Journal of Rehabilitation Practices and Research Volume 1 (2020), Article ID: JRPR-116
https://doi.org/10.33790/jrpr1100116Research Article
Subluxed Shoulders Need “TLC”: A Pilot Study of T-L-C Elastic Taping to Address Hemiplegic Shoulder Pain, Passive Range of Motion, and Sub-luxation
Sara J. Stephenson, OTD, OTR/L, BCPR, CBIS, CSRS1,2*, Steven M. Frey, OTR/L, CKTP1, Mark P. Stumpf, OTD, OTR/L, CKTP2, Jacob D. Wright, OTD, OTR/L, CKTP2 , Lauren B. Loges, OTD, OTR/L2, John L. Wong, OTD, OTR/L2
1Department of Rehabilitation Services, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, Phoenix, Arizona, United States.
2Department of Occupational Therapy, Northern Arizona University, Phoenix Biomedical Campus, Phoenix, Arizona, United States.
Corresponding Author Details: Sara J. Stephenson, OTD, OTR/L, BCPR, CBIS, CSRS, Department of Rehabilitation Services, Barrow Neurological Institute, St. Joseph’s Hospital and Medical Center, 350 W. Thomas Rd., Phoenix, AZ 85013, United States. E-mail: Neuropub@barrowneuro.org
Received date: 11th March, 2021
Accepted date: 19th March, 2021
Published date: 21th March, 2021
Citation:Stephenson, S.J., Frey, S.M., Stumpf, M.P., Wright, J.D., Loges, L.B., & Wong, J.L. (2021). Subluxed Shoulders Need “TLC”: A Pilot Study of T-L-C Elastic Taping to Address Hemiplegic Shoulder Pain, Passive Range of Motion, and Subluxation. J Rehab Pract Res, 2(1):116.
Copyright: ©2021, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Shoulder subluxation, abnormal tone, and hemiplegic shoulder pain are common sequelae for patients with neurological conditions. Therapists have used elastic taping to treat these consequential conditions in rehabilitation settings with inconsistent techniques and results. The use of elastic tape to treat shoulder subluxation, abnormal tone, and hemiplegic shoulder pain may result in increased participation in activities of daily living and functional activities; however, evidence-based approaches to guide taping are still emerging. An elastic taping method called the "T-L-C" method has been developed to establish a consistent approach for the assessment and taping to ensure patients are being treated with evidence-based techniques.
Running Title: Elastic taping for hemiplegic shoulder pain
Keywords: Activities of daily living; biomechanical; elastic tape; hemiplegia; hypertonic; hypotonic; muscle tone; occupational therapy; tension
Abbreviations
ADL, activities of daily living; HSP, hemiplegic shoulder pain; PI, primary investigator; PROM, passive range of motion; ROM, range of motion; VAS, visual analog scale
The onset of a neurological condition that induces hemiplegia may result in various impairments that occupational and physical therapists frequently evaluate and address in a therapy or rehabilitation setting. In particular, reduced range of motion (ROM), shoulder subluxation, and neuromuscular-associated hemiplegic shoulder pain (HSP) are prevalent sequelae following a neurological event that results in hemiplegia. ROM limitations may co-occur with subluxation and HSP and interfere with functional use of the upper extremity in activities such as bathing and dressing [1,2]. Khan and colleagues[3] found the incidence of shoulder subluxation after a neurological event (stroke) of 17-81%. A study in the American Journal of Physical Medicine & Rehabilitation reported that the incidence of shoulder pain is approximately 55% in patients with hemiplegia following the onset of a neurological event such as stroke [4]. Pain is a barrier to rehabilitation efforts and is associated with increased length of hospital stay, reduced quality of life scores, and poor outcomes [5,6]. HSP, coupled with subluxation, reduces arm mobility and ROM and results in reduced independence in activities of daily living (ADLs) such as bathing, dressing, and hygiene and can negatively impact a patient’s quality of life [7,8].
