Journal of Public Health Issues and Practices Volume 10 (2026), Article ID: JPHIP-257
https://doi.org/10.33790/jphip1100257Research Article
Associations Between Unregulated Medical Drug Distribution and Public Health Outcomes in Nigeria
Ibrahim Alliu1*, Dr.PH, Subash Thapa1, MPH, and Hameed Jimoh2, MSc,
1Jiann-Ping Hsu College of Public Health, Department of Biostatistics, Epidemiology and Environmental Sciences, Georgia Southern University, Statesboro, Georgia, United States.
2Independent Researcher, Softmark Technologies Inc., Canada.
Corresponding Author Details: Ibrahim Alliu, Assistant Professor, Jiann-Ping Hsu College of Public Health, Department of Biostatistics, Epidemiology and Environmental Sciences, Georgia Southern University, 501 Forest Dr, Statesboro, GA 30458, United States.
Received date: 09th March, 2026
Accepted date: 21st April, 2026
Published date: 23rd April, 2026
Citation: Alliu, I., Thapa, S., & Jimoh, H., (2026). Associations Between Unregulated Medical Drug Distribution and Public Health Outcomes in Nigeria. J Pub Health Issue Pract 10(1): 257.
Copyright: ©2026, This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Abstract
Unregulated drug distribution and informal healthcare in Nigeria pose severe medication-related risks, driven by a lack of professional oversight and the proliferation of counterfeit medicines. This study identified the determinants of adverse health outcomes through a cross-sectional survey of 663 participants across 15 Nigerian states. Using multivariate logistic regression, we found that uncertainty regarding a licensed pharmacist's presence was the strongest predictor of adverse outcomes (AOR = 18.891; 95% CI: 7.876-45.310), followed by the confirmed absence of a pharmacist (AOR = 13.280; 95% CI: 4.674-37.730). Failing to consult a physician (AOR = 4.877; 95% CI: 2.477–9.599) and sourcing drugs from unlicensed stores (AOR = 2.544; 95% CI: 1.443-4.484) also significantly increased complication risks. Notably, avoiding informal nurse services was highly protective (AOR = 0.015; 95% CI: 0.004-0.055). These findings highlight that a deficit in professional oversight is a primary driver of pharmaceutical harm in Nigeria. Urgent policy interventions are required to strengthen regulatory enforcement, improve access to qualified professionals, and bolster national pharmacovigilance.
Key words: Drug Accessibility; Health Policy; Pharmacovigilance; Self-medication; Unregulated Drug Distribution; Informal Healthcare; Nigeria; Medication Safety;
Introduction
Access to safe, regulated medicines is fundamental to public health. In many low- and middle-income countries (LMICs), the widespread use of drugs from unregulated channels presents a persistent public health challenge. The World Health Organization (WHO) estimates that at least one in ten medical products in LMICs is substandard or falsified [1], with Africa having the highest regional prevalence at 18.7% [2]. This issue is exacerbated in developing nations where regulatory frameworks may be weaker, allowing poor-quality medicines to disproportionately affect public health [3].
In Nigeria, this problem is particularly acute. The easy availability of a wide array of medications, from analgesics and antibiotics to controlled substances like tramadol, without professional oversight, has led to increased medication abuse and adverse reactions [4]. The inability to create an effective national system of drug supply has increased the prevalence of substandard drugs, which are often stored in improper conditions that compromise their efficacy and safety. Consequently, individuals often bypass trained healthcare professionals, leading to treatment failures, drug resistance, and other medication-related complications [4].
Even with regulatory authorities like the National Agency for Food and Drug Administration and Control (NAFDAC), institutional shortcomings, such as resource inadequacy and disorganized supply chains, hinder proper enforcement. The regulatory deficit is compounded by the dependence of the population on para-health practitioners, including auxiliary nurses, who might not receive standardized training but prepare and administer medicines, including intravenous medications, with insufficient supervision.
While the pitfalls of illicit drugs and unofficial healthcare in Nigeria have been acknowledged, to date, the field lacks modern, extensive statistics quantifying the public health impacts and elucidating specific, changeable risk factors. The purpose of this study is to fill the void by examining the determinants of unfavorable health consequences from the unofficial drug distribution network of Nigeria and the unofficial healthcare system [4].
