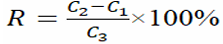Journal of CAM Research Progress Volume 1 (2023), Article ID: JCRP-109
https://doi.org/10.33790/jcrp1100109Research Article
Development and Validation of Residual Amount of Potential Genotoxic Substance 1-Bromo-2-Methylpropane in Febuxostat by GC
Miaomiao Zhang1,2, Mujun Zhang1,2,3*, Fanghua Shi1,2, Yao Deng1,2, Yi Pan1,2,4, and Huiping Zhao5
1Tianjin Institute of Pharmaceutical Research Tianjin 30000, China.
2Tianjin Taipu Pharmaceutical Tianjin 30000, China.
3Research Unit for Drug Metabolism, Chinese Academy of Medical Sciences, Tianjin 30000, China.
4State Key Laboratory of Drug Delivery and Pharmacokinetics, Tianjin Institute of Pharmaceutical Research, Tianjin 300301, People’s Republic of China.
5Teaching Hospital of Tianjin University of Traditional Chinese Medicine, China.
Corresponding Author: Mujun Zhang, Research Unit for Drug Metabolism, Chinese Academy of Medical Sciences, Tian jin 30000, China.
Received date: 13th September, 2023
Accepted date: 23rd September, 2023
Published date: 26th September, 2023
Citation: Zhang , M., Zhang, M., Shi, F., Deng, Y., Pan, Yi., and Zhao, H., (2023). Development and Validation of Residual Amount of Potential Genotoxic Substance 1-Bromo-2-Methylpropane in Febuxostat by GC. J CAM Res Progress, 2(1): 109.
Copyright: ©2023, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
In the present work, an effective and sensitive gas chromatography (GC) method for determination of the residual solvent 1-Bromo-2 Methylpropane in febuxostat was developed using an Agilent DB-624 column (30 m × 0.53 mm × 3.0 μm) and an optimized oven program. Its working range, linearity, the limit of detection (LOD) and the limit of quantification (LOQ), accuracy, precision, robustness, and stability were measured to verify the method. The LOD and the LOQ was 0.4725 μg/mL and 0.9450 μg/mL, respectively. The method has a wide working range of 0.9450 μg/mL (1.89ppm) ~ 21.6216μg/ mL (43.24ppm) for 1-Bromo-2-Methylpropane. And the method has a good linearity with the regression coefficient of the calibration graphs was 0.9998. The accuracy was determined by the recovery rate (80%, 100%, 120%) of three concentration levels, which were in the range of 96.04 to 102.93 %. Precision was expressed as a percentage of relative standard deviation (%RSD), which was 1.34% for determined 1-Bromo-2-Methylpropane. The results showed that the developed GC method had the great specificity, linearity, accuracy and precision, and applicable for the low-level detection of 1-Bromo-2-Methylpropane in febuxostat. This method can provide a new choice for GC determination of potential genotoxic impurities in febuxostat.
Keywords: Febuxostat;1-Bromo-2-Methylpropane; solvent; Genotoxic impurity; Method verification
Introduction
Gout is a metabolic disorder disease caused by the deposition of monosodium urate (MSU) crystals, which can lead to recurrent attacks of chronic inflammatory arthritis, urolithiasis, nephropathy, and acute arthritis and bursitis [1-5]. A variety of complications of gout, including cardiovascular disease, diabetes, obesity, and chronic kidney disease, have added s significant challenges for people with gout [6,7].
