Journal of Rehabilitation Practices and Research Volume 3 (2022), Article ID: JRPR-138
https://doi.org/10.33790/jrpr1100138Research Article
Effectiveness of Surface Electromyographic Biofeedback-triggered Neuromuscular Electrical Stimulation on Quadriceps Femoris Torque and Recruitment
Tony Boucher, PhD, MPT, LAT, ATC
Clinical Associate Professor, Coordinator of Clinical Education, Department of Kinesiology and Sport Management, Texas A&M University, College Station, 135 Gilchrist Building, United States.
Corresponding Author Details: Tony Boucher, PhD, MPT, LAT, ATC, Clinical Associate Professor, Coordinator of Clinical Education, Department of Kinesiology and Sport Management, Texas A&M University, College Station, 135 Gilchrist Building, Tx, United States.
Received date: 20th September, 2022
Accepted date: 05th November, 2022
Published date: 08th November, 2022
Citation: Boucher, T., (2022). Effectiveness of Surface Electromyographic Biofeedback-triggered Neuromuscular Electrical Stimulation on Quadriceps Femoris Torque and Recruitment. J Rehab Pract Res, 3(2):138.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Context: Injury and pathology often result in physiological impairments including decreased strength, muscular performance, and arthrogenic muscle inhibition. Neuromuscular electrical stimulation initiated by a surface electromyographic biofeedback threshold (sEMG-triggered NMES) is effective in neurological populations but has been minimally applied to orthopedic populations.
Objective: The purpose of the study was to investigate the effectiveness of sEMG-triggered NMES on maximal isometric knee extensor torque and neuromuscular recruitment.
Design: Counterbalanced crossover study design.
Setting: University research laboratory.
Participants: Twenty-two healthy persons (14 men, 8 women; age = 22.9 ± 4.7 years; height = 171.7 ± 6.1 cm; mass 76.6 ± 19.4 kg) participated.
Intervention: Maximal isometric knee extensor torque @ 60° knee flexion was assessed with sEMG-triggered NMES applied to the quadriceps femoris compared to voluntary contraction alone on two separate testing sessions 2 to 7 days apart.
Main outcome measures: Peak torque, peak torque to body weight ratio, and time to peak torque were assessed.
Results: Analysis of variance (ANOVA) showed peak torque produced with sEMG-triggered NMES (191.6 ± 59.8 Nm) was significantly greater than without (167.4 ± 54.7 Nm). Average Peak torque to body weight ratio with sEMG-triggered NMES (224.8 ± 66.9 Nm) produced significantly greater torque than without (196.4 ± 51.1 Nm). Time to peak torque with the sEMG-triggered NMES (3.3 ± 1.6 sec) was significantly faster than volitional contractions (4.2 ± 2.1 sec) during the sEMG-triggered NMES testing session and significantly quicker than all volitional contractions (3.9 ± 2.7 sec) for both testing sessions.
Conclusions: sEMG-triggered NMES is more effective in producing greater quadriceps femoris torque while producing quicker neuromuscular recruitment than voluntary contractions alone. Results support the use of sEMG-triggered NMES applications to improve knee extensor torque and recruitment of the QF to influence arthrogenic muscle inhibition, facilitate QF recruitment, and improve strength.
Key Words: sEMG-triggered NMES; Torque; Recruitment; Quadriceps
Introduction
Injury and pathology often result in physiological impairments including decreased strength and muscular performance. Arthrogenic muscle inhibition (AMI) of the quadriceps femoris (QF) muscle, often attributed to pain and swelling, is a limiting factor in full muscle activation and restoration of strength [1-3]. Conventional strength exercise programs do not focus on limiting AMI and may not result in full restoration of the muscle. Disinhibiting the muscle before performing exercise may promote a more optimal neural environment for normal motor patterns. Interventions that decrease inhibition and allow for active exercise enhance recovery [4]. Surface EMG (sEMG) biofeedback and neuromuscular electrical stimulation (NMES) applications are commonly used interventions to limit inhibition, facilitate QF recruitment, and improve strength.
Surface EMG uses sensory feedback to promote normal voluntary asymmetrical motor unit recruitment patterns. Surface EMG has long been demonstrated to be effective for improving isometric strength and recruitment in healthy individuals [5,6]. Surface EMG has also been shown to enhance patellar alignment and recruitment in individuals with knee pathology as well as improve isometric strength, recruitment, and range of motion (ROM) in post surgical patients [7-10]. Systematic review and clinical recommendations reveal inconsistent support of EMG for increasing QF muscle activation or chronic knee conditions but benefits short-term post surgical pain and QF strength [11,12]. Other investigators, however, have not observed strength improvements with sEMG interventions [13].
