Journal of Public Health Issues and Practices Volume 6 (2022), Article ID: JPHIP-204
https://doi.org/10.33790/jphip1100204Review Article
Tocilizumab as a Treatment for Cytokine Storm in COVID-19 Patients: A systematic review
Muhammad Huzaimi Haron1, Mohamad Rodi Isa2*, Hanisa Syahirah Mohd Rashid3, Nur Amanina Adam3, Nur Aliah Awang3, Muhammad Hairul Faez Halip3
1Department of Pharmacology, Faculty of Medicine, Universiti Teknologi MARA, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia.
2Department of Public Health Medicine, Faculty of Medicine, Universiti Teknologi MARA, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia.
3Faculty of Medicine, Universiti Teknologi MARA, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia.
Corresponding Author Details: Mohamad Rodi Isa, MBBS, DAP&E, MPH, DrPH, Department of Public Health Medicine, Faculty of Medicine, Universiti Teknologi MARA, Jalan Hospital, 47000 Sungai Buloh, Selangor, Malaysia.
Received date: 11th April, 2022
Accepted date: 06th May, 2022
Published date: 09th May, 2022
Citation: Haron, M.H., Isa, M.R., Mohd Rashid, H.S., Adam, N.A., Awang, N.A., & Faez Halip, M.H., (2022). Tocilizumab as a Treatment for Cytokine Storm in COVID-19 Patients: A systematic review. J Pub Health Issue Pract 6(1): 204.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Tocilizumab is a competitive interleukin-6 inhibitor agent that has been proposed to combat the COVID-19-related hyperinflammatory state, known as a cytokine storm. This systematic review was conducted to study the treatment of cytokine storm by Tocilizumab in COVID-19 patients. The search strategy (“COVID-19” OR “COVID19” OR SARS-CoV-2”) AND “tocilizumab” AND “cytokine storm” AND “inflammatory markers” AND (“ICU stay duration” OR “intensive care unit stay duration”) AND “mechanical ventilation requirement” AND (mortality OR death) were manually searched through Web of Science, Scopus, and PubMed databases spanned from March 2020 to November 2021. The inclusion criteria were: research articles, human study, clinical trial, and articles in English. The exclusion criteria were: review articles, case reports, early access, editorial materials, letters, short survey, in vivo or in vitro studies. Five articles were included in the analysis. There were four countries had conducted the studies (Italy, China, USA and Netherland) with different study designs (observational (80%) and randomized controlled trials (20%)) involving 649 patients (48% received TCZ) among moderate to severe COVID-19 patients. There were variabilities in the TCZ dosage given with some combination with other medication (methylprednisolone, azithromycin, hydroxychloroquine, lopinavir and ritonavir). TCZ reduce death cases significantly. It improves respiratory function, reduces the incidence of respiratory syndrome and less-invasive mechanical ventilation usage. The level of inflammatory markers such as C-reactive protein, ferritin and lactate dehydrogenase were significantly higher in the TCZ group. Tocilizumab may increase survival and favourable clinical course, improved hypoxia, accelerate respiratory recovery, lower hospital mortality, reduce the likelihood of invasive mechanical ventilation, improve clinical symptoms, represses the deterioration of patients (prolonging survival) and improve inflammation and immune cell function.
Keywords: Tocilizumab, Cytokine storm, COVID-19 patients
Introduction
Coronavirus disease 2019 (COVID-19) is a contagious illness caused by a novel coronavirus which is now known as severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) [1]. The virus spreads primarily by droplets of saliva or discharge from the nose when an infected person coughs or sneezes. The symptoms exhibited varies among COVID-19 patients with the most common symptoms being fever, dry cough, and tiredness. Additionally, some patients may show less typical symptoms such as aches, sore throat, diarrhoea, conjunctivitis, headache, loss of taste or smell, and rashes on the skin [2].
In the wake of COVID-19, the term ‘cytokine storm’ becomes more familiar than ever before. It is a common term among members of the scientific community, but now the general public is also aware of it [3]. It is defined as a severe immune reaction in which the body releases too many cytokines into the blood too quickly. Cytokines play an important role in normal immune responses, but they can be harmful when it released in a large amount simultaneously in the body [4]. A cytokine storm can occur as a result of an infection, autoimmune condition, or other diseases. It may also occur after treatment with some types of immunotherapies. In the development of the cytokine storm, there are many main components involved such as Interferons, interleukins, chemokines, Colony-stimulating factors and TNF-alpha [5]. In COVID-19, approximately 2% of mortality is due to cytokine storms throughout the world [1].