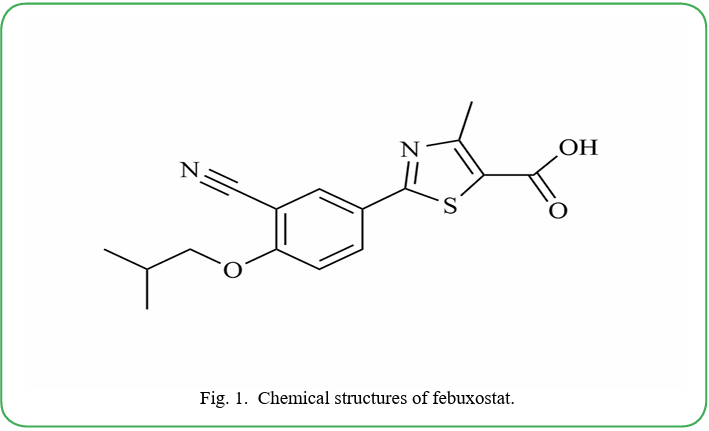
The model drug, febuxostat (Fig. 1) [chemical name: 2-(3-cyano- 4-isobutyloxy)-phenyl-4-methyl -5-thiazolecarboxylic acid], which is the first non-purine non-competitive xanthine oxidase inhibitor (XOI) approved by the European Union in 2008 [8,9]. It is the first drug approved by the United States Food and Drug Administration (FDA) in 2009 for the treatment of chronic gout caused by hyperuricemia [10,11]. Additionally, allopurinol is known to be associated with severe skin reactions and mortality [12]. Unlike allopurinol, febuxostat can significantly improve renal function, oxidative stress, and cardiac function without affecting other activities of purine metabolism [13-17]. Additionally, febuxostat has also been found to ameliorates testosterone-induced benign prostatic hyperplasia [18], and has antioxidant and anti-inflammatory effects [19,20], prevent acute lung injury triggered by uric acid-induced lung inflammation [21], and so on.
Residual solvents in pharmaceuticals refer to volatile organic compounds that are used in the production of active pharmaceutical ingredients (APIs), excipients and preparations, but cannot be completely removed during the process [22]. The residual solvents may affect the physical and chemical properties of APIs, and may pose a risk to human health due to its own potential toxicity or other adverse effects. Therefore, in order to meet the requirements of regulations and ensure the safety of patients, the determination of residual solvents has become an essential procedure for the quality control of APIs and drug products.
Toxicity is the main reason for the control of residual solvent contents, especially genotoxicity. In recent years, genotoxic impurities have attracted the attention of various regulatory agencies, such as the European Medicines Agency (EMA) and the United States Food and Drug Administration (US-FDA) [23,24]. International Council for Harmonization (ICH) also issued a separate guideline M7(R1), involving the limits of genotoxic impurities in newly marketed drugs in 2013 [25]. In the production process of febuxostat, the starting material used a genotoxic impurity with a halogenated aliphatic alkane warning structure: 1-Bromo-2-Methylpropane (Fig. 2). According to ICH M7, it can be classified as three types of genotoxic impurities.
Consequently, a highly sensitive GC method was established to identify and quantify the potential genotoxic impurity viz.1-bromo 2-methylpropane in febuxostat (Drug substance).
2. Experimental
2.1. Chemicals and reagents
The reference substance and samples of febuxostat used for method qualification were acquired from Taipu Pharmaceutical Co., Ltd. 1-Bromo-2-Methylpropane was purchased from Beijing Bailingwei Technology Co., Ltd. N, N-Dimethylformamide (DMF, for HPLC) was purchased from Merck.
2.2. Instrumentation and data acquisition
The system of GC was Agilent GC 7890 (SN: CN15343046) equipped with a flame ionization detector (FID) detector and Agilent 7697 (SN: CN15420001) headspace automatic sampler. Data acquisition and processing were performed using OpenLAB CDS® software (version C.01.07 SR3[465]).
2.3. Chromatographic conditions
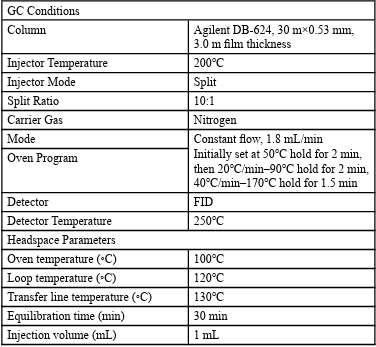
The chromatographic parameters of the GC method were as follows: High purity nitrogen was used as the carrier gas and the flow rate was 1.8 mL/min. The inlet temperature was 200°C and the split ratio was 10:1. The initial temperature of column oven was 50°C hold for 2 min and the temperature was raised to 90°C at a rate of 20°C /min hold for 2 min, then the temperature was raised to 170°C at a rate of 40°C /min hold for 1.5 min. Run for tens of minutes until no chromatographic peak appeared after 240°C high column flow. The FID temperature was 250°C. The equilibrium temperature of the headspace bottle, temperature of the quantitative ring and the transmission line were 100°C, 120°C, 130°C, respectively. The equilibrium time was 30 min and the headspace injection volume was 1 mL. In this work, DMF was used as the exclusive diluent for all the experiments. The concentration of sample was 500 mg/mL.
