path, and increased sway velocity [6] as potential factors altered in those living with HIV. These factors affecting posture have been related to increased postural sway in PLHIV [8,9,23]. One of the reasons for a reduced balance and therefore increased fall risk in PLHIV is poor lower extremity muscle performance [24,25]. Nevertheless, the participants in this study had normal lower limb strength for their age range, indicating that other components are responsible for the gait deficits in this group. Hence, we infer that the participants in the current study exhibit balance impairments affecting gait performance through postural instability and alterations in neuromuscular activation [23,7,26].
The main reason that balance is affected in those living with HIV is the progressive course of HIV and the indirect effects of antiretroviral therapy (ART) medications [2,3,6]. HIV leads to an alteration in the immune system, affecting microglial cells, leading to changes in the nervous system, causing balance deficits [2]. It has been shown that pontocerebellar changes occur during the course of HIV, affecting balance [3]. Rosario et al. concluded that comorbidities, specifically peripheral neuropathy, could contribute to changes in motor components, such as gait in PLHIV [27]; but further research is needed over the correlation between peripheral neuropathy and balance deficits [6,14].
Future investigations could include longitudinal studies examining the alterations in balance and gait. Suppose studies look at the course of HIV over time, and explicitly the onset of balance and gait deficits. In that case, an exercise program could be implemented to limit balance deficits and gait speed alterations, and therefore maintain quality of life in those living with HIV. We recommend balance training to reduce postural sway risk of falls and improve gait velocity. Many inquiries have assessed balance training protocols in older people with variable dosages and heterogeneity [28]. Since HIV accelerated the aging progression of those who live with the virus, the ongoing study proposes tackling balance issues as if they were experiencing advanced aging complications. Based on evidence, balance training protocol should promote stability and reduce falls in community-dwelling older adults when incorporated with strength exercise [29]. Overall, addressing balance deficits will contribute to improvements in gait and improve their quality of life [30].
Prior studies determined that the recommended dosage parameters for balance exercise in older adults are: 11-12 weeks, three sessions per week, 31-45 minutes per session for balance exercises [28]. A balance training protocol could include static, dynamic, reactive, and proactive balances [29]. Dynamic balance training could consist of performing normal gait, narrow gait, overlapping gait, and tandem gait with progressions, as described before, in static balance with the addition of direction and rhythm.
Cognitive component: The second outcome of this study showed that the HVID scale significantly predicted gait speed. Previous studies have shown that the HDS is a reliable tool for measuring dementia or the onset of dementia in those living with HIV [31]. This study suggests HDS can also be associated with gait speed. Previous studies have examined the relationship between physical and cognitive function, indicating that these connections are prevalent and can help create intervention strategies for those living with HIV. PLHIV with a faster gait speed also showed better cognitive function than those with a slower gait speed [32]. Seo et al. [33] also identified an association between slower gait speed and cognitive decline.
Furthermore, these cognitive alterations are associated with various brain areas, such as the frontal lobe. The problem with these alterations is that they affect the central nervous system and provoke cognitive decline, among other complications [2]. Since ART medication slows down the progression of the disease, motor cognitive changes manifest slowly, making it difficult to predict how and when it impacts the lifestyle of these people [34]. Based on our findings, future studies could investigate the benefits of gait training combined with cognitive tasks in different stages of the disease. We recommend treadmill gait training and gait on different challenging surfaces, combined with memory registration, motor speed, psychomotor speed, and memory recall tasks.