Interleukin-6 (IL-6) plays the biggest role in the pathogenesis of cytokine storms and the progression of COVID-19 related cytokine storms [6]. It binds to two forms of IL-6 receptor (IL-6R) i.e., membrane-bound interleukin-6 receptor (mIL-6R) and soluble interleukin-6 receptor (SIL-6R) forming a receptor-ligand complex. The complex will then bind to gp130 on the cell membrane of the alveolar epithelial tissue to proceed with signal transduction resulting in a pro-inflammatory response [7].
Ferritin is an acute protein that escalates in response to the broad spectrum of inflammatory states including infections, malignancy, iron overload, and liver and kidney disease. Hyperferritinaemia can be used as a screening tool for the early diagnosis of cytokine storms (Melo et al, 2021). A significantly elevated ferritin level in the blood of in-patients is often associated with haemophagocytic lymphohistiocytosis (HLH) syndrome but it cannot be considered as a specific marker [8]. HLH is a severe systemic inflammatory syndrome due to the strong activation of the immune system that can be fatal [9]. The SARS-CoV-2 infection can lead to features of cytokine storms that are reminiscent of secondary HLH [10].
Based on several studies, tocilizumab (TCZ), a humanized monoclonal antibody is needed in invasive mechanical ventilation and ICU intervention. It has been suggested as a favourable agent for moderate to severe COVID-19 cases. It was designed to inhibit the JAK-STAT or MAPK/NF-κB-IL-6 signaling pathway and finally stops the cytokine storms syndrome [11]. Therefore, it can reduce the mortality rate and length of hospital stay. This systematic review is conducted to study the treatment of cytokine storms by TCZ in COVID-19. This review was conducted to determine the impact of the use of TCZ on inflammatory marker level, ICU stay duration, mechanical ventilation requirement and mortality rate among patients with COVID-19 related cytokine storm.
Material and Methods
A systematic review was performed and reported according to the reporting standards documented in the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement. This review was registered with the PROSPERO database (Registration number: CRD42022303727).
Comprehensive Literature Review
The searching procedure was conducted using bibliographic databases and other evidence sources which can address the review question. The procedure of comprehensive literature search involved looking for the eligible articles, searching strategy for identification of studies, study selection, and data extraction.
Search strategy
A comprehensive search of the literature was undertaken using three biomedical electronic databases which were Web of Science (WoS), Scopus, and PubMed complete Library to perform our research. The search aimed to identify relevant articles published in peer-reviewed journals written in English, with the assumption that most of the important findings will be reported in English regardless of country of origin. The process of searching strategy for identification of studies included all published studies.
The formulated question was performed by identifying the type of evidence needed to answer the question. The strategy by using Domain, Determinant, and Outcome (DDO) format was used in the study to obtain relevant answers and to identify the question and concentrate the mind. The Boolean search was performed on each database as follows:
(“COVID-19” OR “COVID19” OR SARS-CoV-2”) AND “tocilizumab” AND “cytokine storm” AND “inflammatory markers” AND (“ICU stay duration” OR “intensive care unit stay duration”) AND “mechanical ventilation requirement” AND (mortality OR death).
Eligibility Criteria
This research examined the TCZ as the treatment for Cytokine Storm in COVID-19 patients of published articles. The electronic databases (“COVID-19” OR “COVID19” OR SARS-CoV-2”) AND “tocilizumab” AND “cytokine storm” AND “inflammatory markers” AND (“ICU stay duration” OR “intensive care unit stay duration”) AND “mechanical ventilation requirement” AND (mortality OR death). The studies that did not meet the specific inclusion criteria for this study were eliminated. The decision to include all employees was made so that all available studies could be included in this study. The inclusion criteria in this study are research article, human study, clinical trial stud, and article in English. The exclusion criteria are review articles, systematic review, case report, early access, editorial materials, letters, short survey, in vivo or in vitro study, and other languages.
