Journal of Rehabilitation Practices and Research Volume 1 (2021), Article ID: JRPR-126
https://doi.org/10.33790/jrpr1100126Research Article
Effect of Aerobic Exercise and Dietary Intervention on Visceral and Subcutaneous Adipose tissue in Sedentary Overweight Adults: a Magnetic Resonance Imaging Study
Jui-Hsing Lin1, Michael T.C. Liang2*, Chin-Lung Fang3, Wen-Pao Teng4
1Department of Physical Education, College of Science National Pingtung University No. 4-18, Min Sheng Road, Pingtung, Taiwan 900-03.
2Professor, Department of Kinesiology and Health Promotion, College of Science California State Polytechnic University 3801 West Temple Avenue, Pomona, California 91768, United States.
3Department of Physical Education, National Taiwan Normal University, Ho-ping East Road, Taipei, Taiwan 106.
4Department of Radiology, Pingtung Christianity Hospital, Pingtung, Taiwan 90003.
Corresponding Author Details: Michael T.C. Liang, Ph.D., Professor, Department of Kinesiology and Health Promotion, College of Science California State Polytechnic University 3801 West Temple Avenue, Pomona, California 91768, United States. E-mail: mtcliang@cpp.edu
Received date: 13th August, 2021
Accepted date: 02nd October, 2021
Published date: 05th October, 2021
Citation: Lin, J.H., Liang, M.T.C., Fang, C.L., & Teng, W.P. (2021). Effect of Aerobic Exercise and Dietary Intervention on Visceral and Subcutaneous Adipose tissue in Sedentary Overweight Adults: a Magnetic Resonance Imaging Study. J Rehab Pract Res, 2(2):126.
Copyright: ©2021, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
The purpose of this study was to examine the effect of aerobic exercise and anti-atherosclerotic diet intervention on visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT), lipid profiles and insulin (IN) concentration in overweight adults.
Method: Thirty-six sedentary overweight adults, age 20 to 25 yr, volunteered as study subjects. Subjects were randomly assigned to: 1) exercise training with American Heart Association anti-atherosclerotic Step 1 (AHA-S1) diet (E+D, n=12), 2) exercise training alone (without the AHA-S1 diet) (EX, n=12), and 3) no-treatment control (CON, n=12). The E+D trained 45 min a day at 70% of VO2peak, 3 days/week and consumed the AHA-S1 diet for 12 weeks, while the EX trained at the identical exercise intensity, duration and frequency without the AHA-S1 diet for 12 weeks. The CON did not engage in exercise training and consumed preferred habitual diet for the same study duration. Study outcome measurements include a magnetic resonance imaging scan for visceral adipose tissue (VAT) and subcutaneous adipose tissue (SAT), body weight (BW), VO2peak, insulin concentration (IN), and fasting lipids.
Results: The E+D showed significant (p<0.05) reduction in VAT, SAT, WT and IN as well as increase in VO2peak and HDL-cholesterol. The EX showed significant (p<0.05) reduction in IN and increase in VO2peak and HDL-Cholesterol.
Conclusion: E+D was effective in lowering SAT, VAT, WT and IN as well as enhancing VO2peak and HDL-cholesterol, while EX was effective in lowering IN and enhancing VO2peak and HDL-cholesterol in sedentary overweight adults.
Keywords: Aerobic exercise, American Heart Association Step-One diet, MRI, Abdominal and visceral fat, VO2peak, LDL-cholesterol, HDL-cholesterol,
Introduction
Studies have shown that there is a strong association between excess abdominal adipose tissue and the presence of metabolic risk factors [1,2]which include abdominal obesity, elevated plasma glucose, dyslipidemia, and hypertension [3,4]. Note that intra-abdominal fat includes retroperitoneal and intraperitoneal fat with the latter also known as visceral adipose tissue (VAT) [5,6]. VAT is considered an important biomarker of metabolic dysfunctions that are linked to cardiovascular disease [5-9]. Note that the effect of abdominal obesity including VAT contributes to insulin resistance or insulin sensitivity in overweight and obese people and also in non-obese people of normal weight [5,6]. Researchers have used computed tomography or magnetic resonance imaging (MRI) to categorize adipose tissue mass at the abdominal level into intra-abdominal fat and subcutaneous fat [10,11,12]. The advantage of employing MRI scan in assessing abdominal obesity is that it can accurately quantify VAT distribution and is a safe, non-invasive and non-radioactive technology that can be used in weight control study with pre-intervention and repeated follow-up assessments of regional adipose tissue distribution [10-12].