Vestibular component: The third main finding shows that peripheral vestibular components play a part in gait speed. Evidence from several studies indicates that HIV deteriorates the anatomy and physiology of the vestibular system, which appears to directly affect the central nervous system [35]. Like several pathogens, HIV can localize in the labyrinth, as Smith et al. stated in 1988. The labyrinth is located in the inner ear, consisting of two functions: hearing and balance. In addition, signals travel from the labyrinth to the brain from the vestibulocochlear nerve, which transmits messages to postural muscles to stay balanced. When infection or virus is present, it affects this signal, resulting in disruption of the vestibular system, leading to dizziness or vertigo. (vestibular.org/article/diagnosis-treatment/type of disorders). Previous studies showed a high prevalence of PLHIV present with vestibular dysfunction due to these complications from the disease, based on evidence from Shangase KK et al., [36]. Katijah monitored the vestibular status in a group of PLHIV/AIDS receiving antiretroviral therapy compared to a control group receiving other therapies for the presence of acute vertigo within six months. Their findings revealed that vestibular dysfunction with acute vertigo occurred frequently in the PLHIV/AIDS participant group than in the control group.
The issue is that in light of all other medical conditions associated with HIV, vertigo may be low on the priority list compared to other life-threatening conditions that cause the individual to not report their symptoms for management and assessment [36]. We recommend providing vestibular training exercises that could re-educate the peripheral and central nervous system for those living with HIV by promoting substitution or adaptation to perform their daily living activities. Different exercise approaches have been proposed to address these problems. However, vestibular rehabilitation exercises include many components, such as balance and gait exercises, that help improve postural stability and decrease symptoms [37]. The vestibular exercises include voluntary eye movements, active head movements, active body movements, visual dependency, and somatosensory dependence exercises. For this study, the approaches performed were vestibular exercises for PLHIV, including sensory integration balance training and gaze stabilization training with VOR re-education and cancellation. Finally, vestibular rehabilitation therapy reduces dizziness by provoking head movements in a busy environment, which has been shown to improve mobility, postural stability, and gaze stability [38].
Related to the sensory integration balance training exercises, it included challenging the visual and sensory system by performing standing balance on firm and foam surfaces with eyes open and closed. For the gaze stabilization training, with VOR re-education exercises, we proposed that the patient hold their thumb up an arm's length away, while maintaining a clear visual focus on their thumb while rotating their head left to right in a 45° range. For VOR cancellation, the same technique is performed. However, while moving their head left to right, the arm moves synchronously in the same direction while maintaining eyes on the target (thumb) [38].
Dynamic Gait: The fourth main finding illustrates that the time during turns, but not velocity, impacts gait. The results indicate that an increase in turn duration reduces gait speed in this group. Previous studies have revealed that instability in turns may derive from unique physiological and cognitive demands. It has been speculated that turning is not automatic and involves cognitive processing throughout the whole movement [39]. Studies on specific factors affecting turns have explained that visual acuity affects gait speed and balance in older populations. Therefore, those with poor vision used more cautious walking strategies, such as slower gait and changing midline center of mass, to help them overcome the difficulty of turning [40]. In cases diagnosed with Parkinson's disease, turning is associated with instability, falls, and freezing [41]. Based on our results, we infer that gait is altered in PLHIV due to decreased balance and increased sway, affecting postural control and stability required to execute turns. A review identified that static balance decreased with an increase in overall sway, notably in subjects with severe HIV. Dynamic balance has also been shown to be altered, especially in those who are obese or have chronic HIV symptoms. Factors such as neurocognitive decline have been reported with other common symptoms of HIV, such as executive function, motor skills, and attention/working memory, which have been presented to change gait by reducing speed and increasing initiation time. These components have also contributed to the significant fall rate detected in HIV cases [6]. Future investigations could delve into the more distinct effects of the loss of cognitive function in people with HIV by having subjects with HIV complete cognitive test outcome measures like the MoCA [42] to regulate the rating of cognitive function among HIV cases. Once identifying the information about the level of cognitive function as scored by an outcome measure, we can identify how this influences their ability to turn, as we reviewed that turning is not automatic. We speculate that the reduction in cognitive function in people with HIV could be associated with the duration of the extended turn.