Therapists use various techniques and tools to address HSP and subluxation, including elastic taping. Currently, gaps and inadequacies exist in clinical practice guidelines, evidence-based assessments, and protocols for elastic taping to reduce shoulder symptoms resulting from HSP and subluxation, often due to imbalanced tone. Overall, the literature on elastic taping is ambiguous regarding tension and technique and is inconsistent regarding effectiveness overall. Some clinical efficacy studies and systematic reviews do not support elastic taping for HSP and subluxation [9, 10][9,10]. In contrast, some efficacy studies and systematic reviews support the use of elastic taping 11]-14]. The inconsistent and conflicting literature presents a challenge for OTs to navigate, and therapists must blend the available literature with clinical practice knowledge and experience and apply clinical reasoning to their taping practices.
The available literature to guide elastic taping, which is a combination of direction and tension, includes numerous studies of tape approaches focusing on the mechanical correction of shoulder deficiencies. Although a biomechanical approach to mechanical correction is a key component of elastic taping, overall muscle tone is a key consideration [11,15]. The subject of muscle tone management, which is part of hemiplegia interventions and is a frequently discussed topic in neurological rehabilitation, is not formally addressed in taping research [16-19]. Whether elastic taping is effective is a challenging topic for researchers to study because many factors influence its effectiveness. Tone imbalance as a basis for subluxation and HSP are important foci for therapists to address and can be managed using elastic taping [20,21].
The T-L-C method of tone correction was developed by the primary investigator (S. M. Frey) to address tone management in shoulder subluxation and HSP using elastic taping. The T-L-C method is unique because it combines a biomechanical correction with directional taping that applies tension to manage shoulder tone [21]. The first premise of the T-L-C method is to tape the “T” (tight or hypertonic) shoulder muscles to inhibit their tone; the second is to tape the “L” (loose or hypotonic) shoulder muscles to facilitate their tone. Finally, with the tone addressed, the tape is applied to provide a mechanical correction (“C”) of the subluxation. The basis of taping is that tape applied on the skin along the length of the muscle stretches the skin and fascia, which could affect the muscle fibers below to respond by either inhibiting or facilitating muscle tone [21]. Researchers posit that this effect may be achieved through directional taping with tension on the skin along muscle lengths to provide sensory feedback to the proprioception systems that interact with the skin, fascia, and muscle spindles to facilitate balanced muscle tone. We surmise that taping from insertion to origin along hypertonic muscles provides a reduction in tone. We surmise that taping from origin to insertion along hypotonic muscles facilitates an increase in tone. In this pilot study, we sought to determine if short-term tone management via directional elastic taping and mechanical correction taping impacted HSP, subluxation, and ROM.
Methods
Research Design
This pilot study is a quasi-experimental, AB design with 10 participants. Each participant served as his or her control. Baseline (A) was the control period. During this phase, the amount of subluxation, passive range of motion (PROM) end range due to pain, active range of motion (AROM), and self-reported pain at rest with PROM and AROM were measured within 30 minutes of the implementation of the T-L-C intervention. Clinical observations regarding scapula placement, scapula symmetry, posture, and scapula-humeral rhythm were recorded. The intervention (B) consisted of applying elastic tape to the shoulder musculature based on an assessment of shoulder tone (Table 1) and subluxation. The elastic tape was worn for a total of 3 days as part of the study. Measurements for subluxation, PROM, AROM, and pain were collected immediately after taping, 1 day after taping, and 3 days after taping.
The study was approved by the institutional review board of the primary investigator’s institution. The primary author’s secondary institution ceded approval to the primary institution. The study followed the protocol and standards set forth by the boards.