Neuromuscular electrical stimulation utilizes electrically induced muscle contraction to facilitate symmetrical motor unit recruitment patterns that have the potential to exceed voluntary physiologic contraction. NMES has proven to be effective for improving isometric strength in healthy individuals and post surgical isometric strength and gait [14-19]. Systematic review and clinical practice guidelines support NMES for increasing strength following anterior cruciate ligament reconstruction, meniscal and chondral injury [20,22]. Other authors have found no benefit for NMES intervention [23,25].
There is conflicting evidence regarding the effectiveness of independent NMES and EMG biofeedback rehabilitation applications for improving muscle function. The combination of sEMG and NMES utilized by sEMG-triggered NMES may provide summative advantages. The sEMG component requires a maximum volitional isometric contraction (MVIC) and once the participant exceeds a pre-determined sEMG threshold, the NMES component will elicit an electrically induced contraction. Surface EMG combined with NMES has been shown to improve functional recovery of patients with cerebral vascular accidents [26-31]. Surface EMG-triggered NMES has also shown effectiveness for increasing knee extension ROM in postoperative knee patients [32].
The sensory input of sEMG biofeedback to produce a maximal physiologic contraction and the ability of NMES to augment this contraction can potentially improve strength and neuromuscular recruitment. The proposed effectiveness of sEMG-triggered NMES needs to be determined prior to its application on the pathological population. However, to our knowledge no study has been performed relative to sEMG-triggered NMES application in the orthopedic population.
The purpose of this study was to investigate the effectiveness of sEMG-triggered NMES for improving isometric knee extensor torque and time to peak torque in healthy individuals. It was hypothesized that sEMG-triggered NMES would be more effective for improving isometric torque and time to peak torque versus voluntary contraction alone.
Materials and Methods
Design
The participants completed two separate torque testing sessions 2 to 7 days apart: 1) MVIC with sEMG-triggered NMES test session and 2) MVIC-only test session. The two separate test sessions were compared to determine the effectiveness of the sEMG-triggered NMES on torque outcome measures. No training was administered during the research study. The counterbalanced order of the testing sessions eliminated any systematic and learning effects due to the order of testing. For both test sessions, the same limb was used and randomly determined by a coin flip to eliminate any dominant limb influence.
The investigator examined three dependent variables: 1) Knee extensor isometric peak torque, 2) average isometric peak torque to body weight, and 3) time to peak torque were measured utilizing the Biodex System 3 dynamometer (Shirley, NY). The participants performed ten gravity corrected maximal peak isometric contractions at 60° knee flexion during each test session. Isometric peak torque (Nm) represents the single highest torque of the ten contraction repetitions within a testing session. Average peak torque (Nm) to body weight ratio normalizes the average peak torque data across all ten contraction repetitions within a testing session compared to body weight in kilograms. Time to peak torque is the elapsed time (sec) from the initiation of single contraction repetition to the highest torque produced.
Participants
The study recruited healthy men and women, between the ages of 18 to 35 and moderately active (exercise 3-5 times a week) from a university and local community. Participants with history of knee surgery or injury, cardiovascular or neurologic disease, electrical stimulation contraindications, sensation deficits, pregnancy, or formal strength or power training were excluded. The sponsor university Institutional Review Board for Human Subjects granted study approval.
The study conformed to the ethical considerations of the Helsinki Code and participants signed an approved informed consent. The investigator conducted all procedures in the laboratory setting at the sponsor university.
Participants completed a medical history questionnaire, general physical examination to determine eligibility, and were familiarized to the research equipment and procedures.
Participants participated in normal activities during the course of the study but instructed not to exercise at least 48 hours prior to each testing sessions.
Procedures
The Chattanooga Vectra Gynesis (Hixson, TN) electrotherapy unit provided the sEMG-triggered NMES intervention. The electrical stimulation parameters utilized a constant Russian current symmetrical biphasic square waveform with 2,500-Hz carrier frequency delivered in 50 bursts per second. The duty cycle was 10 second on contraction time with 0.5 second ramp and 50 second off time with no ramp down. Following skin prep by cleansing with 70% isopropyl alcohol, three electrodes were placed on the anterior thigh and lateral leg (Figure 1). The investigators placed one negative 7-cm round self-adhesive Empi (St. Paul, MN) StimCare® carbon cloth surface electrode on the motor point of the distal vastus medialis oblique (VMO) muscle belly and one identical positive electrode on the motor point of the mid rectus femoris halfway between anterior superior iliac spine and superior patella. These commonly accepted motor points are the superfiscial location of the motor nerve where electrical stimulation elicits a stronger contration at a lower stimulation intensity [33]. The VMO and rectus femoris have also shown better reliability than the vastus lateralis for QF sEMG applications [34,35]. Both the negative and postive electrodes have dual capabilities of reading sEMG biofeedback signals and transmitting the NMES current. One identical common ground electrode was placed on the bony prominance of the lateral fibular head. The participant used new surface electrodes to limit any potential electrode degradation and to ensure optimal QF contact for sEMG recording and NMES conduction.