We recommend increasing dynamic and static stability to reduce postural sway help augment faster turns and better dynamic gait. Studies have indicated that implementing a merger of exercises found in the Otago exercise program five times a week for six months showed gains in balance and stability that contributed to improvement in gait. The strength exercises included straight leg raise with ankle weight, prone hamstring curl with ankle weight, side-lying leg raise with ankle weight, and calf and toe raises. The balance exercises included backward walking, walking and turning, sideways walking, heel-toe stand, and heel-toe walking. The program concluded with walking for 30 minutes at an average pace with the usual assistive device. Improvements were observed in turn-time, turn-sway, and balance and strength testing. It is suggested that a combination of strength training of the lower extremity muscle and balance training exercises can decrease postural sway by increasing static and dynamic stability. The improvement of stability will lead to advances in dynamic gait and performing turns [43].
Conclusion
This current study aimed to identify factors that could alter gait variables, such as gait speed. Findings indicate that components such as balance, HIV dementia scale, vestibular, and the time during turns predicted gait speed modifications in PLHIV. This investigation ushers in making a connection between the above variables and gait. Since reduced gait speed is an essential indicator of diminished quality of life, identifying elements to supplement intervention programs is crucial to slow down the progression of HIV complications. Future research should look into including aspects, such as the ones pointed out in this study, in addition to a three-point exercise program (cardiovascular, strengthening, and stretching). Based on the results of this study, we recommend implementing exercises that incorporate vestibular challenge, balance, and dynamic aspects of gait, such as the turns.
Declaration
Funding:
Partially funded by Texas Woman’s University Woodcock Institute Research Grant
Conflicts of interest/Competing interests:
Authors report no conflict or competing interest.
Ethics approval:
IRB approval TWU protocol # FY2020-32
Consent to participate:
The participant gave signed consent for this study
Authors' contributions:
All authors contributed to the study's conception and design.
References
M Elicer, I., Byrd, D., Clark, U. S., Morgello, S., & Robinson-Papp, J. (2018). Motor function declines over time in human immunodeficiency virus and is associated with cerebrovascular disease, while HIV-associated neurocognitive disorder remains stable. Journal of neurovirology, 24(4), 514–522. https://doi. org/10.1007/s13365-018-0640-6View
Rojas-Celis, V., Valiente-Echeverría, F., Soto-Rifo, R., & Toro-Ascuy, D. (2019). New Challenges of HIV-1 Infection: How HIV-1 Attacks and Resides in the Central Nervous System. Cells, 8(10), 1245. https://doi.org/10.3390/cells8101245.View
Sullivan, E.V., Rosenbloom, M.J., Rohlfing, T. et al. Pontocerebellar contribution to postural instability and psychomotor slowing in HIV infection without dementia. Brain Imaging and Behavior 5, 12–24 (2011). https://doi.org/10.1007/ s11682-010-9107-yView
Rosario, M.G., Marshall, J., Herkert, A., Binoy, B., Windham, H., & Orozco, E. (2020). Lower Limb Neuromuscular Modification and Standing Postural Control Alteration in Apparent Asymptomatic People Living with HIV. J Rehab Pract Res 1(1):102.doi: https://doi.org/10.33790/jrpr1100102.