Although the challenges of unregulated drugs and informal healthcare in Nigeria are recognized, there remains a critical need for current, comprehensive data that quantifies the public health consequences and identifies specific, modifiable risk factors. This study aims to address this gap by investigating the determinants of adverse health outcomes arising from Nigeria's unregulated drug distribution system and informal healthcare sector.
The specific objectives of this study were:
1. To assess the patterns of drug accessibility, self-medication, and informal healthcare use across diverse Nigerian settings.
2. To identify key predictors associated with adverse health outcomes, including the role of pharmacist availability and physician consultation.
3. To evaluate the specific risks associated with sourcing drugs from unlicensed vendors and using informal healthcare providers.
Materials And Methods
Study Design and Setting
This study employed a cross-sectional quantitative survey design, conducted between October 2023 and December 2023 across 15 Nigerian states, including the Federal Capital Territory (FCT). The selection of states ensures proportional representation across Nigeria’s six geopolitical zones:
• North-Central: Kwara, Niger, FCT
• North-East: Bauchi, Borno
• North-West: Kano, Kaduna, Katsina
• South-West: Lagos, Oyo, Ogun
• South-South: Edo, Rivers
• South-East: Anambra, Enugu
Urban and rural communities were included to enhance regional diversity and the generalizability of findings.
Study Population and Sampling
Eligible participants were individuals aged 16 years and older residing in the selected states, with healthcare professionals being excluded. The legal basis for autonomous consent from participants aged 16-17 was Nigeria’s Child’s Rights Act 2003 (section 64(2)) [5]. Participants were recruited using a multi-stage, stratified sampling technique in public areas to ensure representativeness. Of 1000 individuals approached, 876 were eligible, and 663 consented and completed the survey (a 75.7% participation rate among those eligible). The sample size was determined using Cochran’s formula, targeting a 95% confidence level and 5% margin of error, with a 10% allowance for non-response.
Data Collection Instrument and Procedures
Data were collected using a structured, interviewer-administered questionnaire developed from existing literature and refined with expert input. The instrument assessed demographics, drug accessibility, self-medication behaviors, healthcare-seeking patterns, and self-reported adverse health outcomes. The questionnaire, which included dichotomous, categorical, and Likert-type scale questions, was pilot-tested for clarity with 30 individuals. It demonstrated acceptable internal consistency. Trained field workers conducted interviews in English or translated them into major local languages to ensure accurate data capture.
Ethical Considerations
This study was approved by the National Health Research Ethics Committee (NHREC) of Nigeria. All participants provided informed consent after a full briefing on the study's purpose and their right to voluntary withdrawal. Written consent was obtained from literate participants, while documented verbal consent was obtained from those unable to write, as approved by the ethics committee. All procedures were conducted in accordance with the Declaration of Helsinki and the National Code for Health Research Ethics in Nigeria.
Statistical Analysis
Data were analyzed using SAS version 9.4. Descriptive statistics (frequencies, percentages, means) were used to summarize data. Associations between categorical variables were explored with chi- square tests. Two multivariate logistic regression models were used to identify independent predictors of dichotomous adverse health outcomes, with adjusted odds ratios (AORs) and 95% confidence intervals (CIs). Model fitness was assessed using the Area Under the receiver operating characteristic (ROC) curve (AUC). For regression models, age was treated as a continuous variable, while other predictors were treated as categorical with defined reference groups. A p-value < 0.05 was considered significant, and cases with missing data (<2%) were excluded via listwise deletion.
Results
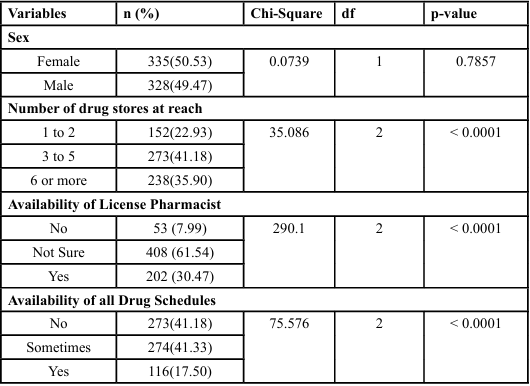
The socio-demographic characteristics of the 663 study participants, with a near-equal gender distribution (50.5% female, 49.5% male). The mean participant age was 31.4 ± 7.8 years. Key characteristics and practices are detailed in Table 1. Notably, a majority of participants (61.5%) were uncertain about the availability of a licensed pharmacist at their usual drugstores, and a high proportion (76.8%) reported using informal nurses for healthcare. Adverse effects were reported by 21.1% of participants after using informal nurse services and by 15.2% after using unprescribed drugs.