Published Articles
The published articles refer to any article that has been published in any published journal articles. The first step was to locate the published articles for this research using a computer-based information search. The established databases in this study are Web of Science (WoS), Scopus, and PubMed. The references of the chosen studies were analysed manually by all researchers to obtain additional research articles for this review.
Screening Titles and Abstract
All citations were identified by screening titles and abstracts of the published articles studies. The relevant articles were downloaded into the Endnote Software. The citations were organized accordingly. Besides, the duplication of the studies was identified and deleted. The coding studies guide was used to screen for relevant articles and theses. Theoretically, study screening looking for suitable titles and abstracts was conducted by at least two independent researchers. When all researchers have agreed on the suitable title and abstract, the articles were considered for full-text retrieval. The articles which did not fulfil the criteria were excluded from the study.
Obtaining Full text Published Articles Studies
All published articles were searched. Full-text articles were obtained and downloaded from the established resources (Web of Science (WoS), Scopus, and PubMed) with a search term. Articles searched in this study were only obtained in a free article. The articles without full text were excluded from the study.
Selecting Suitable Full text Published Articles
The process of selecting suitable full text published articles was done and selected by the corresponding author who are experts in methodology and topic under review. An agreement of inclusion and exclusion criteria was made before starting the reviews process.
After searches were performed, articles were then organized into Endnote Software. Duplication of the articles was identified and removed by Zotero software. This was performed by one reviewer, via the “Find and Remove Duplicate References” function at first, followed by manual screening, as some of the same articles were entered slightly differently into different databases. After duplication of articles was removed, articles were assessed for eligibility independently by two reviewers in two stages. In the first stage, the title and abstract of search results were screened and assessed for relevance. In the second stage, the full text of potentially relevant publications was retrieved and reviewed for inclusion. Both stages of the study selection were performed independently by two reviewers and cross-validated to assess for disagreements. The list of studies included and excluded based on the inclusion and exclusion criteria described earlier was cross-validated to assess for disagreements. If there was disagreement between both reviewers a third reviewer was assigned. Only full-text articles were included in the review to enable quality assessments.
Critical Appraisal
Critical appraisal was performed by two researchers to assess study quality and most methods encompass issues such as appropriateness of study design to the research objective. The articles with poor-quality studies were excluded and discussed in detail. The critical appraisal of articles was performed using the Consolidated Standards of Reporting Trials (CONSORT) checklist for randomized controlled trials and Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) for epidemiological observational study as guidelines to assess to quality of the articles that have been selected. The agreement between the two independent research was assessed using Kappa statistics. The value of kappa with more than 0.80 is considered a good agreement.
Data extraction
Data extractions were performed by all two independent researchers to establish inter-rater reliability and to avoid data entry errors. Study context factors of published articles include the information describing the study and its subjects. Reported findings of the remaining studies were extracted onto a data extraction form. Lists of included studies were created stratified by study name. study design, patient type, dose, sample size and result independently.
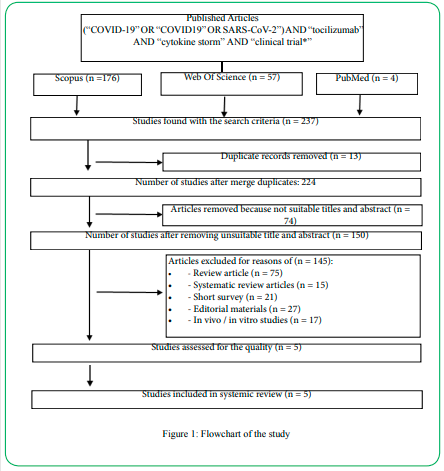
Flowchart of the study
The process of article searches and selection that was performed in this study is shown in Figure 1:
Data Management
All relevant articles were manually coded in Google Docs as described in evidence tables. The electronic Google Docs were utilised to import the data into word form for data analysis.
Statistical analysis
Data were analysed using descriptive statistics. The numerical outcome was be analysed using mean and standard deviation. Categorical outcomes were analysed using frequency and percentage.
Results
A total of 237 records were identified through searches in three databases (Web of Science (WoS), Scopus, and PubMed). After excluding the duplicates, titles and abstracts were screened, 150 articles were selected for the eligibility of the study. However, 140 articles were removed due to various reasons such: review article (n = 75), systematic review articles (n = 15), short survey (n = 21), editorial materials (n = 27) and In vivo / in vitro studies (n = 17). 5 articled were selected for the quality assessment and all articles were included in the study. An overview of these selected studies in research characteristics created in the PRISMA chart is shown in Figure 1.