2.4. Control strategy
In the previous study, we referred to ICH M7 to determine the treatment cycle of febuxostat for 1-10 years, with a maximum acceptable intake (10μg/day) and the maximum daily dose of 80 mg. The limit of 1-Bromo-2-Methylpropane was determined to be 125 ppm by calculating the concentration. Nevertheless, we changed the control strategy because of we identified febuxostat as a long term drug in subsequent studies. Based on the duration of treatment and the maximum clinical dose, the limit level of 1-Bromo-2 Methylpropane needs to be controlled below 18 ppm to meet the threshold of toxicological concern (TTC) of 1.5 μg/day.
2.5. Preparation of sample and standard
About 1000 mg of febuxostat was dissolved with 2 mL DMF to prepare febuxostat sample solution. The solution of standard was prepared by dissolving 1-Bromo-2-Methylpropane in DMF (the concentration was about 9.0 μg/mL); 2.0 mL ofthis solution is equalto 18 ppm (w/w) of 1-Bromo-2-Methylpropane in 1000 mg febuxostat. For neat sample analysis, all solutions were prepared directly in a 10 mL GC headspace vial. After the solution was prepared, all the vials were capped and crimped immediately.
According to gas chromatography (‘Chinese Pharmacopoeia’ 2015 Edition IV Part 0521) determination.
2.6. Method validation
The method verification was carried out according to the guiding principles of methodological verification of ‘Chinese Pharmacopoeia’ 2015 Edition IV Part 9101.
The validation of our GC method was evaluated based on specificity, system suitability, LOD and LOQ, linearity, accuracy, precision, robustness and solution stability. The specificity of the method was assessed by injecting a blank dilute (DMF), standard solutions of 1-Bromo-2-Methylpropane, standard solutions of febuxostat and a specificity mixture solution (1-Bromo-2-Methylpropane was added to febuxostat sample solution at a concentration of 0.0018 % of its working concentration). The separation degree of 1-Bromo-2 Methylpropane and adjacent chromatographic peaks in the system suitability solution is required to meet the requirements (≥ 1.5), and the asymmetry factor of 1-Bromo-2-Methylpropane is between 0.95 and 1.05. The LOD was determined at a concentration of signal to noise (S/N) ratio ≥ 3 and the LOQ was determined at a concentration of S/N ≥ 10. The LOQ solution of six standard substances were continuously injected and the %RSD of peak area response was not more than 10 %. The standard curve of 1-bromo-2-methylpropane was drawn with concentration as abscissa and peak area as ordinate. The accuracy of the method was evaluated by calculating the spiked solutions at three concentration levels (80 %, 100 % and 120 %). The repeatability was evaluated by repeated injection of 100 % spiked solution for 6 times. The intermediate precision of the method was evaluated by the second analyst using different equipment, chromatographic columns, and solutions. The requirements for personal repeatability were the same except for the %RSD ≤ 11 % of the two analysts. Take 1mL of the solutions of standard and sample, inject into the GC by headspace injection, and analyze at 0h, 5h, 15h, 20h, 25h, 30h and 35h respectively. The changes of the peak area of 1-Bromo-2-Methylpropane in the standard solution and the sample solution within 35h were investigated. The acceptance standard for the stability solution was the %RSD (n=6) of the peak area of 1-Bromo-2-Methylpropane in the standard solution and the sample solution should not more than 6.0% within a certain time, and no impurities interfering with 1-Bromo-2-Methylpropane should be produced. Under the initial chromatographic conditions, the column flow rate was ± 0.1mL/min, the initial column temperature was ± 2°C, and the headspace heating temperature was ± 2°C for robustness test.