Studies have shown that in sedentary overweight adults, training with aerobic exercise enhanced cardiorespiratory fitness [13-15], and lowered body weight, VAT [16-20], risk factors for cardiovascular disease [15,21], fasting insulin and triglyceride levels [16]. Although the independent effect of exercise training and weight loss on metabolic disorders has been investigated, the inter-relationship of exercise and abdominal obesity, VAT and cardiovascular disease is unknown [22-26]. It is unclear if exercise training with an anti-atherosclerotic diet can induce weight loss, lower VAT distribution and the risk for the development of metabolic disease in young sedentary and overweight adults. The purpose of this study was to examine the effect of exercise training and anti-atherosclerotic diet intervention on VAT distribution, subcutaneous adipose tissue (SAT), serum lipid profiles and insulin (IN) concentration, and cardiorespiratory fitness in young sedentary overweight adults. We hypothesize that exercise training with dietary intervention will lower VAT, SAT, serum lipid profiles and insulin concentration and enhance cardiorespiratory fitness in young sedentary overweight adults.
Methods and Material
Subjects
Thirty-six sedentary overweight adults volunteered for this study. The inclusion criteria are sedentary adults (men = 19), age 19 – 25 yr, with body mass index (BMI) between 24.0 and 31 kg/m2. The exclusion criteria include those with a BMI ≥32 kg/m2 and having the following diseases or health conditions: cardiovascular, pulmonary, liver, kidney, gastrointestinal disease or cancer, orthopedic disorders or eating disorders, and use of any tobacco product or alcohol. “Sedentary” behavior was defined as participation in less than 30 min per day, 2 or less days per week in aerobic exercise or strength training for the past 2 years (ACSM’s 2018) and having a VO2peak ≤ 35 ml/ kg/min for men or ≤ 28 ml/kg/min for women. All subjects signed a written informed consent approved by the Institutional Review Board of the National Taiwan Normal University and Pingtung Christianity Hospital to serve as study subjects. The research procedures used in this study adhere to the principles of the Declaration of Helsinki.
Study Design
The subjects were randomly assigned to 1) exercise with the American Heart Association anti-atherosclerotic Step 1 (AHA-S1) diet (E+D, n =12, 5 men and 7 women), 2) exercise without the AHA-S1 diet (EX, n =12, 6 men and 6 women), and 3) non-treatment control group (CON, n= 12, 8 men and 4 women). Both E+D and EX groups exercised 45 min a day at 70%VO2peak, 3 days a week for 12 weeks. All training sessions were closely monitored and supervised. In addition, subjects were required to 1) maintain regular habitual activities for daily living, 2) avoid participating in any new physical activity or exercise, 3) maintain preferred habitual diet, except for the E+D group, and 4) submit 7-day food-exchange records and weekly 24-hour dietary recall questionnaires for the entire study period. Satisfactory compliance with the study was defined as attended ≥ 85% of all exercise sessions and submitted ≥ 85% of food-exchange records for the entire study period.
The supervised cycling exercise regimen consisted of 45 min of indoor stationary cycling at 70% of VO2peak, 3 sessions a week for 12 weeks performed at the Exercise Physiology Laboratory. Prior to the beginning of the exercise training, a familiarization period was provided to the exercise groups at the first weeks of the study. This included 3 sessions of stationary cycling exercise at 70% VO2peak to ensure that the prescribed exercise intensity was attained and can be maintained for a 15-20 min period. The purpose was to minimize the risk of muscle soreness or injury. The duration of cycling exercise was gradually increased from 15-20 to 40-45 min per session during the second week at the intensity of 70% VO2peak. At the end of the familiarization period, the training subjects were able to complete the exercise regimen at the prescribed exercise intensity, duration and frequency throughout the remaining weeks of the study. To ensure that the required exercise intensity of 70% VO2peak was maintained, the E+D and EX subjects repeated the VO2peak cycling test every four weeks, and an updated cycling training work rate was calculated and used. The exercise training compliance rates for the E+D and EX were near 95%. There were no subject dropouts throughout the entire 12-week of study.