Participant Selection
Participants were recruited over an 18-month period by occu-pational therapists who identified potential participants with the onset of symptoms <3 months before the study start. The primary investigator (PI) completed the screening to determine eligibility. The participants were screened via electronic health record review and clinical observation by the PI to determine if she or he met the inclusion criteria. The inclusion criteria were (1) current shoulder pain and/or shoulder subluxation, of any amount, as a result of a neurological diagnosis, (2) 18 or more years of age, (3) currently a patient in the rehabilitation unit, and (4) the ability to appropriately communicate pain levels on a visual analog scale (VAS). Exclusion criteria included (1) prior history of allergy to adhesives including elastic tape, (2) bilateral shoulder subluxation, (3) active cancer in the taping area, (4) poor skin integrity, (5) loose or sagging skin, (6) inability to communicate pain levels, (7) current deep vein thrombosis in the taping area, (8) congestive heart failure, and (9) renal insufficiency. Participants were patients in the rehabilitation unit and were receiving concurrent therapy services as part of the routine standard of care. No data were collected to specify the type or amount of therapy services provided. Eligible participants were provided written and verbal information as part of the consent process requirements before enrollment and were provided copies of signed consent forms when they enrolled in the study.
Instruments and Outcome Measures
Before the assessment, participants were positioned upright with their feet on the floor and their arms symmetrically on their laps. The shoulder assessment protocol included measurements for shoulder subluxation, pain, PROM, AROM, observation of scapular placement, and tone (hypertonic, hypotonic, or normal) for the shoulder musculature, including the pectoralis, biceps, triceps, upper trapezius, levator scapulae, rhomboids, teres major, latissimus dorsi, infraspinatus, and deltoids.
Shoulder Subluxation
Anthropometric measurements were used to determine shoulder subluxation (in centimeters) and range of motion (in degrees). Shoulder subluxation was evaluated using a caliper to measure the distance from the inferior aspect of the acromion to the superior aspect of the humeral head, with the hemiparetic extremity hanging unsupported at the participant’s side. This measurement was selected to ensure consistency between potential raters should the PI be unable to continue in the study. Although radiographic imaging is the more commonly used assessment tool for measuring subluxation, its cost was prohibitive [22]. Finger-breadth palpation has demonstrated some reliability; however, this method was not selected in order to establish a consistent clinical measurement in the unlikely event that another researcher would have to assume measurements if the PI became unable to complete the study [23].
Pain
Using the rehabilitation unit's established visual analog scale (VAS), data on self-reported pain at rest, PROM, and AROM were collected four times: (1) before taping; (2) immediately after taping, at the pretaping PROM end-range score due to pain; (3) 1 day after taping, at the pretaping PROM end-range score due to pain; and (4) 3 days after taping, at the pretaping PROM end-range score due to pain. With the VAS, participants indicate their pain level on a scale of zero (left side of the scale), indicating no pain, to 10 (right side of the scale), indicating severe pain [24].
Range of Motion
PROM of shoulder flexion was assessed using goniometric measurement [25]. The PI moved the upper extremity through PROM of shoulder flexion to the end range on the basis of the participant’s pain tolerance. Data were collected four times, as indicated above. AROM was assessed using goniometric measurement for shoulder flexion [26].
Scapular Placement
The position of the scapula on the affected side was compared to that of the unaffected side and was recorded as normal, winging, abducted, or adducted, and an assessment of scapulohumeral rhythm was completed concurrently with ROM. These data were collected for analysis before taping only.
Tone
The PI recorded tone as hypotonic, hypertonic, or normal for the following musculature: pectoralis, biceps, triceps, upper trapezius, levator scapulae, rhomboids, teres major, latissimus dorsi, infraspinatus, and deltoids. The PI used checkmarks to record the tone for the musculature, and pretaping tone data are presented in Table 1. Data were collected for analysis before taping only.
Intervention
Interventions were completed by the PI, and intervention fide-lity was established by having the PI perform all of the pretape assessments; perform reassessments immediately after taping, at day 1, and at day 3; and complete all taping intervention. Participants were seated in an upright and midline sitting position either in a wheelchair or on a firm surface with bilateral upper extremities initially in the participant’s lap symmetrically or unsupported at his/her side temporarily for the assessment portion. Following the assessment and before taping, the participant’s skin was cleaned with an alcohol wipe to remove oils for good tape adherence. Hypertonic muscles were taped first from the muscle’s insertion point to origin using tension range for inhibition [27-29]. Next, hypotonic muscles were taped from origin to insertion using tension range for facilitation [27-29]. Finally, mechanical corrective taping was performed to provide enough force from stretch to adequately correct scapular alignment and approximate the humeral head into the glenohumeral fossa using tension range for mechanical correction [27,29]. The tape was applied following the initial shoulder assessment and remained on the participant for 3 consecutive days.
