Journal of Public Health Issues and Practices Volume 6 (2022), Article ID: JPHIP-212
https://doi.org/10.33790/jphip1100212Research Article
Comparison of Three Diabetes Risk Screening Tools Among Non-Hispanic Asian Indian Americans: ADA DRT, MDRF IDRS and HbA1c
Rupal M Patel, PT, PhD1*, Ranjita Misra, PhD, CHES, FASHA2, Christina Bickley, PT, PhD, BOCO, C/NDT1, & Katy Mitchell, PT, PhD1
1School of Physical Therapy, Texas Woman’s University, 6700 Fannin Street, Houston, Texas, 77030, United States.
2Professor, Department of Social & Behavioral Sciences, School of Public Health, West Virginia University, Morgantown, WV, United States.
*Corresponding Author Details: Rupal M Patel, PT, PhD., Associate Professor, School of Physical Therapy, Texas Woman’s University, 6700 Fannin Street, Houston, Texas, 77030, United States.
Received date: 08th September, 2022
Accepted date: 27th October, 2022
Published date: 29th November, 2022
Citation: Patel, R.M., Misra, R., Bickley, C., & Mitchell, K., (2022). Comparison of Three Diabetes Risk Screening Tools Among Non-Hispanic Asian Indian Americans: ADA DRT, MDRF IDRS and HbA1c. J Pub Health Issue Pract 6(2): 212.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
The purpose of this study was to compare the relationship between two non-invasive screening tools, the American Diabetes Association Diabetes Risk Test (ADA DRT) and the Madras Diabetes Research Foundation Indian Diabetes Risk Score (MDRF IDRS), with glycated hemoglobin (HbA1c) to determine which tool best predicted HbA1c in Asian Indian Americans. Among Asian Americans, Asian Indian Americans have the highest prevalence of Type 2 diabetes (12.6%). Furthermore, they have a unique genetic predisposition as well as cultural and religious lifestyle practices that may contribute to increased risk. Type 2 diabetes screening tools do not address the unique risk characteristics of Asian Indian Americans. We used clinical and survey data from our previous longitudinal study with a convenience sample of adult Asian Indian Americans (n = 70) selected from a community setting. Following the consenting procedure, all participants completed the ADA DRT and MDRF IDRS questionnaires to identify their diabetes risk. After the initial screening, individuals with an MDRF IDRS score of 50 or above (n = 70) were tested for HbA1c within a month. For this, trained volunteers collected capillary blood using standardized protocol. Descriptive statistics were calculated along with Spearman correlations, Chi-Square test of independence, and linear regressions at an alpha level set at p = .05. A significant, positive relationship (rs = .30, p = .01) was found between the MDRF IDRS score and HbA1c. A positive but weak relationship was also found between ADA DRT and HbA1c (rs = .23). The MDRF IDRS was a better predictor of diabetes risk in Asian Indian Americans than ADA DRT. The MDRF IDRS is a validated, simple, low-cost tool for the detection of diabetes risk in high-risk Asian Indian Americans. Hence, it is a good tool to use for community screenings and patient education for the prevention and management of the risk of Type 2 diabetes.
Key Words: Diabetes Risk Factors, Diabetes Screening, Type 2 Diabetes, Asian Indian Americans, Diabetes Prevention, Community-based Diabetes Screening
Introduction
Currently over 34 million people in the United States (US) have diabetes with another 88 million having prediabetes [1]. The increase in diabetes prevalence is associated with a substantial health and financial burden on the US economy. For example, the estimated cost of diagnosed diabetes was $327 billion in 2017, a 26% increase from 2012 [2]. This cost is expected to rise as the number of individuals diagnosed with diabetes is projected to nearly double by 2060 [3]. Racial and ethnic minorities have a higher prevalence of type 2 diabetes (12.3% – 24.6%) than non-Hispanic Whites (NHW) (12.1%). Furthermore, the non-Hispanic Asian American (NHAA) group is diverse and includes multiple racial groups originating from East Asia, Southeast Asia, and/or South Asians from the Indian subcontinent (which include individuals originating from India, Pakistan, Nepal, Sri Lanka, and Bangladesh). The overall prevalence of diabetes for NHAA is 19.1% and within this diverse group, South Asians have the highest prevalence (23.3%)[4]. Among South Asians, Non-Hispanic Asian Indian Americans (NHAIA), are one of the fastest-growing subgroups that are disproportionately burdened by the disease [1,5].
Awareness and screening of risk factors for diabetes and prediabetes are critical since 25% of all Americans have diabetes and don’t know it. Screening and diagnosis are especially important for NHAA as the rate of undiagnosed diabetes is estimated to be three times higher in this group than that of NHWs [6]. Secondary prevention i.e., screening and early detection can reduce diabetes and related-cardiovascular and heart disease as well as reduce the health and economic burdens of diabetes.