The American Heart Association Step One Diet
Subjects in the E+D group were initially instructed to strictly follow the AHA-S1 Diet by a registered dietician and thereafter every four weeks by the same dietician to ensure that they closely followed with the AHA-S1 diet. During the study period, all meals were prepared and provided for the E+D group by the study. Their meals were served at the university’s cafeteria at no cost to the participants. The AHA-S1 diet meals were prepared by the university’s cafeteria staff. For the EX and CON, their personally preferred habitual meals were consumed at the university cafeteria or off-campus local food stores. The subjects were required to strictly follow their preferred habitual daily meals (Taiwanese, Chinese or Western-style foods) throughout the study period. In addition, the subjects were required to submit a seven-day food exchange record which was reviewed by the registered dietician to check for dietary compliance or attrition and omitted information.
The AHA-S1 diet was aimed at preventing risk of cardiovascular disease (CVD) by lowering low-density lipoprotein cholesterol (LDL-C), total cholesterol (TC), and triglycerides (TG). The percentage of total daily calorie intake was <30% from dietary fat, <10% from saturated fat, 10% from monounsaturated fats, and up to 10% polyunsaturated fat. The total dietary protein and carbohydrate from the AHA-S1 diet was, respectively, 15% and 55% of total daily calories. Other dietary modifications in the AHA-S1 diet included dietary cholesterol < 300 mg per day, sodium < 2,400 mg per day, and fiber 20 to 30 g per day. The source of protein in the AHA-S1 diet includes plant or lean animal protein, and the source of complex carbohydrates includes vegetables, fruit and whole grains (https:// www.slideshare.net/heart.step.diet.plan/american-heart-association-step-1-diet).
Study Outcome Measurements
Pre- and post-training outcome measurements included:
1. Visceral adipose tissue (VAT), subcutaneous adipose tissue (SAT) and total abdominal fat (TAF) were assessed using a magnetic resonance imaging (MRI, Sigma Advantage, Model 1.5 Tesla Signa Cvi, General Electric Medical System, Milwaukee, WI, USA). Briefly, subjects were examined in a supine position and all images were obtained using the radio frequency (RF) coil. A series of four spin echo T1 weighted transverse sections were obtained through the abdominal region. The parameters for the transverse series was: repetition time (TR) =300ms, time to echo (TE) =12ms, 256×128 matrix, 4 excitations (NEX), field of view 48 cm, slice thickness 10 mm, and inter-slice gap 10 mm. Calibration of the MRI instrument was performed daily prior to the study measurements. The MRI instrument and the phantom interception at the navel alignment in one centimeter and downward 19 centimeters with every other one centimeter, respectively, were cut once, altogether 20 slides were obtained (Figure 1a and 1b). After obtaining the scanned images, MRI image data files were analyzed separately for calculating subcutaneous adipose tissue and visceral adipose tissue. The abdomen subcutaneous fat was estimated using the mean value of 20 transverse cutting area. A single investigator (WPT) who was blinded to the subject group assignments performed all measurements and MRI image analyses.
2. Blood lipid concentrations including triglycerides (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDL-C), low-density lipoprotein cholesterol (LDL-C), and serum insulin concentration (IN) were measured by the Clinical Reference Laboratory following the National Clinical Laboratory Procedures and Standards at the Pingtung Christianity Hospital, Pingtung, Taiwan.
3. Body weight was measured using a clinical balance scale to the nearest 0.1 kg with the subjects dressed in light clothing. Standing height was measured to the nearest 0.1 cm using a wall-mounted stadiometer. Body mass index (BMI) was calculated as weight/height2and expressed as kg/m2.