Data Collection
Measurements were taken by the PI, who was not blinded to the study participants or purpose. Measurements were taken four times for pain, PROM, AROM, and subluxation, before taping, immediately after taping, 1 day after taping, and 3 days after taping. After 3 days of taping, no further measurements were taken. Day 3 ended the study, and the participants could continue with shoulder taping at the discretion of their primary OT.
Data are presented as mean (standard deviation) or median (interquartile range [IQR]). Charts are based on the medians of the measurements due to the skewness of the data. Wilcoxon signed-rank tests were used to compare the changes in PROM, pain, and subluxation between before and after taping to assess immediate relief and then from before taping to day 3 after taping to assess reversion. An alpha of 0.05 (two-tailed) was used as the criterion for statistical significance. SPSS ver 26 (IBM Corp, Armonk, NY) was used for data analysis.
Results
Participant Demographics
Eleven participants were enrolled in the study, three men and eight women. One participant had the shoulder tape inadvertently removed during a shower and was removed from the study. The 10 participants ranged in age from 27-73 years (mean = 51.8 years, SD = 18.8 years). Six right and four left upper extremities were taped. All participants had an onset of symptoms less than 3 months before the study start. The broad diagnosis categories included stroke (5), brain tumor (2), spinal cord injury (2), and central nervous system lymphoma (1). Participants received concurrent interventions from an occupational therapist and other rehabilitation team members during the study. Table 1 includes highlights of participants’ upper extremity characteristics.
Active Range of Motion Shoulder Flexion
Participants’ mean (SD) end-range AROM before taping was 1.6 (2.7) degrees, and immediately after taping, it was 3.9 (4.0) degrees. The day 1 mean was 4.4 (4.4) degrees, and the day 3 mean was 5.4 (4.9) degrees (Table 1). The differences between measurements before and after taping were not significant (all p≥0.05).
Passive Range of Motion Shoulder Flexion
Participants’ median end-range PROM before taping was 75 (IQR: 65, 100) degrees. PROM increased to 105 (IQR: 100, 122.5) degrees immediately after taping (p=0.005). The day 3 median of 125 (IQR: 107.5, 132.5) degrees remained significantly greater than the pre-taping measurement, p=0.005 (Figure 1).
Pain Level During Passive Range of Motion of Shoulder Flexion
Participants’ median pain before taping was 5.5 (IQR: 4.0, 8.25), and immediately after taping, it was 0.0 (IQR: 0.0, 0.0) (p=0.07). The day 3 median was 0.0 (IQR: 0.0, 0.0) (p=0.07) (Figure 2)
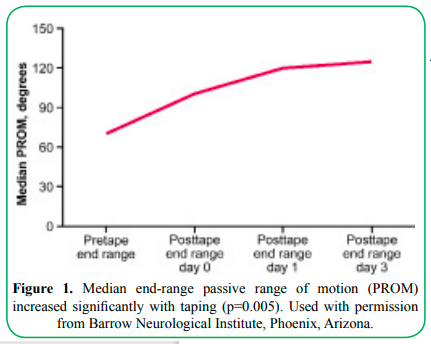
Figure 1. Median end-range passive range of motion (PROM) increased significantly with taping (p=0.005). Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
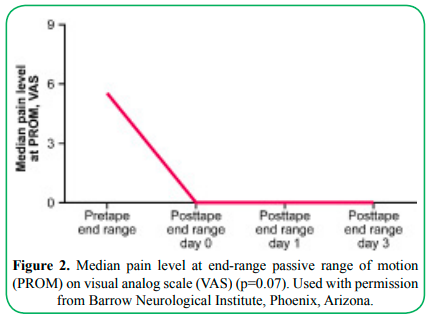
Figure 2. Median pain level at end-range passive range of motion (PROM) on visual analog scale (VAS) (p=0.07). Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
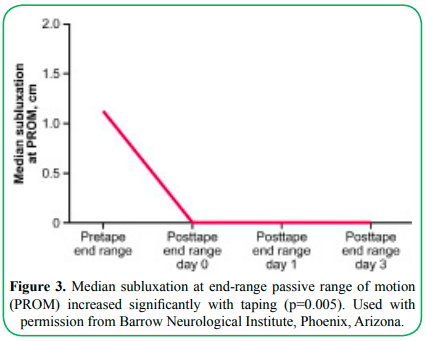
Figure 3. Median subluxation at end-range passive range of motion (PROM) increased significantly with taping (p=0.005). Used with permission from Barrow Neurological Institute, Phoenix, Arizona.