A total of five articles were reviewed in this study and summarized in Table 1. The studies were conducted in Italy [12], China [13,14], the Netherlands [15], and the United States of America (USA) [16]. All of the studies involved patients infected with COVID-19 ranging from severe COVID-19 patients [16], moderate to the severe stage (cytokine storm syndrome) associated with either pneumonia [12], moderate (bilateral pulmonary lesions) or severe COVID-19 [13], Severe COVID-19 patients with extensive lung lesions [14] and Severe COVID-19 associated cytokine storm syndrome [15].
Two of the studies (40%) were conducted at a single centre and three of the studies (60%) were conducted in a multi-centre study. One of the studies (20%) was conducted via a randomized controlled trial [13] and the other four studies (80%) were conducted via an observational study i.e., matched case-control study [12], retrospective cohort study [14], matched cohort study [16] and retrospective case-control study [15]. The total number of COVID-19 patients involved was 649, where 312 (48%) patients received TCZ and 338 (52%) patients was in the control group.
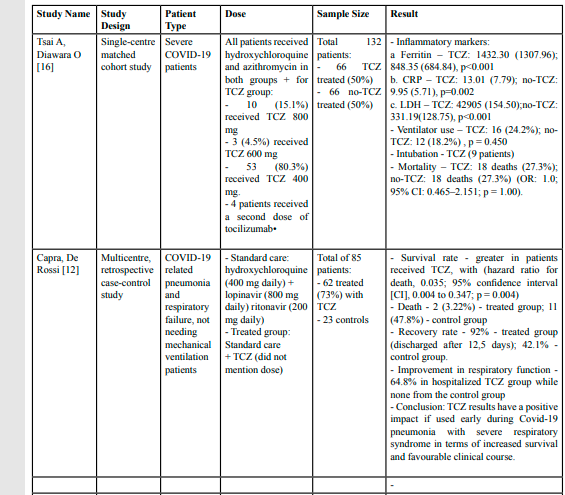
There were variabilities of the TCZ dosage given to the treated patients. A study by Wang D, Fu B [13] gave 400mg TCZ diluted in 100 ml of 0.9% saline for the first dose and the second dose was only given if the patients still had febrile for 24 hours. Tian J, Zhang M [14] intervened by giving intravenous (IV) 400mg TCZ and infusing for one hour and an additional dose was given if the fever continue within 12 hours. Ramiro S, Mostard RLM [15] intervened by giving IV 8mg/kg body weight with a combination of 250 mg IV 250 methylprednisolone (MP) on day 1 and 80mg MP on day -2 to day-5. A study by Tsai A, Diawara O [16] intervened by giving hydroxychloroquine and azithromycin and different mg of TCZ i.e. 10 (15.1%) patients received TCZ 800 mg, 3 (4.5%) patients received TCZ 600 mg, 53 (80.3%) patients received TCZ 400 mg and four patients received a second dose of TCZ. However, a study by Capra, De Rossi [12] did not mention the dosage of TCZ but it was given in a combination of hydroxychloroquine (400 mg daily), lopinavir (800 mg daily) and ritonavir (200 mg daily).
Capra, De Rossi [12] found a greater survival rate in patients who received TCZ (HR: 0.035; 95%CI: 0.004, 0.347) and a 92% recovery rate with discharged after 12.5 days compared to just 42.1% in the control group. Wang D, Fu B [13] found 94.12% cure rate in TCZ group compared to 87.10% in the control group (p=0.4133, 95%CI: 7.19%, 21.23%). There was no significant difference in the ICU stay between TCZ and the control group (10 days versus 12 days, p=0.270). However, there was a significant difference in hospital stays where TCZ groups had longer stays compared to the non-TCZ group (40 days versus 26.5 days, p=0.001)[14].