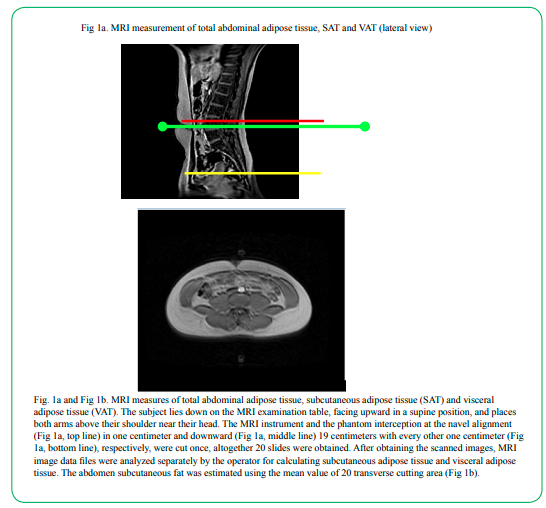
Fig. 1a and Fig 1b.MRI measures of total abdominal adipose tissue, subcutaneous adipose tissue (SAT) and visceral adipose tissue (VAT). The subject lies down on the MRI examination table, facing upward in a supine position, and places both arms above their shoulder near their head. The MRI instrument and the phantom interception at the navel alignment (Fig 1a, top line) in one centimeter and downward (Fig 1a, middle line) 19 centimeters with every other one centimeter (Fig 1a, bottom line), respectively, were cut once, altogether 20 slides were obtained. After obtaining the scanned images, MRI image data files were analyzed separately by the operator for calculating subcutaneous adipose tissue and visceral adipose tissue. The abdomen subcutaneous fat was estimated using the mean value of 20 transverse cutting area (Fig 1b).
4. Peak oxygen uptake (VO2peak) was measured using a 4-minute per stage cycle ergometry (Monark model 818) protocol and a metabolic cart for measuring O2 and CO2 content (SensorMedice 2900, Yorba Linda, CA). VO2peak was determined when subjects achieved two of the following three conditions: 1) maximal heart rate reached 90% of age-predicted maximal heart rate (maximal heart rate =220-age), 2) respiratory exchange ratio ≥ 1.00, and 3) plateau in VO2 (<2.0 ml/kg/min with increased workload). During the exercise test, maximal heart rate was determined electrocardiographically every minute.
5. Cardiorespiratory function parameter. Resting heart rate (RHR) was obtained with the subject sitting quietly for 15 minutes and then recorded every 5 minutes using a Polar Vantage heart rate monitor. The lowest RHR value was recorded and used for statistical analysis. Following the heart rate measurement, resting systolic and diastolic pressure were measured using a mercurial sphygmomanometer (Sanken Model SM-301, Tokyo, Japan) with the subjects resting in a sitting position. Duplicate blood pressure measurements were taken, separated by a 2-min interval. The lowest values of the systolic and diastolic blood pressure readings were recorded and used for statistical analysis.
Statistical Procedure
All statistical analyses were performed with the SPSS Statistical Software (version 16). Initially, group differences in baseline values were compared using analysis of variance (ANOVA). These comparisons were done among those who completed the 12-week study and had follow-up measurement for the MRI data. All variables tested between groups using the ANOVA were homogenous and continuously distributed. Analysis of covariance (ANCOVA), with repeated measures across all factors, was used to examine for independent group difference in changes from baseline to 12-week in the primary outcome variables. In addition to the baseline measure of outcomes being assessed, covariates included in the statistical analyses were gender (male = 1 and female = 2), VO2peak, and body weight. When a significant groups difference in any outcome was present, a repeated measure ANCOVA was performed for that variable, with the group entered as the between subject factors and the 2 time points entered as within subject factors. Statistical significance was set at α < 0.05. Results are presented as means ± SD.
Result
The randomization process balanced all characteristics, with the exception of VO2peak and body weight. At baseline, VO2peak was significantly higher in the CON, compared to E+D and EX (p < 0.05, Table 2), and body weight was significantly greater in the E+D, compared to EX. There were no significant differences in other baseline cardiorespiratory function parameters, and in anthropometric and lipid parameters.