Subluxation
The median pretaping subluxation score was 1.1 (IQR: 0.5, 3.0) cm. This score decreased to 0.0 (IQR: 0.0, 0.0) immediately after taping (p=0.005) and remained at 0.0 (IQR: 0.0, 0.25) at day 3, p=0.005 (Figure 3). Participants 2 and 5 had a slight increase in subluxation on day 3. However, their scores remained better than their pretaping subluxation scores. These participants presented with dense hemiplegia, and the investigators surmised that the weight of the upper extremity could have overcome the effects of the tape.
Discussion
This pilot study indicates that the clinician-developed T-L-C elastic taping method demonstrated significantly reduced subluxation and increased shoulder flexion PROM in the participants tested. The median end-range PROM pretape to posttape scores and subluxation median pretape to posttape scores differed significantly. Participants 2 and 5 had a slight increase in subluxation on day 3 but nowhere near the pretaping subluxation scores. These participants presented with significant hemiplegia and large upper extremities. This slight increase in subluxation could be due to the weight of the extremity overcoming the effects of the tape. This information is useful for informing clinical practice, indicating that the tape should be changed sooner than 3 days for patients with significant hemiplegia and a large upper extremity. Given the small sample size, the T-L-C method did not reach statistical significance for improving AROM. We did not identify any clinical significance from the data for AROM.
Although changes in participant’s VAS pain scores did not meet the threshold of statistical significance, we regard these changes to be highly clinically significant because a reduction in pain score has a positive and measurable effect on client well-being and engagement in the rehabilitation process. This information suggests that following the T-L-C method of shoulder assessment and taping evaluation for patients with neurological issues can provide an effective intervention that impacts symptoms and positively influences the client’s ability to actively participate in future occupational therapy sessions.
Limitations
Limitations include small sample size, limited effect size, no control group, participants not being blinded to the intervention, and the use of a convenience sample. Although the study lends itself to practice-oriented implementation and interventions, it poses issues with external validity. We are unable to generalize to a larger population and are unable to compare intervention groups. Potential sampling bias could have occurred because other occupational therapists referred clients to the PI for consideration in the study. Observer bias cannot be ruled out because the outcome measurements were all performed by the PI, although a strict study protocol was followed. The use of the VAS with responses given verbally rather than marking pain levels on a line impacts the VAS scoring. Marking the VAS was considered for the study protocol, but we elected to use a verbal indication instead. The VAS is consistently used in the rehabilitation unit with a verbal report, and participants were familiar with this process.
Future studies should include a larger sample size and comparison of T-L-C to other techniques. The content validity and interrater reliability should be established for the T-L-C method to ensure the reliability of the results.