Despite their higher risk of developing diabetes, NHAA have a 34% lower odds of being screened for diabetes compared to NHWs [7]. Hence, in 2015, the American Diabetes Association (ADA) changed its guidelines to recommend screening for diabetes among Asian Americans to improve the recognition of diabetes in this population [8]. In addition, ADA lowered the BMI cut off for being categorized as overweight (≥ 23kg/m2BMI) therefore increasing the rate of screening in this population [9]. This change was particularly helpful for NHAIA as they have greater abdominal obesity despite lower BMI when compared to NHWs in the US [10]. The higher burden of diabetes and coronary artery disease, at the same BMI, in NHAIA as compared to other ethnic groups is well documented [10,11]. However, emerging cardiometabolic risk factors such as waist circumference should be considered when screening for diabetes among NHAIA [12]. Studies have shown waist circumference to be a stronger correlation with diabetes risk factors, independent of BMI [13,14]. Waist circumference of >87 cm in males and >82 cm in females has been suggested as the cut-off point for risk for type 2 diabetes for NHAIA [15].
Adverse physiologic characteristics also make the NHAIAs more susceptible to type 2 diabetes. Among South Asians in the US, lower beta cell function and inability to compensate for higher glucose levels due to insulin resistance are additional factors that lead to higher rates of type 2 diabetes despite lower BMI [16]. There is ample evidence that type 2 diabetes is largely preventable, thus, increased efforts to appropriately screen NHAIA for diabetes is needed. Non-invasive, patient questionnaires can be a helpful first step to screen individuals at risk of type 2 diabetes.
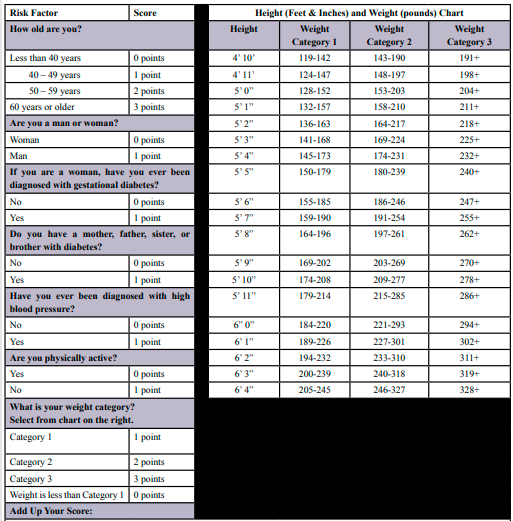
One commonly used non-invasive diabetic screening tool is the American Diabetes Association Diabetes Risk Test (ADA DRT). This instrument is a seven-question survey designed to identify and sum the number of risk factors an individual possesses. The score on the ADA DRT ranges from 0 to 11 with greater than five points deemed as high risk for diabetes. The current ADA DRT is an adaptation of a previously developed diabetic screen by Bang and associates [17] which was developed from a risk-prediction model using representative cross-sectional data from the 1999-2004 National Health and Nutrition Examination Survey (NHANES) questionnaire that inquired about common risk factors among the US population. Risk factors included in the ADA DRT are age (starting risk at age 40), gender, family history of type 2 diabetes, history of gestational diabetes, hypertension, physical activity, and weight (using BMI cut-off of <25kg/m2) [18]. Recently, the ADA DRT was validated in a low-income African American population [19], and in a community-based sample of Latinas [20]. Also, the recommended BMI cut-off to determine diabetes risk for Asian Americans is now ≥23 kg/m2 [18], yet the ADA DRT does not use ethnic specific BMI cut-offs to determine obesity risk. Use of ethnic specific BMI cut-offs has been established as an important risk factor for development of type 2 diabetes in native and migrant Asian Indians, thus the ADA DRT may not provide an accurate risk score for Asian Indians who tend to be leaner in body mass but still have a higher risk for type 2 diabetes [21]. Hence, the utility of the ADA DRT in quantifying risk of type 2 diabetes among a prospective clinical or community based cohort of NHAIA is not known.
There are two non-invasive diabetes risk scores specifically developed and validated for native Asian Indian populations [22,23]. One tool is a five item questionnaire developed by Ramchandran and colleagues [22] utilizing cohorts (n = 10,003) from the National Urban Diabetes Survey (NUDS) in India. Ramchandran’s risk score utilizes age (starting risk at age 30), BMI (using cut-off of <23kg/ m2), waist circumference (using 85 cm as cut-off for males and 80 cm as cut-off for females), family history of diabetes, and sedentary physical activity as risk factors for type 2 diabetes to determine risk with an optimal risk score of greater than 21. This tool was also tested on 567 migrant Asian Indians in the UK revealing that distribution of risk factors for populations of the same ethnic origin living in a different cultural context must be considered when determining appropriate cut-points. This is because in this UK study using cut-points from the native Indian population resulted in high sensitivity but very low specificity, with a high proportion of the migrant Asian Indian UK population testing positive for type 2 diabetes, of whom, a majority would be false positive [22].