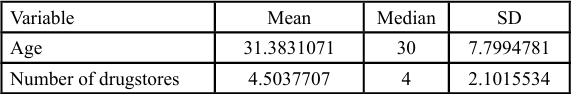
Table 2 presents the descriptive statistics for the study's continuous variables: participant age and drugstore accessibility. The participants had a mean age of 31.38 ± 7.80 years (Median = 30). Regarding accessibility, participants reported a mean of 4.50 ± 2.10 available drugstores (Median = 4).
Two multivariate logistic regression models were developed to identify factors associated with these adverse health outcomes. The first model (Table 3) assessed predictors of adverse outcomes following consultation with informal healthcare providers. The second model (Table 4) evaluated predictors related to the use of unprescribed or potentially substandard medications.
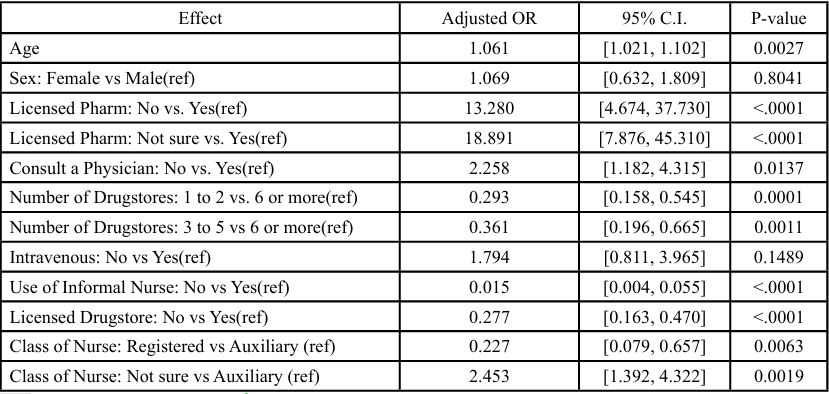
Table 3: The odds ratio of the predictors of adverse health outcomes associated with using informal nurses.
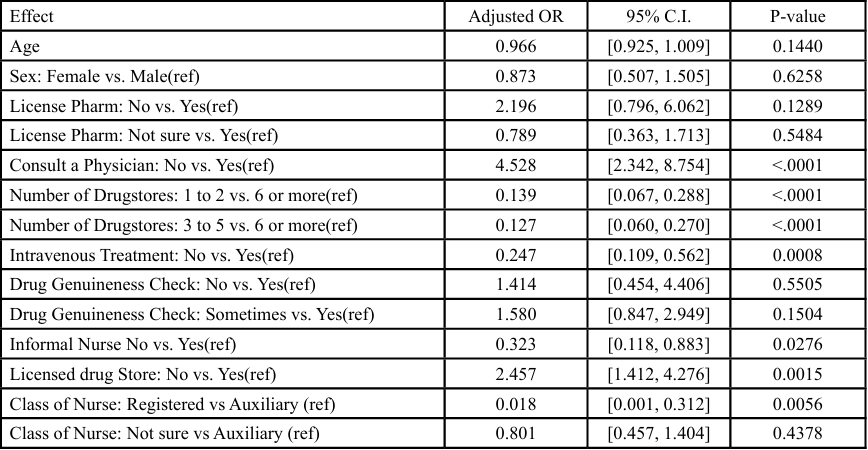
Table 4: The odds ratio of the predictors of adverse health outcomes associated with the accessibility of drugs
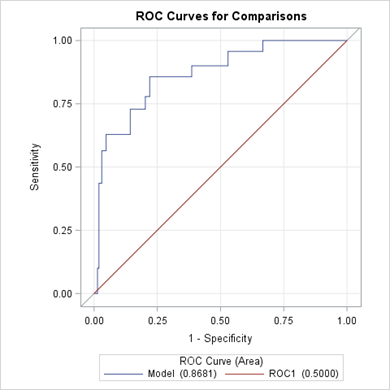
In the first model (Table 3), increasing age was a significant risk factor (AOR = 1.061, 95% CI: 1.021-1.102). Compared to drugstores with a confirmed licensed pharmacist, the odds of adverse effects were dramatically higher when a pharmacist was absent (AOR = 13.280, 95% CI: 4.674-37.730) or when participants were unsure of the pharmacist's presence (AOR = 18.891, 95% CI: 7.876- 45.310). Not consulting a physician also increased risk (AOR = 2.258, 95% CI: 1.182-4.315). Accessing drugs from 1–2 drugstores (AOR = 0.293, 95% CI: 0.158-0.545) or 3-5 drugstores (AOR = 0.361, 95% CI: 0.196-0.665) compared to 6 or more was significantly protective. Not using informal nurse services was strongly protective (AOR = 0.015, 95% CI: 0.004-0.055), and registered nurses were associated with significantly lower odds of adverse outcomes compared to auxiliary nurses (AOR = 0.227, 95% CI: 0.079-0.657). The model demonstrated excellent predictive capability (AUC = 0.8681) as seen in Figure 1 below.