Implications for Practice
• Therapists with experience using elastic tape can use this method to assess the neurologically impaired shoulder and to guide taping
• Using the T-L-C method and taping technique could extend the treatment impact depending on client reaction and staff adherence
• A focus on the proximal stability of the shoulder in neurology-based rehabilitation is important, and the T-L-C method serves as an adjunct that can be applied in conjunction with other interventions
• Participant subluxation was decreased along with an increase in PROM, allowing continued participation in neuromuscular shoulder re-education when using the T-L-C method
• Patient-reported decreased pain and reduced hypertonicity could reduce the use of analgesics and potentially lead to improved participation in the therapy process
• Improved posture and proper shoulder alignment (scapular symmetry and humeral head alignment) reduces the risk of future microtrauma
Researchers have highlighted that this method can begin to close the gap between knowledge and evidence-based practice. This pilot study suggests that T-L-C is a promising method for an adjunctive therapy to address HSP, PROM limitations, and subluxation. Exploring methods of evaluating current intervention practices such as T-L-C is important to give clinicians evidence-based tools that can be easily applied to practice. Although clinic-based research poses numerous challenges, therapists need to continue exploring translational rehabilitation research such as the T-L-C method and incorporating it into clinical care practices to improve patient outcomes.
Disclosures:
The authors have no personal, financial, or institutional interest in any of the drugs, materials, or devices described in this manuscript. Preliminary pain and range of motion findings were presented at the American Occupational Therapy Association Conference, New Orleans, Louisiana, April 4-7, 2019.
Financial Support:
None
Acknowledgments:
We thank Lawrence Greene, PhD, for writing guidance, and Luis Escalante, DPT, and Kristin Palmiscno, OTD, OTR/L, for support in consenting participants. We thank Curt Bay, PhD, for assistance with the statistical analyses. We thank the staff of Neuroscience Publications at Barrow Neurological Institute for assistance with manuscript preparation.
Competing interests:
The authors of this paper declare no competing interests.
References
Griffin, C. (2014). Management of the hemiplegic shoulder complex. Top Stroke Rehabil, 21(4):316-318.
Hardwick, D.D., & Lang, C.E. (2011). Scapular and humeral movement patterns of people with stroke during range-of-motion exercises. J Neurol Phys Ther. Mar, 35(1):18-25.View
Khan, N., Khan, S.A., Ahmed, N., Akram, M.J., & Mehboob, S. (2020). Frequency of shoulder subluxation among stroke patients. Rawal Medical Journal, 45(1):74-76.View
Kalichman, L., & Ratmansky, M. (2011). Underlying pathology and associated factors of hemiplegic shoulder pain. Am J Phys Med Rehabil, 90(9):768-780.View
Lindgren, I., Gard, G., & Brogardh, C. (2018). Shoulder pain after stroke - experiences, consequences in daily life and effects of interventions: a qualitative study. Disabil Rehabil, 40(10):1176-1182.View
Turner-Stokes, L., & Jackson, D. (2002). Shoulder pain after stroke: a review of the evidence base to inform the development of an integrated care pathway. Clin Rehabil, 16(3):276-298.View
Wolbrecht, E.T., Rowe, J.B., & Chan, V., Ingemanson, M.L., Cramer, S.C., Reinkensmeyer, D.J. (2018). Finger strength, individuation, and their interaction: Relationship to hand function and corticospinal tract injury after stroke. Clin Neurophysiol, 129(4):797-808.View
Hung, C.S., Hsieh, Y.W., Wu, C.Y., et al. (2019). Hybrid rehabilitation therapies on upper-limb function and goal attainment in chronic stroke. OTJR (Thorofare N J), 39(2):116- 123.View
Mason, H., Vandoni, M., Debarbieri, G., Codrons, E., Ugargol, V., & Bernardi, L. (2013). Cardiovascular and respiratory effect of yogic slow breathing in the yoga beginner: what is the best approach? Evidence-Based Complementary and Alternative Medicine, 2013.View