Pollock, A. S., Durward, B. R., Rowe, P. J., & Paul, J. P. (2000). What is balance? Clinical Rehabilitation, 14(4), 402-6. doi:http://dx.doi.org.ezp.twu.edu/10.1191/0269215500cr342oa.View
Berner, K., Morris, L., Baumeister, J., Low, Q. (2017). Objective impairments of gait and balance in adults living with HIV-1 infection: a systematic review and meta-analysis of observational studies. BMC Musculoskeletal Disorders.18(325):2-26. http:// doi.org/10.1186/s12891-017-1682-2View
Rosario, M. (2020d). Gastrocnemius and tibialis anterior neuromuscular modification recruitment during postural standing in people living with HIV. AIDS Review, 19(4), 260– 266. https://doi.org/10.5114/hivar.2020.101633View
Trenkwalder, C., Straube, A., Paulus, W., Krafczyk, S., Schielke, E., & Einhäupl, K. M. (1992). Postural imbalance: an early sign in HIV-1 infected patients. European archives of psychiatry and clinical neuroscience, 241(5), 267–272. https://doi.org/10.1007/ BF02195975View
Pimenta, B. J. F., Moura, F. A., Santos, C. R., Martins, R. M., Cardoso, J. R., and Marques, I. (2019). Assessment of postural sway in people living with HIV/AIDS. ABCS Health Sciences, 44(3), 180-187. Doi: https://dx.doi.org/10.7322/abcshs. v44i3.1196View
Bauer, L. O., Ceballos, N. A., Shanley, J. D., & Wolfson, L. I. (2005). Sensorimotor dysfunction in HIV/AIDS: effects of antiretroviral treatment and comorbid psychiatric disorders. AIDS (London, England), 19(5), 495–502. https://doi. org/10.1097/01.aids.0000162338.66180. 0b. View
Bauer, L. O., Wu, Z., & Wolfson, L. I. (2011). An obese body mass increases the adverse effects of HIV/AIDS on balance and gait. Physical therapy, 91(7), 1063-1071.View
Qun Liu, Zhi-Yi Xu, Xiao-Li Wang, Xiao-Mei Huang, Wen-Lin Zheng, Mei-Jun Li, Fan Xiao, Pei-Wen Ouyang, Xiao-Hua Yang, Yu-Hong Cui, Hong-Wei Pan; (2021). Changes in Conjunctival Microbiota Associated With HIV Infection and Antiretroviral Therapy. Invest. Ophthalmol. Vis. Sci. 62(12):1. doi: https://doi.org/10.1167/iovs.62.12.1.View
Teggi, R., Giordano, L., Pistorio, V., & Bussi, M. (2006). Vestibular function in HIV patients:preliminary report. Acta otorhinolaryngologica Italica : organo ufficiale della Societa italiana di otorinolaringologia e chirurgia cervico-facciale, 26(3), 140–146.View
Rosario, M. G., Jamison, L., & Gines, G. (2020c). The Role of HIV Antiretroviral Medication on Motor-Cognitive and Neurological Alterations in Hispanic People Living with HIV. J Pub Health Issue Pract 4(1):160. doi: https://doi.org/10.33790/ jphip1100160.
Bjerk, M., Brovold, T., Skelton, D. A., & Bergland, A. (2017). A falls prevention programme to improve quality of life, physical function and falls efficacy in older people receiving home help services: study protocol for a randomised controlled trial. BMC health services research, 17(1), 559. https://doi.org/10.1186/ s12913-017-2516-5 View
Erlandson, K. M., Allshouse, A. A., Jankowski, C. M., Duong, S., MaWhinney, S., Kohrt, W. M., & Campbell, T. B. (2012). Risk factors for falls in HIV-infected persons. Journal of acquired immune deficiency syndromes (1999), 61(4), 484–489. https://doi.org/10.1097/QAI.0b013e3182716e38View
Schrack, J. A., Althoff, K. N., Jacobson, L. P., Erlandson, K. M., Jamieson, B. D., Koletar, S. L., & Margolick, J. B. (2015). Accelerated longitudinal gait speed decline in HIV-infected older men. Journal of acquired immune deficiency syndromes (1999), 70(4), 370.View