Factors Associated with Adverse Health Outcomes from Unprescribed or Potentially Substandard Drug Use
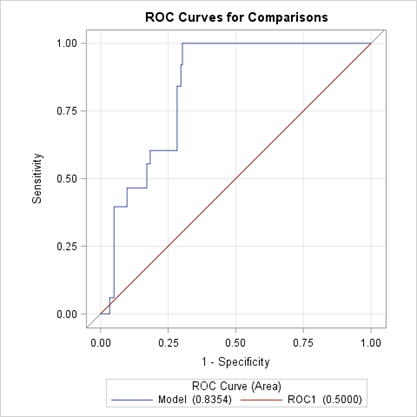
In the second model (Table 4), Firth’s penalized maximum likelihood estimation was applied due to quasi-complete separation associated with the nurse classification variable, yielding stable and fully interpretable estimates for all predictors. Not consulting a physician remained the strongest predictor of complications (AOR = 4.528, 95% CI: 2.342-8.754). Purchasing drugs from unlicensed stores was also significantly associated with increased risk (AOR = 2.457, 95% CI: 1.412-4.276). Having access to fewer drugstores was significantly protective: those visiting 1-2 stores (AOR = 0.139, 95% CI: 0.067- 0.288) and 3-5 stores (AOR = 0.127, 95% CI: 0.060-0.270) had markedly lower odds than those with 6 or more. Not receiving intravenous drips was also protective (AOR = 0.247, 95% CI: 0.109 0.562). Registered nurses had significantly lower odds of adverse drug outcomes than auxiliary nurses (AOR = 0.018, 95% CI: 0.001- 0.312). This model demonstrated excellent predictive capability (AUC = 0.8359, Nagelkerke R² = 0.326; Figure 2).
Discussion
This study aimed to identify the determinants of adverse health outcomes arising from Nigeria's unregulated drug distribution system and informal healthcare sector. Our analysis revealed that the lack of a licensed pharmacist at the point of sale and the failure to consult a physician are the most critical predictors of medication-related harm. These findings, contextualized below, provide a robust evidence base for targeted public health interventions.
The finding that the absence of a licensed pharmacist was associated with a more than 13-fold increase in the odds of adverse effects is a confirmation of the role of pharmacists in drug safety. This aligns with global research demonstrating that pharmacist-led medication management is pivotal in reducing adverse drug reactions and improving medication adherence [6,7]. In an environment with substandard drugs, the pharmacist serves as a crucial barrier, providing patient counseling, identifying potential drug interactions, and screening for counterfeit products. The most concerning finding was that the uncertainty about a pharmacist's presence carried an even higher risk (AOR ≈ 19) in the informal nurse model, suggesting these ambiguous environments may represent the most severe lapse in regulatory adherence. This emphasizes the urgent need for stricter enforcement of regulations mandating pharmacist presence in all drug outlets.