Wang, Y., Kondo, T., Suzukamo, Y., Oouchida, Y., & Izumi, S. I. (2010). Vagal nerve regulation is essential for the increase in gastric motility in response to mild exercise. The Tohoku Journal of Experimental Medicine, 222(2), 155-163.View
Becker, E.W. (2007). Micro-algae as a source of protein. 25(2), 207-210. https://doi.org/10.1016/j.biotechadv.2006.11.002View
Kok, B. E., Coffey, K. A., Cohn, M. A., Catalino, L. I., Vacharkulksemsuk, T., Algoe, S. B.,& Fredrickson, B. L. (2013). How positive emotions build physical health: Perceived positive social connections account for the upward spiral between positive emotions and vagal tone. Psychological Science, 24(7), 1123-1132.View
Scott, F. J., Baron-Cohen, S., Bolton, P., & Brayne, C (2002). The CAST (Childhood Asperger Syndrome Test): Preliminary development of a UK screen for mainstream primary-school age children. Autism, 6, 9-31. View
Gibson, J. J., & Pick, A. D. (1963). Perception of another person's looking behavior. The American Journal of Psychology, 76(3), 386-394.View
Dawson, G., & Adams, A. (1984). Imitation and social responsiveness in autistic children. Journal of Abnormal Child Psychology, 12(2), 209-226.View
Stone, W. L., Lemanek, K. L., Fishel, P. T., Fernandez, M. C., & Altemeier, W. A. (1990). Play and imitation skills in the diagnosis of autism in young children. Pediatrics, 86(2), 267- 272.View
Stahmer, A. C., & Schreibman, L. (1992). Teaching children with autism appropriate play in unsupervised environments using a self-management treatment package. Journal of Applied Behavior Analysis, 25(2), 447-459.View
Zak, P., & Barraza, J. (2018, May 5) Immersion neuroscience platform. Retrieved from https://www.immersionneuro.com/ our-team/
Norris, C. J., Larsen, J. T., & Cacioppo, J. T. (2007). Neuroticism is associated with larger and more prolonged electrodermal responses to emotionally evocative pictures. Psychophysiology, 44(5), 823–826.View
Barraza, J. A., Alexander, V., Beavin, L. E., Terris, E. T., & Zak, P. J. (2015). The heart of the story: Peripheral physiology during narrative exposure predicts charitable giving. Biological Psychology, 105, 138-143.View
S, Baron-Cohen., S, Wheelwright., J, Hill., Y, Raste., I, Plumb.,(2001). The "Reading the Mind in the Eyes" Test revised version: a study with normal adults, and adults with Asperger syndrome or high-functioning autism. J Child Psychol Psychiatry, 42(2):241-51.View
Kooij, D. T. A. M., Bal, P. M., & Kanfer, R. (2014). Future time perspective and promotion focus as determinants of intraindividual change in work motivation. Psychology and Aging, 29(2), 319–328. https://doi.org/10.1037/a0036768View
A, Kushki.,E, Drumm., Michele, P. M., Nadia, T.,Annie, D.,Tom, C., Evdokia, A., (2013). Investigating the Autonomic Nervous System Response to Anxiety in Children with Autism Spectrum Disorders. https://doi.org/10.1371/journal.pone.0059730 View
Guo-li, M., Hongjun, S., (2005). Adult neurogenesis in the mammalian central nervous system. Annu Rev Neurosci, 28:223-50. doi: 10.1146/annurev.neuro.28.051804.101459.View
Michelle, A. P., Angela, S., Bruce, H. F., Stephen, W. P., (2013). Respiratory sinus arrhythmia: A marker for positive social functioning and receptive language skills in children with autism spectrum disorders. https://doi.org/10.1002/dev.21002View
Kok, B. E., & Fredrickson, B. L. (2010). Upward spirals of the heart: Autonomic flexibility, as indexed by vagal tone, reciprocally and prospectively predicts positive emotions and social connectedness. Biological Psychology, 85(3), 432-436.View
Fragala-Pinkham, M., Haley, S. M., & O’Neil, M. E. (2008). Group aquatic aerobic exercise for children with disabilities. Developmental Medicine & Child Neurology, 50(11), 822-827.View
Getz, M., Hutzler, Y., & Vermeer, A. (2006). Effects of aquatic interventions in children with neuromotor impairments: a systematic review of the literature. Clinical Rehabilitation, 20(11), 927-936.View
Nichols, W. J. (2015). Blue mind. Little Brown.View