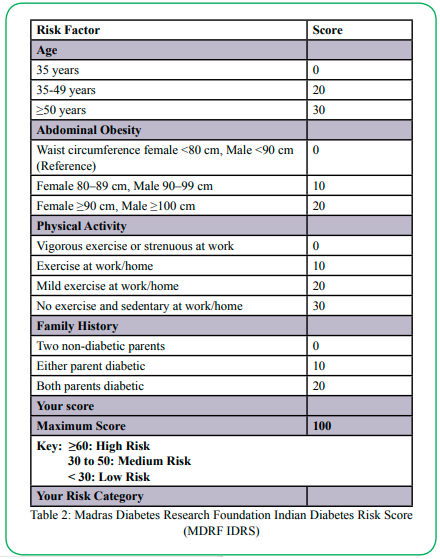
The Madras Diabetes Research Foundation Indian Diabetes Risk Score (MDRF IDRS) is another non-invasive diabetes screening tool developed for use specifically with the native Asian Indian community [23]. The MDRF IDRS has been validated and utilized in multiple sub populations of native and migrant Asian Indians [13,24]. The score (derived from the Chennai Urban Rural Epidemiology Study [CURES]) ranges from 0 to 100 with risk categories of low (<30 score), moderate (30-50 score) and very high (>60 score) [23]. The MDRF IDRS assesses risk factors such as age (starting risk at age 35), abdominal obesity (using waist circumference cut-offs of >90 cm for males and >80 cm for females), family history of diabetes, and leisure time physical activity [23].
The American Diabetic Association 2022 Standards [25] have been revised and currently recommend that screening for prediabetes and diabetes should begin at age 35 years.
Additionally, the new standards recommend screening with an informal assessment of risk factors or validated risk calculator in all asymptomatic adults but in asymptomatic Asian Americans of any age who are overweight (BMI ≥23 kg/m2) or obese (BMI ≥25 kg/m2) with one or more risk factors.
Both the ADA DRT and MDRF IDRS were selected for this study because they are easy to administer and have been widely studied in various populations. Given the higher incidence of type 2 diabetes among NHAIA, it is imperative to determine which diabetes risk screening tool is the best at predicting and/or detecting diabetes among this at-risk population. Much of the evidence that supports use of non-invasive screening tools has been derived from nationally representative data [20] with limited application in the clinical or community setting.
Hence, this study examined and compared two diabetes screening questionnaires (MDRF IRDS and ADA DRT) to determine their sensitivity and predictive ability in a NHAIA sample living in the Southeast Texas region of the United States. The objectives of this study were to determine (1) if there are NHAIA identified as low risk according to the ADA DRT yet are considered as high risk according to the MDRF IDRS, (2) the association between MDRF IDRS and the ADA DRT scores among NHAIA, (3) the association between MDRF IDRS and HbA1c among NHAIA, (4) the association between ADA DRT and HbA1c among NHAIA, and (5) the predictive ability of the MDRF IDRS or ADA DRT on HbA1c, both independently and collectively among NHAIA.
Methods
Study Design
We used clinical and survey data from our previous longitudinal study, a prospective observational study that was conducted in the Southeast Texas region of the United States. The study protocol was approved by the institutional review boards of Rocky Mountain University of Health Professions and Texas Woman's University-Houston. This study was part of a larger randomized controlled trial that compared the effectiveness of a group-based culturally tailored 12-week lifestyle intervention program and standard of care [26].
Participants
A convenience sample of at-risk adult participants was selected from the community setting, a Hindu temple, or mandir (a place of worship) in Houston, Texas. Individuals were eligible for the intervention trial if they were >18 years of age, had a diabetes risk score ≥ 50 as per the MDRF IDRS, HbA1c value < 6.4% (≤47 mmol/ mol), and willing to be randomized and complete all intervention assessments. Participants were excluded if they (a) self-reported diagnosed diabetes, unstable chronic diseases (e.g., cardiac disease or cancer and/or were undergoing treatment), (b) were unable to participate in regular moderate intensity physical activity, (c) were pregnant (self-report) or planning a pregnancy in next 6 months, and/or (d) were currently involved in a supervised program for weight loss. The current study was conducted during the screening and recruitment phase of the randomized controlled intervention trial. Several screening events were held at the mandir. A print flyer was distributed to create awareness and encourage participation in the screening. Purpose of this screening phase was to determine eligibility for the intervention trial. Trained volunteers administered the paper and pen version of the ADA DRT orally in English and the MDRF IDRS in English or Gujarati to 200 individuals during the scheduled screening events at the mandir. Those who met the eligibility criteria (n = 158) were invited to participate in the intervention study. Of those eligible, 78 NHAIA consented to participate and completed baseline testing, during the pre-intervention phase of the trial, including confirming eligibility via HbA1c testing. For the current study, we excluded NHAIA who were found to have diabetes based on subsequent HbA1c testing (n = 4) and those who were not randomly allocated to the study arms (n = 4) due to reaching study’s approved participant limit. The final study sample for data analysis included 70 participants.