Hinkin, C. H., Castellon, S. A., & Hardy, D. J. (2000). Dual task performance in HIV-1 infection. Journal of clinical and experimental neuropsychology, 22(1), 16–24.https://doi. org/10.1076/1380-3395(200002)22:1;1-8;FT016View
Hausdorff, J. M. (2007). Gait dynamics, fractals and falls: Finding meaning in the stride-to-stride fluctuations of human walking. Human Movement Science, 26(4), 555-589. doi:10.1016/j.humov.2007.05.003View
Rosario, M. G., Bowman, C. ., Vega-Calderon, I. ., & Orozco, E. . (2021c). Dual Cognitive Tasks Provoke Temporo-Spatial Gait and Anticipatory Postural Adjustments in Healthy Young Adults. Journal of Sports Research, 8(1), 26–34. https://doi. org/10.18488/journal.90.2021.81.26.34View
Fritz, S., & Lusardi, M. (2009). White paper: "walking speed: the sixth vital sign". Journal of geriatric physical therapy (2001), 32(2), 46–49.View
Takayanagi, N., Sudo, M., Yamashiro, Y., Lee, S., Kobayashi, Y., Niki, Y., & Shimada, H. (2019). Relationship between daily and in-laboratory gait speed among healthy community-dwelling older adults. Scientific Reports, 9(1). https://doi.org/10.1038/ s41598-019-39695-0View
Rosario, M.G., Marshall, J., Herkert, A., Binoy, B., Windham, H., & Orozco, E. (2020b). Lower Limb Neuromuscular Modification and Standing Postural Control Alteration in Apparent Asymptomatic People Living with HIV. J Rehab Pract Res 1(1):102. https://doi.org/10.33790/jrpr1100102.
Richert, Lauraa; Dehail, Patrickb,c; Mercié, Patricka,c; Dauchy, Frédéric-Antoinec; Bruyand, Mathiasa; Greib, Carinec; Dabis, Françoisa,c; Bonnet, Fabricea,c; Chêne, Genevièvea,c for the Groupe d'Epidémiologie Clinique du SIDA en Aquitaine (GECSA) High frequency of poor locomotor performance in HIV-infected patients, AIDS: March 27, 2011 - Volume 25 - Issue 6 - p 797-805 doi: 10.1097/QAD.0b013e3283455dffView
Richert, Lauraa,b,c; Brault, Mathildeb,c; Mercié, Patricka,b,c; Dauchy, Frédéric-Antoineb,c; Bruyand, Mathiasa,c; Greib, Carinec; Dabis, Françoisa,b,c; Bonnet, Fabricea,b,c; Chêne, Genevièvea,b,c; Dehail, Patrickb,c,d for the Groupe d’Epidémiologie Clinique du SIDA en Aquitaine (GECSA) Decline in locomotor functions over time in HIV-infected patients, AIDS: June 19, 2014, - Volume 28 - Issue 10 - p 1441- 1449. doi: 10.1097/QAD.0000000000000246View
Rosario, M.G., Gines, G., Jamison, L. (2020e). Lifestyle, Physical and Cardiovascular Components Associated with Immune Profile in Hispanic-Latino People Living with HIV. J Ment Health Soc Behav 2(1):121. https://doi.org/10.33790/ jmhsb1100121
Martín G. Rosario, Leah Jamison, Gabriel Gines. (2021b). Peripheral Neuropathy Impacts Gait Motor Components in Hispanic-Latinx living with HIV International Journal of Sports Medicine and Rehabilitation. International Journal of Sports Medicine and Rehabilitation. 2021; 4:20View
Lesinski, M., Hortobágyi, T., Muehlbauer, T. et al. (2015). Effects of Balance Training on Balance Performance in Healthy Older Adults: A Systematic Review and Meta-analysis. Sports Med 45, 1721–1738. https://doi-org.ezp.twu.edu/10.1007/ s40279-015-0375-yView
Gschwind, Y.J., Kressig, R.W., Lacroix, A. et al. (2013). A best practice fall prevention exercise program to improve balance, strength / power, and psychosocial health in older adults: study protocol for a randomized controlled trial. BMC Geriatr 13, 105. https://doi.org/10.1186/1471-2318-13-105 View