3. Results and Discussion
3.1. Method development
The purpose of this study is to establish a gas chromatography method with good selectivity, high tolerance and applicability for the potential genotoxic impurity 1-Bromo-2-Methylpropane. The details and key points of the method development process are discussed in turn below.
3.1.1. Chromatographic conditions
There are two programmed temperatures were set in the development process of GC method. We chose the second heating procedure for subsequent method development: the solution does not interfere with the determination of 1-Bromo-2-Methylpropane under this method. The chromatographic peak of the diluent did not appear at the end of the oven programming and were all blowout during post run. The oven program was optimized and determined with the total running time was about 9.5 min because of the need to remove the diluent (DMF) after the elution of the residual solvent, but the post-running time is increased.
3.1.2. Diluent
The diluent is a key factor affecting the headspace equilibration temperature, the sample peak response and sensitivity of GC method. DMF and N-Methyl pyrrolidone (NMP) were studied as potential diluents. The results showed that DMF provided the best baseline and did not interfere with the target peak. Therefore, DMF was selected as the diluent of the GC method.
3.1.3. Volume of diluent
3 mL and 2 mL of NMP interfered with the determination of 1-Bromo-2-Methylpropane, while 2 mL and 3 mL of DMF did not interfere with the determination of 1-Bromo-2-Methylpropane during the development process. Comprehensive consideration, the volume of the diluent was selected to be 2 mL.
3.1.4. Concentration of sample
Appropriate amount of 1-Bromo-2-Methylpropane was precisely measured, dissolved with DMF and quantitatively diluted to a con centration of S/N ratio close to 10.0. The LOQ of 1-Bromo-2-Meth ylpropane was 0.9450 μg/mL, which it can meet the sensitivity re quirements of the method. The concentration of febuxostat sample was 500 mg/mL was selected in the present study because of the limit of 1-Bromo-2-Methylpropane is 18 ppm.
Using the above strategy, a new GC method for the determination of the residual amount of the potential genotoxic substance 1-Bromo 2-Methylpropane in febuxostat was successfully developed. GC chromatographic conditions and headspace parameters of the final method are summarized in Table 1.
3.2. Method validation
3.2.1. Specificity
Specificity refers to the ability of the analytical method to accurately determine the analyte in the presence of some impurities, degradation products, excipients and other components. DMF did not interfere with the determination of 1-Bromo-2-Methylpropane and the resolution of 1-Bromo-2-Methylpropane in the specificity mixture solution was 16.21 (≥ 1.5) under the selected chromatographic conditions.
3.2.2. System Suitability
The system suitability test in the methodology validation is to examine the instrument and equipment, test operation and the analyzed sample as a whole. The main purpose is to ensure that the chromatographic system used in the analysis is effective and applicable. Under the selected chromatographic conditions, the resolution of 1-Bromo-2-Methylpropane in the solution of system suitability was 16.44 (≥ 1.5), and the asymmetry factor of 1-Bromo 2-Methylpropane in the solution of system suitability was 0.99, between 0.95 and 1.05. All the parameters were indicated that the established chromatographic system was applicable for the analysis of 1-Bromo-2-Methylpropane in febuxostat.
3.2.3. Limits of detection and quantitation
Under the selected chromatographic conditions, the concentration of LOD of 1-Bromo-2-Methylpropane was 0.4725 μg/mL. The RSD (n = 2) of the retention time of 1-Bromo-2-Methylpropane in 2 copies solutions of LOD was 0% (< 1.0%), and the two S/N were not less than 3.0. The RSD (n = 6) of the retention time of 1-Bromo-2-Methylpropane in 6 parts of the solution of LOQ was 0.01 % (< 1.0%), and the RSD (n = 6) of the peak area of 1-Bromo 2-Methylpropane was 2.0% (< 8.0%). The six S/N were not less than 10.0.
3.2.4. Linearity
The linear equation of 1-Bromo-2-Methylpropane was: Y = 2.7917X + 0.2122, the correlation coefficient (R) was 0.9998, the absolute value of Y axis intercept was less than 25 % of 100 % response, and the % RSD of response factor is 1.49 % (< 6.0%).