Our study strongly reinforces the dangers of self-medication, as participants who did not consult a physician had nearly five times the odds of experiencing medication-related complications. This is consistent with studies by Osemene & Lamikanra and Zheng et al. [7,8] that link self-medication to inappropriate drug use, antimicrobial resistance, and drug toxicity. Having access to fewer drugstores is found to be protective. A higher density of outlets, likely including many unregulated vendors, encourages more frequent and haphazard drug procurement, thereby increasing risk, a phenomenon reported in other poorly regulated markets [9].
A high dependence on informal nurses emerged as a primary predictor of adverse outcomes in Model 1. The pronounced protective effect observed among participants who avoided informal nurse services (AOR = 0.015) underscores the systemic risks associated with unregulated clinical practices. Furthermore, the significant distinction between consulting a registered nurse versus an auxiliary nurse or an unverified practitioner (AOR = 0.227) confirms that formal professional training is a fundamental determinant of patient safety in nurse-led interventions. These data emphasize the necessity for standardized national policies regarding the certification and supervised scope of practice for auxiliary health workers.
However, Model 2, which specifically addressed adverse drug outcomes from non-prescribed medications, presented a more complex statistical landscape. Registered nurses were associated with significantly lower odds of drug-related complications compared to auxiliary nurses (AOR = 0.018, 95% CI: 0.001-0.312). While a similar trend was observed for participants unsure of their practitioner's qualifications (AOR = 0.801), this did not reach statistical significance (p = 0.438).
Several structural and statistical factors may explain this pattern. First, the auxiliary nurse subgroup was relatively small (n = 70), and despite the application of Firth’s penalized likelihood, the resulting wide confidence intervals suggest statistical instability rather than a definitive protective effect. Second, Model 2 isolates outcomes from non-prescribed drug use; it is plausible that non-registered practitioners in these settings recommend fewer or less complex drug combinations, inadvertently reducing the risk of polypharmacy- related harm. Third, a systematic underreporting bias may exist, as patients utilizing non-registered practitioners often possess lower health literacy and may be less equipped to recognize or report adverse drug reactions. Finally, residual confounding likely plays a role, as these practitioners predominantly operate in resource- constrained environments where limited drug availability inherently reduces the frequency of medication-related incidents. Consequently, these findings reflect the intricate nature of Nigeria’s informal healthcare sector and suggest that future longitudinal research with larger subgroup cohorts and objective clinical measures is required to further delineate these associations.
That drug sourcing through unlicensed stores doubled the risk of adverse effects by over two times becomes a direct indictment of the unregulated market. This reflects the cumulative risk of receiving substandard or counterfeit drugs, improper storage conditions, and advice from unqualified staff, reinforcing the need for aggressive regulatory enforcement [10]. Observe that self-reported drug authenticity verification was not a significant predictor in our model. That does not eliminate the risk of drug counterfeits, but it does suggest consumer-level verification as yet another effective drug counterfeit prevention measure that becomes ineffective in the era of highly advanced counterfeit technologies, and a system-level solution, like intensified pharmacovigilance and safe supply chains, must become the answer of preference [11].
Strengths and Limitations
This study’s strengths include its large, geographically diverse sample across Nigeria’s six geopolitical zones and its use of multivariate analysis to control for confounders. However, several limitations must be acknowledged. "While this study provides robust associations, the cross-sectional design focuses on point-in-time correlations rather than longitudinal causality. Additionally, the use of self-reported data introduces the potential for recall and social desirability biases, which are common in large-scale community surveys. The measurement of adverse effects was based on participant reports rather than clinically validated scales, which, while pragmatic for a large survey, lacks clinical specificity. Finally, the use of convenience sampling for participant recruitment at the final stage may limit the generalizability of the findings. Future research should employ longitudinal designs and objective measures to confirm these associations and explore the efficacy of regulatory interventions.
Conclusion
This study provides critical evidence that the absence of professional oversight from pharmacists and physicians is a primary driver of adverse health outcomes in Nigeria's unregulated drug market. These findings indicate that systemic reforms prioritizing patient safety over unregulated accessibility are essential to mitigating medication- related harm. Ultimately, creating a safer pharmaceutical landscape requires a committed, multi-sectoral approach to rebuild trust in the medicine supply chain and ensure every citizen has access to safe, effective, and properly regulated healthcare.