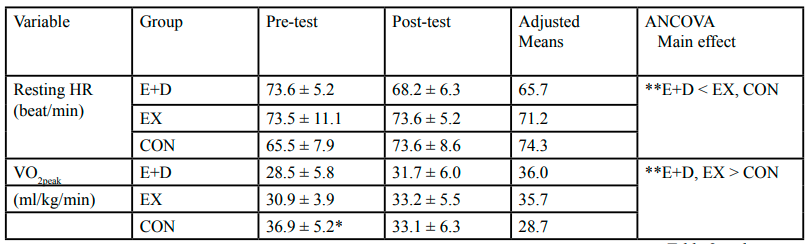
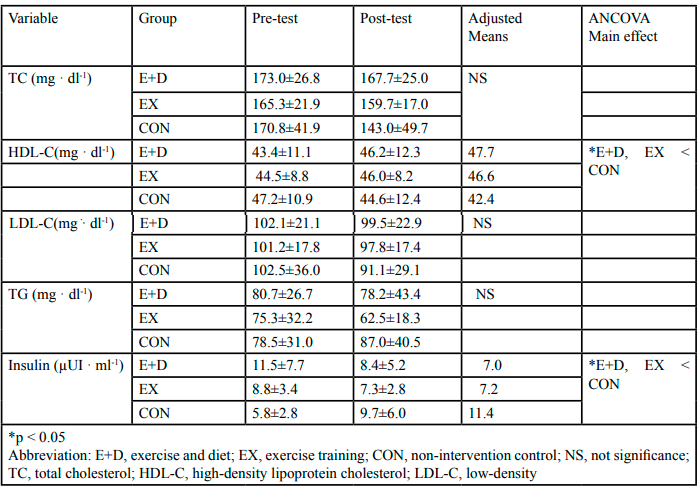
Table 1, 2 and 3 compare group differences in changes from baseline to post-intervention, adjusted for covariates which include gender, VO2peak, and body weight. The pre-post test results show that the E+D group exhibited significant decrease (all p < 0.05) in body weight (4.6%), abdomen transverse cutting area (10.8%), SAT (9.4%), and VAT (15.6%) (Table 1). These changes were not observed in EX and CON. The pre-post test results in VO2peak showed improvement (p < 0.05) in both E+D (11.2%) and EX (7.4%), not the CON (Table 2). Results also show improvements (p<0.05) in HDL-C (6.5%) and IN (26.9%) in E+D, and in HDL-C (3.7%) and IN (17%) in EX (Table 3).
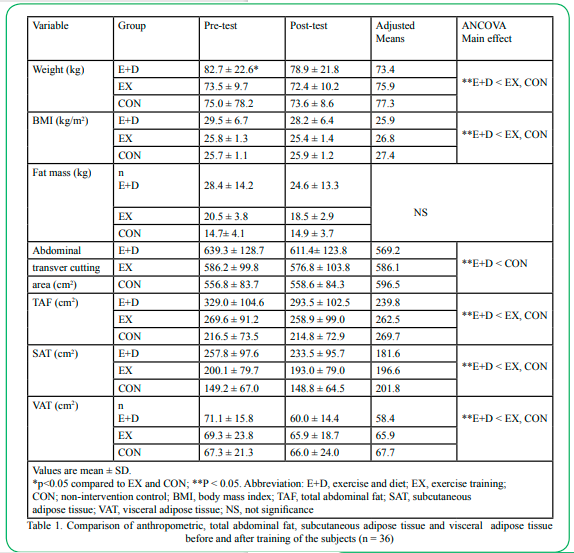
Table 1. Comparison of anthropometric, total abdominal fat, subcutaneous adipose tissue and visceral adipose tissue before and after training of the subjects (n = 36)
The compliance rate of the E+D with the AHA S1diet was 94%. Note that two E+D subjects failed to follow through with the AHA-S1 diet and their results were excluded from the study. One EX subject missed two food-exchange records throughout the 12-week study period and their data were included for the final analysis. All CON subjects fully complied with the food-exchange records. The overall subjects’ compliance rate for the entire study was >90% which was considered highly satisfactory.
Discussion
The purpose of this study was to assess the effect of exercise training with the AHA S1 diet intervention on visceral and central adipose tissue distribution, serum lipid profile, and cardiorespiratory fitness in young sedentary overweight adults. Our results support the hypothesis that 12 weeks of exercise training with the AHA-S1 diet intervention resulted in significant reductions in weight, TAF, SAT, VAT, and IN as well as increase in HDL-C and VO2peak (All p < 0.05). The findings suggest that the combination of aerobic exercise training and AHA-S1 diet intervention were effective for reduction of body weight, VAT, SAT, and TAF, and improvement of HDL-C and IN in young sedentary overweight adults.