Instrumentation
Two paper and pen surveys were concurrently and prospectively administered by trained volunteers in English (ADA DRT) and English and Gujarati (MDRF IDRS). Those with a score of 50 or higher on the MDRF IDRS were invited to receive HbA1c test one to four weeks after the surveys were administered and reviewed to determine eligibility for the intervention trial. The ADA DRT collects self-reported data about seven categorical diabetes risk factors that are depicted in Table 1. Assigned scores for each risk factor are summed for a maximum score of 11. A score of 5 or above indicates risk for diabetes. For this study Asian American BMI categories (normal = >23 kg/m2, overweight = < 23 – 24.9 kg/m2, obese = < 25 kg/m2) were used in the calculation of weight status for the ADA DRT score.
The MDRF IDRS collects self-reported data about four risk factors that are depicted in Table 2. Total score for the MDRF IDRS ranges from 0 to 100 with it categorized as low (<30 score), moderate (30-50 score) and very high (>60 score) risk for diabetes. For this study, an actual measurement of waist circumference was taken to determine the score for the abdominal obesity risk factor on the MDRF IDRS. Those who had a score of 50 or more on the MDRF IDRS were invited back to undergo the HbA1c test as well as complete a demographic questionnaire.
The Bio-Rad Hemoglobin Capillary Collection System for HbA1c Testing was used to obtain a drop of whole blood sample via finger stick to assess glycosylated hemoglobin (HbA1c) to determine glycemic status. HbA1c was selected for this study since it only requires a drop of capillary blood that can be drawn by trained volunteers, can be collected at any time of the day, and does not require an overnight fast or the drawing of multiple blood samples, as is the case for Fasting Plasma Glucose and Oral Glucose Tolerance Test. Thus, this method is appropriate for community settings and reflects the average blood glucose levels in the past three months [27]. American Diabetes Association diagnostic criteria was used to determine prediabetes (HbA1c value 5.7–6.4%), and diabetes (HbA1c value ≥6.5%) [18].
Procedures
Participants were recruited via announcement at the end of religious services at the mandir as well as via flyers posted throughout the mandir campus. Following the consenting procedure, all participants completed the ADA DRT and MDRF IDRS questionnaires in order to identify potential participants’ diabetes risk. After this initial screening, those with an MDRF IDRS score of 50 or above were tested with HbA1c between one to four weeks later. Blood collection was performed by trained volunteers at the mandir using the manufacturer’s standardized protocol for specimen collection, labeling, storage, and shipping. Diabetes Diagnostic Laboratory (University of Missouri, Columbia) conducted the analysis for HbA1c.
Data Analysis
The data was entered into Excel 2016 from paper copy data sheets and double checked by two research team members. For analyses, the data was transferred to SPSS Statistics Version 23 (IBM, USA). Descriptive statistics were calculated along with Spearman correlations, Chi-Square test of independence, and linear regressions at an alpha level set at .05.
Results
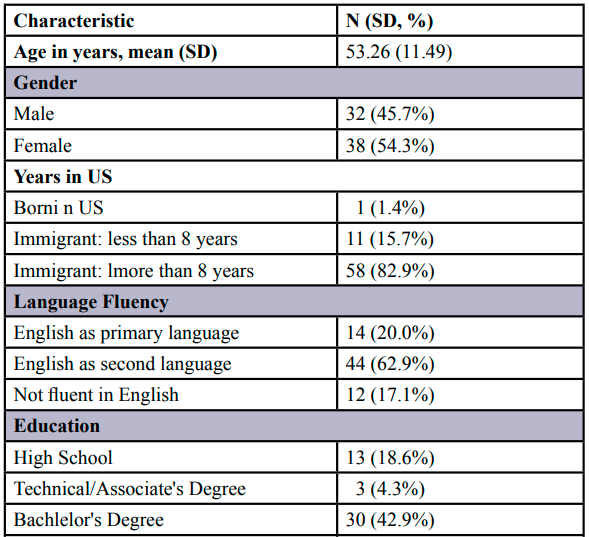
Table 3 presents the demographic characteristics of the study sample (n = 70). The mean age of the 70 participants was 53.26 ± 11.49 years (range: 30-79); the majority (54.3%) were female, married (94.3%), reported English as their second language (62.9%), lived in the US for more than 8 years (82.9%), and were college educated (77.2%). All the participants were currently living in the US. Two-thirds or 64.3% of the participants reported they worked full-time, and 75.7% followed a diet that was lactovegetarian, 20.0% vegan, and 4.3% ovo-lacto vegetarian.