Quiles, N.N., Rosario, M., & Ortiz, A. (2019). Balance as an assessment of health-related quality of life in people living with HIV. Journal of Human Sport and Exercise, 14(2), 492-499. doi:https://doi.org/10.14198/jhse.2019.142.20View
Power, C., Selnes, O.A., Grim, J.A., & McArthur, J.C. (1995). HIV Dementia Scale: a rapid screening test. J Acquir Immune Defic Syndr Hum Retrovirol.1;8(3):273-8. doi: 10.1097/00042560-199503010-00008. PMID: 7859139.View
Derry, H.M., Johnston, C.D., Burchett, C.O., Siegler, E.L., & Glesby, M.J. (2020). Gait Speed Is Associated with Cognitive Function among Older Adults with HIV. J Aging Health.32(10):1510-1515. doi: 10.1177/0898264320943330. Epub 2020 Jul 22. PMID: 32697615; PMCID: PMC7768797.View
Seo, M., Won, C.W., Kim, S., Yoo, J.H., Kim, Y.H., Kim, B.S. (2020). The Association of Gait Speed and Frontal Lobe among Various Cognitive Domains: The Korean Frailty and Aging Cohort Study (KFACS). J Nutr Health Aging. 24(1):91-97. doi: 10.1007/s12603-019-1276-9. PMID: 31886814.View
Fazeli, P. L., Woods, S. P., Heaton, R. K., Umlauf, A., Gouaux, B., Rosario, D., Moore, R. C., Grant, I., Moore, D. J., & HNRP Group (2014). An active lifestyle is associated with better neurocognitive functioning in adults living with HIV infection. Journal of neurovirology, 20(3), 233–242. https://doi. org/10.1007/s13365-014-0240-zView
Pappas, D., Roland, J., Lim, J., Lai, A., & Hillman, D. (1995). Ultrastructural findings in the vestibular end-organs of AIDS cases. American Journal of Otology.16:140–5. [PubMed] [Google Scholar]
Shangase, K.K. Vestibular Function in a group of adults with HIV/AIDs on HARRT. 11AD. https://www.ncbi.nlm.nih.gov/ pmc/articles/PMC5733256/View
Kinnealey, M., Riuli, V., & Smith, S. (2015). Case Study of an Adult With Sensory Modulation Disorder. Sensory Integration. 38(1).View
Tee, L.H., Chee, N.W.C. (2005). Vestibular Rehabilitation Therapy for the Dizzy Patient. https://annals.edu.sg/ pdf/34VolNo4200505/V34N4p289.pdfView
Bayot, M., Dujardin, K., Tard, C., Defebvre, L., Bonnet, C., Allart, E., & Delval, A. (2018). The interaction between cognition and motor control: A theoretical framework for dual-task interference effects on posture, gait initiation, gait and turning. Neurophysiologie Clinique, 48(6), 361-375.View
Shin, S., An, D., & Yoo, W. (2014). Effects of balance recovery after square and semicircular turns on gait velocity and center of mass acceleration in older adults with differing visual acuity. European Geriatric Medicine, 6(2), 103-108.View
Nieuwboer, A., Baker, K., Willems, A., Jones, D., Spildooren, J., Lim-de Vries, L., . . . Rochester, L. (2009). The short-term effects of different cueing modalities on turn speed in people with Parkinson's disease. Neurorehabilitation and Neural Repair, 23(8), 831-836.View
Alford, K., Banerjee, S., & Nixon, E., et al. (2019). Assessment and Management of HIV-Associated Cognitive Impairment: Experience from a Multidisciplinary Memory Service for People Living with HIV. Brain Sci. 9(2):37. Published 2019 Feb 8. doi:10.3390/brainsci9020037View
Yang, X. J., Hill, K., Moore, K., Williams, S., Dowson, L., Borschmann, K., Simpson, J. A., & Dharmage, S. C. (2012). Effectiveness of a targeted exercise intervention in reversing older people's mild balance dysfunction: a randomized controlled trial. Physical therapy, 92(1), 24–37. https://doi. org/10.2522/ptj.20100289View