For death, Capra, De Rossi [12] found 3.22% in TCZ group compared to 47.8% in control group. Whereas, Tian J, Zhang M [14] found 21.54% death in TCZ group and 32.31% in non-TCZ group (p = 0.012); hazard ratio = 0.47; 95% CI: 0.25, 0.90; p = 0.023) for the in-hospital death. Ramiro S, Mostard RLM [15] found 65% lower hospital mortality in TCZ group than control groups (HR: 0.35; 95% CI: 0.19, 0.65). However, Tsai A, Diawara O [16] did found any significant different in the death case between both (OR: 1.0; 95% CI: 0.465–2.151; p=1.000).
Capra, De Rossi [12] found an improvement in respiratory function in 64.8% in the TCZ group but none in the control group and Ramiro S, Mostard RLM [15] found a 79% higher likelihood of improvement in respiratory status compared to the control group (HR: 1.79; 95%CI: 1.20, 2.67), 7 days earlier and Tian J, Zhang M [14] found less incidence of respiratory distress syndrome in TCZ group (36.92%) compared to the control group (70.77%) (adj OR: 0.23 (95%CI: 0.11, 0.45; p < 0.001).
Ramiro S, Mostard RLM [15] found 71% less-invasive mechanical ventilation usage in the TCZ group (HR: 0.29; 95% Cl 0.14 to 0.65). There was also a significant difference in the incidence of mechanical ventilation use were less in the TCZ group (1.3% vs 5.4% (p=0.0003). However, Ramiro S, Mostard RLM [15] reported that once the patients were mechanically ventilated, the duration of mechanical ventilation was not significantly different. Tsai A, Diawara O [16] found no significant difference in ventilator use (24.2% versus 18.2%, p=0.450) although nine patients in the TCZ group need intubation. Wang D, Fu B [13] found an improvement in the hypoxia from day 4 to day 12 (p=0.0359) in severe COVID-19 patients. In moderate COVID-19 patients, only 8.33% of hypoxia was reported compared to 66.67% in control group (p=0.0217; 95%CI: -99.17%, -17.50). However, Wang D, Fu B [13] found 58.82% mild temporary adverse effects in the TCZ group than only 12.90% in the control group.
There were some inflammatory markers monitored in the treatment for cytokine storm in COVID-19 patients. The level of C-reactive protein (CRP) was significantly higher in the TCZ group than in the non-TCZ group [16]. However, a study by Tian J, Zhang M [14] did not find the same result. The ferritin level and lactate dehydrogenase (LDH) were significantly higher in the TCZ group than in the non-TCZ group [16]. However, the IL-10 level was significantly higher in the non-TCZ group than TCZ group [14].
In the overall conclusion, most of the findings show a positive impact of TCZ on the COVID-19 patients such as increased survival and favourable clinical course [12], improved hypoxia without unacceptable side effects and influences on the time virus load becomes negative [13] and may accelerate respiratory recovery, lower hospital mortality and reduce the likelihood of invasive mechanical ventilation [15]. TCZ also can improve clinical symptoms and represses the deterioration of patients (prolonging survival), lower the risk of an in-hospital proportion of death and improve inflammation and immune cell function [14]. However, only a study by Tsai A, Diawara O [16] did not support the use of TCZ for the management of cytokine storms in patients with COVID-19.
Discussion
In this systematic review, we summarised and analysed the effect of TCZ as a treatment for COVID-19 patients related to cytokine storms. There were many studies found and have been conducted in five countries in various types of moderate and severe COVID-19. The activation of various cytokines is reflected by clinical manifestations in COVID-19 infection. Pyrexia, malaise and chills may convey an increase in the interferon levels while acute symptoms such as vascular damage and lung contusions along with general symptoms may indicate high TNF levels [17]. A high concentration of interleukins may lead to cytokine storms.
Randomized controlled trials are considered to be the “gold-standard” method to estimate the treatment effect [18]. However, in the emergency of COVID-19 worldwide, RCT was not possible to conduct. Many researchers conducted an observational study to determine the impact of TCZ with one group as a comparison group [12,14-16]. However, ethical approval and informed consent must be obtained before conducting the study. The ethical review protects participants and also the researcher. By obtaining ethical approval, the researcher is demonstrating that they have adhered to the ethical standard of a research study [19].