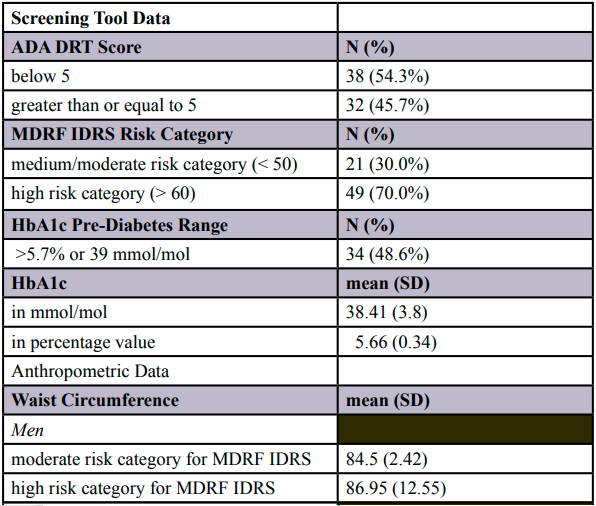
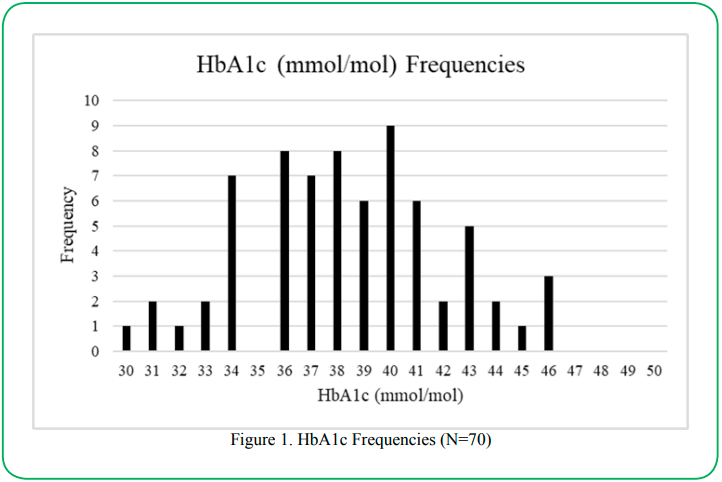
Table 4 presents the anthropometric and screening tool data of the study sample. Mean HbA1c was 38.41 ± 3.8 mmol/mol (range: 30- 46) or 5.66% ± .34 (range: 4.9-6.4). Mean waist circumference for males and females was 88.07 ± 11.03 cm (range: 58-113) and 78.92 ± 8.33 cm (range: 61-99), respectively. Mean systolic and diastolic BP were 123.96 mmHg ± 17.98 (range: 89-188), and 79.57 ± 8.94 mmHg (range: 57-98), respectively. In addition, 84.8% of the sample was overweight or obese per Asian Indian BMI cut-off guidelines with an overall average BMI of 26.29 ± 3.97 (range: 17.17-36.81) [26]. 48.6% of the study sample was categorized as pre-diabetic per American Diabetes Association diagnostic criteria (HbA1c value 5.7–6.4%). This is illustrated in Figure 1.
Table 5 provides the distribution of risk status by screening tools and correlation w HbA1c. The first objective was to determine if there were individuals identified as low risk according to the ADA DRT yet high risk according to the MDRF IDRS. Using the ADA DRT risk score, 54.3% of the sample (N = 38) was categorized as low risk (below 5). Of these low-risk participants, 44.7% were categorized in the medium risk category (score of 30-50) according to the MDRF IDRS. Furthermore, 55.3% of individuals (N = 21) were categorized as low risk using ADA DRT, but high risk (>60 score) according to the MDRF IDRS.
The second objective was to determine if there was an association between ADA DRT and MDRF IDRS scores. Chi-square analysis was conducted to determine if the ADA DRT and the MDRF IDRS were independent of each other, and a Spearman correlation was calculated to determine the exact relationship. The Chi-Square test of independence was found to be significant at χ2 = 56.363, p = .001 suggesting that the two tools measure similar constructs and are not independent of each other. A Spearman correlation revealed a significant positive association between the ADA DRT and MDRF IDRS of rs = .564, p ≤ .0005. However, the moderate rs value suggests that the two tools are capturing different aspects of risk.
The third objective was to determine the association between MDRF IDRS and HbA1c. There was a significant, positive relationship between the two variables at rs = .300, p = .012. The fourth objective was to determine the association between ADA DRT and HbA1c. There was not a significant relationship at rs = .230, p = .056.