Recommendations Based on the findings of this study, a multi- pronged strategy is recommended, focused on three core areas:
1. Regulatory and Policy Reform: A robust regulatory overhaul is essential. This includes expediting the full implementation of the National Drug Distribution Guidelines (NDDG) to create a secure supply chain and empowering agencies like NAFDAC and the Pharmacists Council of Nigeria (PCN) with greater resources and authority for stricter, routine enforcement against unlicensed vendors.
2. Healthcare Workforce Strengthening: The healthcare workforce at the community level must be strengthened. Policies should be implemented to ensure licensed pharmacists are present at all drug outlets. Concurrently, national standards for the training, certification, and supervised integration of auxiliary nurses into the primary healthcare system must be established to formalize their role and improve patient safety.
3. Public Health and Safety Systems: National pharmacovigilance programs and sustained public awareness campaigns must be enhanced. These initiatives are critical to monitor medication safety effectively and to educate citizens on the dangers of self medication and the importance of seeking advice from qualified healthcare professionals.
List of Abbreviations
• AOR: Adjusted Odds Ratio
• AUC: Area Under the Curve
• CI: Confidence Interval
• FCT: Federal Capital Territory
• LMICs: Low- and Middle-Income Countries
• NAFDAC: National Agency for Food and Drug Administration and Control
• NDDG: National Drug Distribution Guidelines
• NHREC: National Health Research Ethics Committee
• OTC: Over-the-Counter
• PCN: Pharmacists Council of Nigeria
• ROC: Receiver Operating Characteristic
• SAS: Statistical Analysis System
• WHO: World Health Organization
• SD: Standard Deviation
Sources of Financial Support:
This research did not receive any grants from any funding agencies.
Conflict of Interest:
The authors declare that they have no affiliations with or involvement in any organization or entity with any financial interests in the subject matter or materials discussed in this manuscript.
References
Daniela Bagozzi Communication Officer WHO. (2017, November 28). Medical products in developing countries is substandard or falsified. View
Ozawa, S., Evans, D. R., Bessias, S., Haynie, D. G., Yemeke, T. T., Laing, S. K., & Herrington, J. E. (2018). Prevalence and Estimated Economic Burden of Substandard and Falsified Medicines in Low- And Middle-Income Countries: A Systematic Review and Meta-analysis. In JAMA Network Open (Vol. 1, Number 4). American Medical Association. View
Nayyar, G. M. L., Breman, J. G., Newton, P. N., & Herrington, J. (2012). Poor-quality antimalarial drugs in southeast Asia and sub-Saharan Africa. In The Lancet Infectious Diseases (Vol. 12, Number 6, pp. 488–496). View
Onyima, B. N. (2023). Prescription opioids, consumption cultures and “informal governing images” among “young street guys” in Nigeria. Drugs, Habits and Social Policy, 24(4), 270- 284. View
Nigeria: Act No. 26 of 2003, Child’s Rights Act, 2003. (2003). View
Alaa Eddine, N., Schreiber, J., El-Yazbi, A. F., Shmaytilli, H., & Amin, M. E. K. (2023). A pharmacist-led medication review service with a deprescribing focus guided by implementation science. Frontiers in Pharmacology, 14. View
McGettigan, S., Curtin, D., & O’Mahony, D. (2024). Adverse Drug Reactions in Multimorbid Older People Exposed to Polypharmacy: Epidemiology and Prevention. Pharmacoepidemiology, 3(2), 208–222. View
Osemene, K. P., & Lamikanra, A. (2012). A study of the prevalence of self-medication practice among university students in southwestern Nigeria. Tropical Journal of Pharmaceutical Research, 11(4), 683–689. View
Wafula, F. N., Miriti, E. M., & Goodman, C. A. (2012). Examining characteristics, knowledge and regulatory practices of specialized drug shops in Sub-Saharan Africa: A systematic review of the literature. In BMC Health Services Research (Vol. 12, Number 1). View
Renschler, J. P., Walters, K. M., Newton, P. N., & Laxminarayan, R. (2015). Estimated under-five deaths associated with poor quality antimalarials in sub-Saharan Africa. The American Journal of Tropical Medicine and Hygiene, 92(6), 119–126. View
Mackey, T. K., & Liang, B. A. (2011). The global counterfeit drug trade: Patient safety and public health risks. Journal of Pharmaceutical Sciences, 100(11), 4571–4579. View