TCZ is a key cytokine leading to an inflammatory storm act by inhibiting the IL-6 receptor which may result in increased alveolar-capillary blood glass exchange dysfunction [20]. There are about more than 30 drugs have been introduced to treat COVID-19 including TCZ and many research investigate further the efficacy of TCZ for different patients [21]. A review by Samaee, Mohsenzadegan [21] of 15 studies found many variabilities of dosages prescribed to the patients for their treatment which is also the same finding in this review. The US Food and Drug Administration (FDA) recommended dosage of TCZ is a single 60-minute intravenous infusion for patients less than 30kg weight is 12mg/kg and to patients at or above 30kg weight is 8mg/kg with maximum dosage is 800mg per infusion for COVID-19 patients [22].
Untreated cytokine storms can progress from respiratory failure to cardiovascular collapse, multiorgan dysfunction, and death [23]. In this review, the survival rate was greater in the treated group with a 92% recovery rate and discharged after 12.5 days [12]. However, Wang D, Fu B [13] did not find any significant cure rate although the percentage in the TCZ group was higher than the non-TCZ group and there was a significant difference in hospital stays where TCZ groups had longer stays compared to the non-TCZ group (40 days versus 26.5 days, p=0.001) [14]. Gokhale Yojana, Mehta Rakshita [23] concluded that the warranted improvement was due to a hyperinflammatory state.
Mortality in COVID-19 patients has been linked to the presence of the so-called “cytokine storm” induced by the virus [24]. The majority of the studies reviewed found that TCZ can reduce death, lower hospital mortality or in-hospital death [12,14,15] except a study by Tsai A, Diawara O [16] did not found any significant difference in death. The mortality is due to an excessive production of pro-inflammatory cytokines which leads to ARDS aggravation and widespread tissue damage [24,25]. In addition to that, fatal pneumonia can also be a result of mortality in COVID-19 patients after the SARS-CoV-2 completes replication in the lower respiratory tract [25].
Invasive mechanical ventilation is a lifesaving tool commonly used in the care of hospitalized patients [26]. Adults with COVID-19 who develop acute respiratory distress syndrome (ARDS) are managed by intubation and invasive mechanical ventilation as a conventional oxygen therapy may be insufficient to fulfil their oxygen needs [27]. ARDS is frequently characterized by high levels of plasma IL-6, which will stimulate lung epithelial cells and inflammatory cells response thus leading to pulmonary damage COVID-19 patients with cytokine storm syndrome treated with TCZ had 71% less used and incidence of invasive mechanical ventilation [15]. Several studies have shown that inhibition of IL-6 with TCZ may help to improve the respiratory status and survival of patients with COVID-19 requiring mechanical ventilation [28]. However, Tsai A, Diawara O [16] found no significant difference in ventilator use in treated and without treated TCZ group and Ramora et al. [15] found no difference found no difference in the duration of using mechanical ventilation once the patients were mechanically ventilated. Although advanced aged COVID19 patients are presented with greater pneumonia severity scores, need for oxygen therapy, lymphopenia and need of mechanical ventilation it was not associated with age [29].
Prolong intensive care unit (ICU) admission led COVID-19 patients to worse outcomes. Studies from early in the pandemic have found statistically one in five infected individuals are hospitalised and one in ten may get admitted to an ICU, with most of these critically ill patients experiencing ARDS and requiring mechanical ventilation [30]. However, Tian J, Zhang M [14] did not find any significant difference in the ICU stay between TCZ and control group but TCZ groups had longer stays compared to the non-TCZ group.
Hypoxia and respiratory failure were considered as important causes of exacerbation and even death of COVID-19 [31]. The “Cytokine storm” leads to immune dysregulation and tissue damage in the lungs that cause hypoxia and respiratory failure [32]. With the treatment by TCZ, it was found an improvement in the hypoxia in day-4 to day-12 in severe and moderate COVID-19 patients and it revealed that an increased IL-6played an important role in the “cytokine storm” [13]. Although Wang D, Fu B [13] found TCZ group had more adverse effects including leukopenia, neutropenia and abnormal hepatic function, all of these side effects remitted spontaneously and no serious adverse events occurred.