The final objective was to determine if the MDRF IDRS or ADA DRT could predict HbA1c independently and/or collectively. The ADA DRT was a significant positive predictor of HbA1c at p = .043, r = .243. In addition, MDRF IDRS was a significant predictor of HbA1c at p = .012, r = .299. When entered into the model together, the two tools predicted 9.5% of HbA1C at p = .035, r = .309.
Discussion
This is the first prospective study to compare two non-invasive diabetes screening tools in a community-based sample of NHAIA, with the highest diabetes risk among all NHAA subgroups. In terms of diabetes risk, on the ADA DRT, 45.7% of the study sample (n = 32) was deemed at risk (score >5), whereas, on the MDRF IDRS, 84.3% of the study sample (n = 59) was deemed at medium/high risk (score >50). Despite using the ADA recommended lower overweight cut-point of 23 kg/m2 for BMI, the ADA DRT did not capture higher number of NHAIA as being at risk for diabetes as the MDRF IDRS screening tool. Previous studies on Asian Indians have shown that BMI may not be a good indicator of risk because it does not capture excess fat mass of an individual who is considered not overweight or nonobese per BMI; excess fat mass has shown to increase metabolic and cardiovascular risks among Asian Indians [10,28,29]. The MDRF IDRS includes waist circumference as a risk factor (instead of BMI) and confirms it is a better predictor of diabetes risk for NHAIA [2830]. While both the MDRF IDRS (rs = .30) and ADA DRT (rs =.23) correlations with HbA1c were weak, the MDRF IDRS was found to have significant and slightly higher correlation to HbA1c in this sample population of NHAIA.
We also examined the ability to predict HbA1c levels for both screening tools. Both the ADA DRT and MDRF IDRS were found to be significant predictors of HbA1c. Similar to the accuracy of risk-assessments, the MDRF IDRS performed slightly better than the ADA DRT in this sample. However, future studies should explore the use of both screening tools for a better HbA1c prediction since our analysis showed that when both tools were entered into the model, performance of prediction improved.
Comparison of the two screening tools showed that differing risk categories can be assigned to the same subject when using two different risk tools. For example, 30% of individuals were categorized as low risk using ADA DRT but high risk according to the MDRF IDRS. A significant but moderate association between the ADA DRT and MDRF IDRS indicates that while they are similar, these two screening tools are not the same. To further examine how these two tools relate to each other, a significant Chi-Square test of independence showed the two screening tools are measuring similar but not the same constructs that help explain the differing risk categories assigned to the same subjects.
The finding that the ADA DRT and MDRF IDRS screening tools are measuring differing constructs of diabetic risk is logical when comparing the specific domains and questions contained in each. While the ADA DRT has seven domains (as compared to the four domains of the MDRF IRDS), it lacks a specific question on abdominal obesity (that is included in the MDRF IDRS). Furthermore, in our study, the abdominal obesity was not self-reported, but waist circumference was measured by trained volunteers as part of the screening phase of the larger trial. Recent studies have illustrated that using BMI to determine body fat percentage may not be as accurate as previously thought [10,29]. Other anthropometrics such as waist circumference may reflect better correlation to abdominal body fatness compared to BMI [11,28,30]. In addition to the lack of an abdominal obesity question, the physical activity question on the ADA DRT, is a yes/no question as opposed to the four-level physical activity question included on the MDRF IDRS. The more detailed physical activity question on the MDRF IRDS provides additional information that may be valuable in creating a more reliable screening tool for NHAIA.
The MDRF IDRS assesses family history of diabetes. However, it does not explore diabetes prevalence among siblings or gestational diabetes (which is found on the ADA DRT). In addition, the MDRF IDRS also does not assess high blood pressure as found on the ADA DRT. Age ranges also differ between the two screening tools. For example, the ADA DRT has four age levels that range from less than 40 years to greater than 60 years while the MDRF IDRS has three age levels that range from less than 35 years to greater than 50 years. This is because studies have shown that Asian Indians are at risk for diabetes at a younger age than NHWs and other ethnic groups [31,32]. Comparing the domains as well as specific question content and type between these two diabetes screening tools helps to explain the moderate correlations found between them as well as the reason different risk categories were assigned to the same participants. The inclusion of abdominal obesity in the MDRF IDRS may help explain the slightly better association with HbA1c in our sample of NHAIA and is congruent with previous studies that have demonstrated presence of increased abdominal adiposity while having a lower BMI [10,28,29,30]. The inclusion of waist circumference as a screening question that is answered via measurement could be a beneficial risk factor to assess quickly in a clinical or community setting, not just for NHAIA but also for other ethnicities and/or body types with high incidence of diabetes [33]. Additional tool differences that could explain the slightly better performance of the MDRF IDRS in this study could be the development of the MDRF IRDS in this sample’s native country of India as well as offering a version of the MDRF IDRS in the Gujarati language, the native or first language of many of the participants of this study.