Our study results supported other studies using MRI scanner that showed exercise in combination with diet intervention lowered VAT, SAT and TAF [27,28]. Kamel et al. [28] observed changes in intra-abdominal adipose tissue volume during weight loss in obese men and women and reported high correlations between MRI and anthropometric measurements. Specifically, Kamel et al. [28] reported that a reduction in endo-abdominal fat distribution is correlated with SAT and TAF. Marks [27] using MRI to study metabolic effects of exercise training on VAT distribution in abdominally obese men and found that 12 weeks of exercise intervention lowered VAT by 21.0% and BMI by 5.8% (Both p<0.05), not SAT and fat mass. Another study by Giannopoulou et al. [29] found in women with type 2 diabetes that moderate weight loss (4.5 kg) resulted in TAF and VAT loss, suggesting that exercise training was the contributing factor for the reduction of VAT. Results obtained from the present study support the contention that AHA-S1 diet and aerobic exercise training decrease VAT, SAT, TAF, and insulin concentration as well as increase HDL-cholesterol and cardiorespiratory fitness.
Note that a 12-week of aerobic training regimen using stationary cycling exercise at intensity of 70% VO2peak exhibits significant increase in VO2peak and HDL-cholesterol as well as decrease in insulin concentration. The results suggest that health benefits of aerobic exercise in sedentary young overweight adults include lowering of cardiovascular and metabolic disease risk factors. As far as we know, this is one of the few studies that employed MRI technology to document changes in VAT and SAT distribution from AHA-S1 diet and exercise training. The employment of the MRI technology for assessing visceral and subcutaneous adipose tissue on the subjects might have had an influence on subjects’ behavior for maintaining high compliance rates with the diet and exercise training, i.e., both were >90%.
Other studies did not observe positive changes in VAT, serum lipid profile, insulin concentration, and body weight with aerobic exercise training. For example, Grundy et al. [26] found that exercise training alone did not lower TAF and VAT. Rimmer et al. [30] investigated the role of aerobic exercise on serum lipid profile in adolescents and found no change in HDL-C after a 15-week exercise intervention. Gutin et al. [20] found no significant improvement in fasting glucose and insulin concentration. On exercise training and dietary intervention and changes in VAT and body weight parameters, Slentz et al. [16] and Gutin et al [20] observed that 8 months of aerobic exercise was an effective mode of exercise for lowering VAT and fasting insulin concentration in sedentary overweight adults. Slentz et al. [16] , and Mayo et al. [19] observed that exercise training was an effective means for weight control. Therefore, it is unclear as to the independent effects of diet- or exercise-induced weight loss in overweight and obese individuals especially as they relate to cardiovascular disease risk and comorbidity. Our study has limitations that must be addressed. First, this cross-sectional study is limited for drawing any conclusion on causal relationships. Second, a lack of existing research in the area of abdominal obesity and MRI study with cycling exercise limiting our findings to be generalized to sedentary abdominal obesity adults.
We conclude that for young sedentary overweight adults, exercise training with the AHA-S1 diet is effective for inducing significant reductions in VAT, SAT, body weight and insulin concentration, and increase in VO2peak and HDL-cholesterol. In addition, aerobic exercise training is effective for improving VO2peak and HDL-C as well as lowering insulin concentration. Further study is warranted to examine the independent effects of diet- or exercise-induced weight loss in sedentary overweight and obese adults as they relate to cardiovascular disease risk and comorbidity.
Competing interest statement:
Jui-Hsing Lin, Michael T.C. Liang, Chin-Lung Fang, and Wen-Pao Teng declare that they have no competing financial interests and have no professional relationships with the manufacturer of the MRI instrument used in this study that may benefit from the results of this manuscript.
Author’s contributions:
All authors contributed to the study conception and design. Material preparation and data collection were performed by JH Lin, CL Fang, WP Teng, and MTC Liang. Statistical analysis was performed by JH Lin and MTC Liang. WP Teng performed all MRI scans and supervised all clinical laboratory analysis. The first draft of the manuscript was written by JH Lin, subsequent versions of the manuscript were revised by MTC Liang and JH Lin, and all authors commented on previous and current versions of the manuscript. All authors read and approved the final manuscript.
Acknowledgements:
We would like to acknowledge our graduate students for their assistance in the project, and the participants for their time, effort and commitment in completing the study.