Mostly, serum CRP will increase in bacteria compared to viral infection but a current study on COVID-19 patients revealed a significant rise of serum CRP levels in severe cases than in non-severe patients. It can be suggested that CRP can be the biomarker for assessing disease progression and mortality in COVID-19 patients [33]. Besides CRP, ferritin is another biomarker for inflammation in COVID-19 patients. Ferritin has been characterized as an acute phase reactant and mediator of immune dysregulation in severe COVID-19 [34]. The remarkable increase of serum ferritin level routinely indicates liver damage. However, a study by Tsai A, Diawara O [16] found the level of ferritin raise than the normal level in COVID-19 patients which specifies more risk of liver injury and severe illness. Therefore, analysis of ferritin at an early stage can recognise liver damage, disastrous disease, and prognosis of COVID-19 patients [35]. Cytokine can induce ferritin expression and ferritin can induce expression of pro-inflammatory cytokine as well. Patients with higher ferritin levels have been associated with more severe lung involvement in COVID-19 due to the development of lung damage resulting from activation of the inflammatory response induced by high ferritin levels [34].
There are several limitations in conducting this systematic review. The summary provided is only reliable depending on the methods used to estimate the effect in each of the primary studies. The systematic review also relied on the relatively limited number of databases for the identification of potentially eligible studies therefore there may not be enough research in the literature to analyse. The systematic review can quickly become outdated because COVID-19 is a newly emerged disease in which many ongoing types of research are conducted. The quality of the systematic review depends on what has been published in the literature
Conclusion
It can be concluded that TCZ was associated with cytokine storm syndrome. It may reduce mortality rate, mechanical ventilation requirement, ICU stay duration, and IL-6 level if it is administered before entering the moderate to severe inflammatory state as a treatment for COVID-19.
Competing of interest:
All authors declare that there is no conflict of interest.
List of abbreviation
ARDS: Acute Respiratory Distress Syndrome; CONSORT: Consolidated Standards of Reporting Trials; COVID-19: Coronavirus disease 2019; CRP: C-reactive protein; DDO: Domain, Determinant, and Outcome; FDA: The US Food and Drug Administration; ICU: Intensive care unit; HLH: Haemophagocytic lymphohistiocytosis; IL=6: Interleukin-6; IL-6R: IL-6 receptor; IV: Intravenous: LDH: Lactate dehydrogenase; OR: Odds ratio; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; STROBE: Randomized controlled trials and Strengthening the Reporting of Observational Studies in Epidemiology; TCZ: Tocilizumab; WoS: Web of Science;
Acknowledgement
This abstract has been presented in the International Conference on Post COVID Healthcare, Medical Research and Education 2022 and Creation de UiTM, International Mega Innovation Carnival 2022.
Funding
This is self-funded research and did not receive any funding.
References
Cron, R.Q, Caricchio, R., and W.W. Chatham, (2021). Calming the cytokine storm in COVID-19. Nature Medicine, 27(10): p. 1674–1675.View
World Health Organization Coronavirus. 2021.View
Tisoncik JR, et al., (2012). Into the Eye of the Cytokine Storm. Microbiology and Molecular Biology Reviews, 76(1): p. 16–32.View
National Cancer Institute (2021). Cytokine strom. View
Coperchinia, F., et al., (2020). The cytokine storm in COVID-19: An overview of the involvement of the receptor system. Cytokine and Growth Factor Reviews, 53: p. 25-32.View
Giannakodimos, I., et al., (2021). The Role of Interleukin-6 in the Pathogenesis, Prognosis and Treatment of Severe COVID-19. Curr Med Chem, 28(26): p. 5328-5338.View
Zhang SY, et al., (2020). Rational Use of Tocilizumab in the Treatment of Novel Coronavirus Pneumonia. Clinical Drug Investigation, 40(6): p. 511–518.View
Melo AKG, et al., (2021). Biomarkers of cytokine storm as red flags for severe and fatal COVID-19 cases: A living systematic review and meta-analysis. PLOS ONE, 16(6): p. e0253894.View