The prospective nature of this study is a strength as previous and similar studies on diabetes screening tools were mainly retrospective in design [13,23,24]. The collection of corresponding HbA1c levels along with risk scores is another strength of this study. The gap in timing of survey completion to HbA1c collection varied from one to four weeks, which was a weakness of this study; this was due to time and space availability at the mandir and the availability of trained volunteers to collect HbA1c blood samples. The homogenous nature of this study’s sample is both a strength and weakness. The strength of examining a homogeneous sample is the ability to determine the specific risk factors and needs of a population of NHAIA. As the need for precision medicine in diabetes [34,35,36] becomes evident having diabetes screening tools that capture ethnic-specific risk factors may improve the precision [34] aspect of overall delivery of clinical and community based health promotion, risk reduction, risk detection, diagnosis, and disease management programs for diabetes and its related cardiovascular conditions. At the same time, the weakness of having an NHAIA sample from one Houston, Texas area mandir limits the generalizability of this study’s results. Future studies should replicate our methodology in a larger sample across multiple US regions including a wider South Asian American population, beyond the NHAIAs in our sample, as this will reflect the varied ethnicity and cultural practices of the larger South Asian diaspora.
Conclusion
Overall, while the MDRF IDRS performed slightly better in this study than the ADA DRT, both tools correlated surprising low with HbA1c levels. Future research should determine if participants scoring high risk on both screens have better correlations to HbA1c levels. Comparing and contrasting the domains and specific questions of both screening tools may yield positive aspects and room for improvement on each tool. Future research should also explore the creation of a new and improved diabetes screening tool that combines Asian specific BMI and waist circumference [33] to assess risk for NHAIAs. This would combine the strengths of both the MDRF IRDS, and the ADA DRT, and such a precise tool can be easily implemented in a clinical or community setting. Finally, future prospective research should also continue to compare screening tool results with HbA1c levels in a larger, ethnically, and culturally varied, as well as geographically diverse population of South Asians living in the US. Developing an accurate diabetes risk screening tool is a crucial step in improving the health of those at risk of developing this debilitating and costly chronic condition. With Type 2 Diabetes being the most common and most preventable type of diabetes, developing a cross-cultural and precise screening tool has the potential to have a significant positive impact on the health outcomes of Asian Indian Americans.
Competing Interests:
The authors declare that they have no competing interests.
Acknowledgements
Briana Durand, PT, DPT and Huzefa Tayabali, PT, DPT helped with aspects of this research project including literature review in partial fulfillment of the requirements for their Doctor of Physical Therapy (DPT) degree from Texas Woman’s University-Houston, Texas.
References
Prevention, C.f.D.C.a. (2020). National Diabetes Statistics Report. ;Available from: https://www.cdc.gov/diabetes/data/ statistics-report/index.html.View
Association, A.D., (2018). Economic Costs of Diabetes in the U.S. in 2017. Diabetes Care, 41(5): p. 917-928.View
Lin, J., et al., (2018). Projection of the future diabetes burden in the United States through 2060. Population Health Metrics, 16(1): p. 9.View
Cheng, Y.J., et al., (2019). Prevalence of Diabetes by Race and Ethnicity in the United States, 2011-2016. JAMA, 322(24): p. 2389-2398.View
DHHS, O.U. (2021). Diabetes and Asian Americans - The Office of Minority Health; Available from: https://minorityhealth.hhs. gov/omh/browse.aspx?lvl=4&lvlid=48.View
Menke, A., et al., (2015). Prevalence of and Trends in Diabetes Among Adults in the United States, 1988-2012. JAMA, 314(10): p. 1021.View
Tung, E.L., et al., (2017). Racial and Ethnic Disparities in Diabetes Screening Between Asian Americans and Other Adults: BRFSS 2012–2014. Journal of General Internal Medicine, 32(4): p. 423-429.View
Association, A.D., (2015). Standards of Medical Care in Diabetes—2015 Abridged for Primary Care Providers. Clinical Diabetes, 33(2): p. 97-111.View
Araneta, M.R.G., et al., (2015). Optimum BMI Cut Points to Screen Asian Americans for Type 2 Diabetes. Diabetes Care, 38(5): p. 814-820.View
Misra, A., (2015). Ethnic-Specific Criteria for Classification of Body Mass Index: A Perspective for Asian Indians and American Diabetes Association Position Statement. Diabetes Technology & Therapeutics, 17(9): p. 667-671.View
Bajaj, H.S., et al., (2014). Comparison of Relative Waist Circumference between Asian Indian and US Adults. Journal of Obesity, 2014: p. 1-10. View
Kotha, P., et al., (2012). Modified criteria for determining cardiometabolic syndrome in Asian Indians living in the USA: Report from the diabetes among Indian Americans national study. International journal of cardiology., 155(2): p. 343-345.View
V, M., et al., (2007). A diabetes risk score helps identify metabolic syndrome and cardiovascular risk in Indians - the Chennai Urban Rural Epidemiology Study (CURES-38). Diabetes, obesity & metabolism, 9(3).View
M, V., et al., (2020). A composite of BMI and waist circumference may be a better obesity metric in Indians with high risk for type 2 diabetes: An analysis of NMB-2017, a nationwide cross-sectional study. Diabetes research and clinical practice, 161.View
Thomas, A. and A. Ashcraft, (2013). Type 2 Diabetes Risk among Asian Indians in the US: A Pilot Study. Nursing Research and Practice, 2013: p. 1-8.View
Kanaya, A.M., et al., (2014). Understanding the High Prevalence of Diabetes in U.S. South Asians Compared With Four Racial/ Ethnic Groups: The MASALA and MESA Studies. Diabetes Care, 37(6): p. 1621-1628.View
H, B., et al., (2009). Development and validation of a patient self-assessment score for diabetes risk. Annals of internal medicine, 151(11).View
Association, A.D., (2009). 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care, 2021. 44(Supplement_1): p. S15-S33.View
Chima, C.C., et al., (2020). 1442-P: Validity of the American Diabetes Association Diabetes Risk Test in a Low-Income African-American Population | Diabetes | American Diabetes Association. Diabetes, p. 1442-P.