References
Bray GA, Jablonski KA, Fujimoto WY, Barrett-Connor E, Haffner S, Hanson RL, Hill JO, Hubbard V, Kriska A, Stamm E, Pi-Sunyer FX. (2008). The Diabetes Prevention Program Research Group. Relation of central adiposity and body mass index to the development of diabetes in the Diabetes Prevention Program. Am J Clin Nutr, 87: 1212–1218.View
Klein S, Allison DB, Heymsfield SB, Kelley DE, Leibel RL, Nonas C, Kahn R. (2007). Waist circumference and cardiometabolic risk: a consensus statement from Shaping America's Health: Association for Weight Management and Obesity Prevention; NAASO, The Obesity Society; the American Society for Nutrition; and the American Diabetes Association. Am J Clin Nutr, 85: 1197–202.View
Grundy, S., Cleeman, J., Daniels, S., et al. (2005) Diagnosis and Management of the Metabolic Syndrome—An American Heart Association/National Heart, Lung, and Blood Institute Scientific Statement. Circulation, 112, 2735-2752. http://dx.doi. org/10.1161/CIRCULATIONAHA.105.169404View
Pouliot, M.C., Despres, J.P., Nadeau, A., Moorjani, S., Prud'Homme, D., Lupien, P.J., Tremblay, A., & Bouchard, C. (1992). Visceral obesity in men: associations with glucose tolerance, plasma insulin, and lipoprotein levels. Diabetes, 41: 826–834.View
Gastaldelli, A., Miyazaki, Y., Pettiti, M., Matsuda, M., Mahankali, S., Santini, E., DeFronzo, R.A., & Ferrannini, E. (2002). Metabolic effects of visceral fat accumulation in type 2 diabetes. J Clin Endocrinol Metab, 87: 5098–5103.View
Banerji, M.A., Chaiken, R.L., Gordon, D., Kral, J.G., & Lebovitz, H.E. (1995). Does intra-abdominal adipose tissue in black men determine whether NIDDM is insulin-resistant or insulin-sensitive? Diabetes, 44: 141–146.View
Kuk, J. L., Katzmarzyk, P. T., Nichaman, M. Z., Church, T. S., Blair, S. N., & Ross, R. (2006). Visceral fat is an independent predictor of all-cause mortality in men. Obesity, 14: 336-341.
Yusuf, S., Hawkens, S., Ounpuu, S., Dans, T., Avezum, A., Lanas, F., McQueen, M., Budaj, A., Pais, P., Varigos, J., Lisheng, L., INTERHEART Study Investigators. (2004). Effect of potentially modifiable risk factors associated with myocardial infarction in 52 countries (the INTERHEART study): Case-control study. Lancet, 364(9438): 937-952 DOI: 10.1016/ S0140-6736(04)170189.View
Bjorntorp P: Portal adipose tissue as a generator of risk factors for cardiovascular disease and diabetes. (1990). Arteriosclerosis, 10: 493–496.View
Szabo, C., Babinszky, L., Verstegen, M. W. A., Vangen, O., Jansman, A. J. M., & Kanis, E. (2000). The application of digital imaging techniques in the in vivo estimation of the body composition of pigs: a review. Livestock Production Science, 60, 1-11. View
Gronemeyer, S. A., Steen, R. G., Kauffman, W. M., Reddick, W. E., & Glass, J. O. (2000). Fast adipose tissue (FAT) assessment by MRI. Magnetic Resonance Imaging, 18, 815-818.View
Ross, R., & Janssen, I. (1999). Is abdominal fat preferentially reduced in response to exercise-induced weight loss? Medicine and Science in Sports and Exercise, 31(11), S568-S572.View
Grundy, S.M., Barlow, J.I., Farrell, S.W., Vega, G.L., & Haskell, W.L. (2012). Cardiorespiratory fitness and metabolic risk. Am J Cardiol, 109:988-993.View
Gutin, B., Barbeau, P., Owens, S. et al. (2002). Effects of exercise intensity on cardiovascular fitness, total body composition, and visceral adiposity of obese adolescents. Am J Clin Nutr, 75:818- 826.View
Suzuki, I., Yamada, H., Sugiura, T., Kawakami, N., & Shimizu, H. (1998). Cardiovascular fitness, physical activity and selected coronary heart disease risk factors in adults. J Medicine and Physical Fitness, 38(2), 149-157.View