Immune Deficiency Foundation (2017). Hemophagocytic Lymphohistiocytosis (HLH).View
Hakim, N.N., et al., (2021). Secondary hemophagocytic lymphohistiocytosis versus cytokine release syndrome in severe COVID-19 patients. Experimental Biology and Medicine, 246(1): p. 5-9.View
Saha, A., et al., (2020). Tocilizumab: A Therapeutic Option for the Treatment of Cytokine Storm Syndrome in COVID-19. Arch Med Res, 51(6): p. 595-597.View
Capra, R., et al., (2020). Impact of low dose tocilizumab on mortality rate in patients with COVID-19 related pneumonia. European Journal of Internal Medicine, 76: p. 31–35.View
Wang, D., et al., (2021). Tocilizumab in patients with moderate or severe COVID-19: a randomized, controlled, open-label, multicenter trial. Frontiers of Medicine, 15(3): p. 486–494.View
Tian, J., et al., (2021). Repurposed tocilizumab in patients with severe COVID-19. Journal of Immunology, 206(3): p. 599–606.View
Ramiro, S., et al., (2020). Historically controlled comparison of glucocorticoids with or without tocilizumab versus supportive care only in patients with COVID-19-associated cytokine storm syndrome: Results of the CHIC study. Annals of the Rheumatic Diseases, 79(9): p. 1143–1151.View
Tsai, A., et al., (2020). Impact of tocilizumab administration on mortality in severe COVID-19. Scientific Reports, 10: p. 19131 View
Rabaan, A.A., et al., (2021). Role of Inflammatory Cytokines in COVID-19 Patients : A Review on Molecular Mechanisms , Immune Functions , Immunopathology and Immunomodulatory Drugs to Counter Cytokine Storm. Vaccines (Basel), 29(9): p. 436.View
Kendall, J.M., (2003). Designing a research project: randomised controlled trials and their principles. Emergency Medicine Journal, 20(2): p. 164-168.View
Gelling Leslie, (2016). Applying for ethical approval for research: the main issues. Nurs Stand, 13(30): p. 40-44.View
Yang Xiaobo, et al., (2020). Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respiratory Medicine, 8(5): p. 475-481.View
Samaee, H., et al., (2020). Tocilizumab for treatment patients with COVID-19: Recommended medication for novel disease. International Immunopharmacology, 89(Part A): p. 107018.View
Medscape (2021). Fact sheet for healthcare providers: Emergency use authorization for ACTEMRA(R) (Tocilizumab). View
Gokhale Yojana, et al., (2021). Tocilizumab improves survival in severe COVID-19 pneumonia with persistent hypoxia: a retrospective cohort study with follow-up from Mumbai, India. BMC Infectious Diseases, 21: p. 241.View
Ragab Dina, et al., (2020). The COVID-19 Cytokine Storm; What We Know So Far. Frontiers in Immunology, 11: p. 1446.View
Hojyo, S., et al., (2020). How COVID-19 induces cytokine storm with high mortality. In Inflammation and Regeneration BioMed Central, 40: p. 37.View
Walter, J.M., Corbridge, T.C., and SInger, B.D., (2018). Invasive Mechanical Ventilation. South Med J, 111(12): p. 746–753.View
National Institutes of Health (2021). Oxygenation and Ventilation - COVID-19 Treatment Guidelines.View
Somers, E.C., et al., (2021). Tocilizumab for Treatment of Mechanically Ventilated Patients With COVID-19. Infectious Diseases Society of America, 73(2): p. e445-e454.View
Farzan Nina, et al., (2021). Invasive mechanical ventilation and clinical parameters in COVID19 patient: Can age be a factor?*. International Journal of Surgery Open, 32: p. 100344.View
Hosey, M.M. and Needham, D.M., (2020). Survivorship after COVID-19 ICU stay. In Nature Reviews Disease Primers Nature Research, 6(1): p. 60View
Moore, J.B. and June, C.H., (2020). Cytokine release syndrome in severe COVID-19. Science, 368(6490): p. 473-474.View
Seguin, A., et al., (2016). Pulmonary Involvement in Patients With Hemophagocytic Lymphohistiocytosis. Chest, 149(5): p. 1294-1301.View
Sadeghi-Haddad-Zavareh, M., et al., (2021). C-Reactive Protein as a Prognostic Indicator in COVID-19 Patients. Interdisciplinary Perspectives on Infectious Diseases, p. 557582,.View
Carubbi Francesco, et al., (2021). Ferritin is associated with the severity of lung involvement but not with worse prognosis in patients with COVID-19: data from two Italian COVID-19 units. Scientific Reports, 11: p. 4863.View
Hussein, A.M., et al., (2021). Dimer and Serum Ferritin as an Independent Risk Factor for Severity in COVID-19 Patients. Materials Today: Proceedings.View