3.2.5. Accuracy
The accuracy of the method was expressed by spiked recovery. The spiked recovery was calculated according to the following formula:
Among them, R was the spike recovery; c1 was the initial concentration of the sample; c2 was the measured concentration of the spiked sample; c3 was the spiked amount.
The recovery rate of 80 % ~ 120 % limit concentration of 1-Bromo- 2-Methylpropane in the solution of 9 recovery rates were in the range of 80 % ~ 115 %, 9 recovery data RSD (n = 9) was 2.03% (< 6.0%).
3.2.5. Precision
The precision of the method was evaluated by repeatability and intermediate precision. The recoveries of 1-Bromo-2-Methylpropane in 6 repeated solutions were in the range of 80 % ~ 115 %, and the %RSD (n = 6) of 6 recoveries was 0.91 % (< 6.0%). The recovery rates of 1-Bromo-2-Methylpropane in 12 precision solutions were in the range of 80 % ~ 115 %, and the %RSD (n = 12) of 12 recovery rates was 1.34 % (< 11.0%).
3.2.6. Robustness
The robustness of the method was appraised by slightly changing the chromatographic conditions of GC and headspace parameters such as the column flow rate was ± 0.1mL/min, the initial column temperature was ± 2°C, and the headspace heating temperature was ± 2°C. The robustness of the analysis method was compared with the results under conventional method conditions under each variable condition. Under each slightly changed method conditions, the %RSD (n = 6) of the six 1-Bromo-2-Methylpropane contents in the sample solutions were less than 6.0%. These study results established that the test method is robust, the key instrument parameters fluctuate slightly, and the performance of the method will not be affected.
3.2.7. Solution stability
The standard solution and sample solution were stable within 35 h under conventional laboratory storage conditions. Within 35 hours, the RSD (n = 6) of the peak area of 1-Bromo-2-Methylpropane in the standard solution was 1.68 % (< 6.0%), and no impurities interfering with 1-Bromo-2-Methylpropane were produced. 1-Bromo-2-Methylpropane was not detected in the sample solution, and no impurities interfering with 1-Bromo-2-Methylpropane were produced.
4. Discussion
The method was fully validated, indicating that it can be successfully applied to the determination of the residual solvent 1-Bromo-2-Methylpropane in febuxostat. The method has the advantages of simple operation, high sensitivity and low cost, which provides convenience for further determination of the residual solvent 1-Bromo-2-Methylpropane in febuxostat.
Competing interest:
The author declares that they have no competing interests.
References
Mackenzie I. S., Ford I., Nuki G., Hallas Je., Hawkey C. J., Webster J., Ralston S. H. (2020). Long-term cardiovascular safety of febuxostat compared with allopurinol in patients with gout (FAST): a multicentre, prospective, randomised, open label, non-inferiority trial. Lancet, 396(28), 1745-1757.View
Al-Abdouh A., Khan S. U., Barbarawi M., Upadhrasta Si., Munira S., Bizanti A., Elias H. (2020). Effects of Febuxostat on Mortality and Cardiovascular Outcomes: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Mayo Clinic Proceedings, 4(4), 434-442.View
Doherty M. (2009). New insights into the epidemiology of gout. Rheumatology, 48, i2-ii8.View
Alexander S. (2008). Developments in the scientific and clinical understanding of gout. Arthritis Research & Therapy, 10, 221.View
Gim G. T., Raj N., Kenneth G. S. (2006). Pathophysiology, Clinical Presentation and Treatment of Gout. Drugs, 66 (12), 1547-1563.View
Lin S. X., Zhang T., Zhu L. X., Pang K., Lu S. S., Liao X., Ying S. H. (2021). Characteristic dysbiosis in gout and the impact of a uric acid-lowering treatment, febuxostat on the gut microbiota. Journal of Genetics and Genomics, 48, 781-791.