Slentz, C.A., Bateman, L.A., Willis, L.H., Shields, A.T., Tanner, C.J.,& Piner, L.W., et al. (2011). The effects of aerobic versus resistance training on visceral and liver fat stores, liver enzymes and HOMA from STRRIDE AT/RT: A Randomized Trial. Am J Physiol Endocrinol Metab, 301(5):E1033-1039.View
Hunter, G.R., Brock, D.W., Byrne, N.M., Chandler-Laney, P.C., Del Corral, P., & Grower, B.A. (2010). Exercise training prevents regains of visceral fat for 1 year following weight loss. Obesity (Silver Spring), 18(4):690-695.View
Ross, R., Janssen, I., Dawson, J., Kungl, A. M., Kuk, J. L., Wong, S. L., Nguyen-Duy, T. B., Lee, S., Kilpatrick, K., & Hudson, R. (2004). Exercise-induced reduction in obesity and insulin resistance in women: a randomized controlled trial. Obesity Research, 12(5), 789-798.View
Mayo, M. J., Grantham, J. R., & Balasekaran, G. (2003). Exercise induced weight loss preferentially reduces abdominal fat. Medicine and Science in Sports and Exercise, 35(2), 207- 213.View
Gutin, B., Barbeau, P., Owens, S., et al. (2002) Effects of Exercise Intensity on Cardiovascular Fitness, Total Body Composition, and Visceral Adiposity of Obese Adolescents. American Journal of Clinical Nutrition, 75, 818-826.View
Canoy, D. (2008). Distribution of body at and risk of coronary heart disease in men and women. Current Opinion in Cardiology, 23(6), 591-598.View
Murphy, J.C., McDaniel, J.L., Mora, K., Villareal, D.T., Fontana, L., & Weiss, E.P. (2012). Preferential reduction in intermuscular and visceral adipose tissue with exercise-induced weight loss compared with calorie restriction. J Appl Physiol, 112(1): 79-85. DOI: 10.1152/japplphysiol.00355.2011.View
Kriska, A. (2003). Can a physically active lifestyle prevent type 2 diabetes? Exerc Sports Sci Rev, 31: 132-137.View
Goodpaster, B.H., Katsiaras, A., & Kelley, D.E. (2003). Enhanced fat oxidation in physical activity is associated with improvements in insulin sensitivity in obesity. Diabetes; 52:2191-2197. View
Irwin, M.L., Mayer-Davis, E.J., & Addy, C.L., et al. (2000). Moderate-intensity physical activity and fasting levels in women: the Cross-Cultural Activity Participation Study. Diabetes Care, 23:449-454 JAMA (January issue 2009).View
Grundy, S. M., Blackburn, G., Higgins, M., Lauer, R., Perri, M. G., & Ryan, D. (1999). Physical activity in the prevention and treatment of obesity and its comorbidities: evidence report of independent panel to assess the role of physical activity in the treatment of obesity and its comorbidities. Medicine and Science in Sports and Exercise, 31(11), 1493-1500.View
Marks, S. J., Chin, S., & Strauss, B. J. G. (1998). The metabolic effects of preferential reduction of visceral adipose tissue in abdominally obese men. International Journal of Obesity, 22, 893-898.View
Kamel, E. G., McNeill, G., & Wijk, M. V. (2000). Change in intra-abdominal adipose tissue volume during weight loss in obese men and women: correlation between magnetic resonance imaging and anthropometric measurements. International Journal of Obesity, 24, 607-613.View
Giannopoulou, I., Ploutz-Snyder, L.L., Carhart, R., Weinstock, R.S., Fernhall, B., Goulopoulou, S., Kanaley, J.A. (2005). Exercise is required for visceral fat loss in postmenopausal women with type 2 diabetes. J Clin Endocrinol Metab, 90(3):1511-8. doi: 10.1210/jc.2004-1782.View
Rimmer, J. H., Looney, M. A. (1997). Effect of an aerobic activity program on the cholesterol levels of adolescents. Research Quarterly for Exercise and Sport, 68(1), 74-79.