Scanlan, A.B., et al., (2018). Diabetes Risk Assessment in Latinas: Effectiveness of a Brief Diabetes Risk Questionnaire for Detecting Prediabetes in a Community-Based Sample. Diabetes Spectrum, 31(1): p. 31-36.View
A, M. and K. L, (2009). The metabolic syndrome in South Asians: epidemiology, determinants, and prevention. Metabolic syndrome and related disorders, 7(6).View
A, R., et al., (2005). Derivation and validation of diabetes risk score for urban Asian Indians. Diabetes research and clinical practice, 70(1).View
V, M., et al., (2005). A simplified Indian Diabetes Risk Score for screening for undiagnosed diabetic subjects. The Journal of the Association of Physicians of India, 53.View
Nugawela MD, S.S., Mohan V, Rajalakshmi R, Netuveli G, (2020). Evaluating the Performance of the Indian Diabetes Risk Score in Different Ethnic Groups. Diabetes Technol Ther.View
Standards of Medical Care in Diabetes—2022 Abridged for Primary Care Providers. Clinical Diabetes, 2022. 40(1): p. 10-38.View
Patel, R.M., et al., (2017). Effectiveness of a Group-Based Culturally Tailored Lifestyle Intervention Program on Changes in Risk Factors for Type 2 Diabetes among Asian Indians in the United States. Journal of Diabetes Research, 2017: p. 1-13.View
Misra, R., et al., (2016). Community-Based Diabetes Screening and Risk Assessment in Rural West Virginia. Journal of Diabetes Research, 2016: p. 1-9.View
Dudeja, V., et al., (2001). BMI does not accurately predict overweight in Asian Indians in northern India. British Journal of Nutrition, 86(1): p. 105-112.View
Vikram, N.K., et al., (2003). Non-obese (body mass index < 25 kg/m2) Asian Indians with normal waist circumference have high cardiovascular risk. Nutrition, 19(6): p. 503-509.View
Banerji, M.A., et al., (1999). Body Composition, Visceral Fat, Leptin, and Insulin Resistance in Asian Indian Men1. The Journal of Clinical Endocrinology & Metabolism, 84(1): p. 137-144.View
Gujral, U.P., et al., (2013). Type 2 diabetes in South Asians: similarities and differences with white Caucasian and other populations. Annals of the New York Academy of Sciences, 1281(1): p. 51-63.View
Shah, A. and A.M. Kanaya, (2014). Diabetes and Associated Complications in the South Asian Population. Current Cardiology Reports, 16(5).View
Venkatrao, M., et al., (2020). A composite of BMI and waist circumference may be a better obesity metric in Indians with high risk for type 2 diabetes: An analysis of NMB-2017, a nationwide cross-sectional study. Diabetes research and clinical practice., 161: p. 108037.View
Chung, W.K., et al., (2020). Precision medicine in diabetes: a Consensus Report from the American Diabetes Association (ADA) and the European Association for the Study of Diabetes (EASD). Diabetologia, 63(9): p. 1671-1693.View
Klonoff, D.C., et al., (2020). The Need for Precision Medicine to be Applied to Diabetes. Journal of Diabetes Science and Technology, 14(6): p. 1122-1128.View
Xie, F., J.C. Chan, and R.C. Ma, (2018). Precision medicine in diabetes prevention, classification and management. Journal of Diabetes Investigation, 9(5): p. 998-1015.View