Sun R. X., Lu J., Li H., Cheng X. Y., Xin Y., Li C. G. (2020). Evaluation of febuxostat initiation during an acute gout attack: A prospective, randomized clinical trial. Joint Bone Spine, 87, 461-466.View
Asmaa M. A., Nashwa F. G. E., Medhat A. S. A., Mostafa M. M., Ahmed I. A., Yasmine F. I. (2020). Amelioration of testosterone induced benign prostatic hyperplasia using febuxostat in rats: The role of VEGF/TGFβ and iNOS/COX-2. European Journal of Pharmacology, 889, 173631.View
Rashad A. Y., Daabees H. G., Elagawany M., Shahin M., Abdel Moneim A. E., Rostom S. A. (2023). Towards the Development of Dual Hypouricemic and Anti-inflammatory Candidates: Design, Synthesis, Stability Studies and Biological Evaluation of Some Mutual Ester Prodrugs of Febuxostat-NSAIDs. Bioorganic Chemistry, 135, 106502.View
Kang Y. L., Gu J. M., Hu X. R. (2017). Syntheses, structure characterization and dissolution of two novel cocrystals of febuxostat. Journal of Molecular Structure, 1130, 480-486.View
Burns C. M., Wortmann R. L. (2011). Gout therapeutics: new drugs for an old disease. Lancet, 377, 165-177.View
Liu C. W., Chang W. C., Lee C. C., Shau W. Y., Hsu F. S., Wang M. L., Chen T. C. (2019). The net clinical benefits of febuxostat versus allopurinol in patients with gout or asymptomatic hyperuricemia e A systematic review and meta-analysis. Nutrition, Metabolism & Cardiovascular Diseases, 29, 1011 1022.View
Wu Y. L., Mao Z. S., Liu Y. P., Wang X., Di X. (2015). Simultaneous determination of febuxostat and its three active metabolites in human plasma by liquid chromatography–tandem mass spectrometry and its application to a pharmacokinetic study in Chinese healthy volunteers. Journal of Pharmaceutical and Biomedical Analysis, 114, 216-221.
Faruque L. I., Ehteshami-Afshar A., Wiebe N., Tjosvold L., Homik J., Tonelli M. (2013). A systematic review and meta-analysis on the safety and efficacy of febuxostat versus allopurinol in chronic gout. Seminars in Arthritis and Rheumatism, 43, 367-375.View
Terkeltaub R. (2010). Update on gout: new therapeutic strategies and options. Nature Reviews Rheumatology, 6, 30-38.View
Chohan S., Becker M. A. (2009). Update on emerging urate lowering therapies. Current Opinion In Rheumatology, 21, 143 149.View
Edwards N. L. (2009). Febuxostat: a new treatment for hyperuricaemia in gout. Rheumatology, 48, ii15-ii19.View
Abo-Youssef A. M., Afify H., Azouz A. A., Abdel-Rahman H. M., Abdel-Naim A. B., Allam S. (2020). Febuxostat attenuates testosterone-induced benign prostatic hyperplasia in rats via inhibiting JAK/STAT axis. Life Sciences, 260, 118414.View
Amirshahrokhi K. (2019). Febuxostat attenuates ulcerative colitis by the inhibition of NF-κB, proinflammatory cytokines, and oxidative stress in mice. International Immunopharmacology, 76, 105884.View
Ahmed M. A., Morsy E.M., Ahmed A. A. (2019). Protective effects of febuxostat against paraquat-induced lung toxicity in rats: Impact on RAGE/PI3K/Akt pathway and downstream inflammatory cascades. Life Sciences, 221, 56-64.View
Maresh M.M., Abdelaziz R. R., Ibrahim T. M. (2020). Febuxostat mitigates concanavalin A-induced acute liver injury via modulation of MCP-1, IL-1β, TNF-α, neutrophil infiltration, and apoptosis in mice. Life Sciences, 260, 118307.View
ICH (International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use). (2021). Impurities: Guideline for residual solvents, Q3C (R8).View
European Medicines Agency. (2007). Evaluation of medicines for human use, in: Guideline on the Limits of Genotoxic Impurities.View
U.S. Department of Health and Human Services, Food and Drug Administration. (2008). Genotoxic and Carcinogenic Impurities in Drug Substances and Products: Recommended Approaches.View
ICH (International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use). (2013). Assessment and control of DNA reactive (mutagenic) impurities in pharmaceuticals to limit potential carcinogenic risk (draft), M7.View