Setup
Each participant performed a 60 rpm lower extremity cycle ergometer 5-min warm-up followed by supervised quadriceps and hamstrings stretches held for 30 seconds, three repetitions each. Participants then sat on the dynamometer chair with the trunk erect and hips at 90° flexion secured by a thigh strap, waist strap, and 2 chest straps with the force pad secured one inch superior to medial malleoli. The investigator then aligned the dynamometer's axis of rotation with the lateral femoral condyle, the knee was extended from 90° to 0° to ensure axis of rotation alignment, and gravity correction procedures performed at 0° knee flexion. With the knee positioned at 60° knee flexion, the participant practiced one MVIC for ten seconds at 100% of their perceived MVIC.
Testing phase
The testing phase for each testing session consisted of 10 maximal contractions. Each contraction lasted 10 seconds in duration followed by a 50-second rest period. The investigators instructed the participant to extend their knee by pushing as hard and quickly as possible into the dynamometer force pad. No verbal encouragement was provided and the participant was blinded to visual and audible output from the dynamometer unit and data screen. During the sEMG-triggered NMES test session, the participant was able to view the Vectra Gynesis EMG display for sensory biofeedback input. For the MVIC-only test session, the participant solely performed ten MVIC’s without the sEMG-triggered NMES. Participants returned after 48 hours and within 1 week of the first test session to repeat the testing procedures for the other test testing condition with identical procedures.
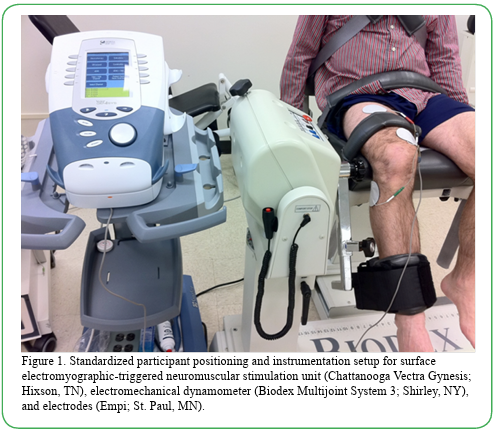
Figure 1. Standardized participant positioning and instrumentation setup for surface electromyographic-triggered neuromuscular stimulation unit (Chattanooga Vectra Gynesis; Hixson, TN), electromechanical dynamometer (Biodex Multijoint System 3; Shirley, NY), and electrodes (Empi; St. Paul, MN).
sEMG-triggered NMES test session
Dosing phase for NMES
The investigators incrementally increased the NMES stimulus intensity until it produced a visible contraction then followed by MVIC. The NMES stimulus was continually increased in intensity amplitude over a ten second period until the participant indicated that their limit of stimulus tolerance based on self-perceived pain sensation had been attained and did not want any further increase. This NMES treatment dosing technique is standard clinical practice and similar to numerous previous clinical research designs [20].
Threshold Phase for sEMG
The investigator determined the threshold value to initiate the NMES from the participant’s sEMG output. Participants performed a MVIC for ten seconds with concurrent monitoring by the sEMG component of the Vectra Gynesis unit capturing the maximum EMG signal produced. Using this maximum EMG signal, the target sEMG threshold to trigger and initiate the NMES was then set at 90% of this MVIC sEMG value. Previous research shows for effects to occur with electrical stimulation, the load on the muscle must be within 10% of MVIC [36].
MVIC-only test session
For the MVIC-only test session, the participant performed the exact MVIC procedures for the dosing phase for NMES and threshold phase for sEMG but without the sEMG-triggered NMES unit.
Statistical analysis
The investigator extracted knee extensor isometric peak torque and average isometric peak torque to body weight ratio from the Biodex dynamometer output. Time to peak torque for each individual contraction repetition was extracted from the Biodex dynamometer graphical data. Data was analyzed using the SPSS 26.0 statistical software program (SPSS Inc., Chicago, IL) with the α level set at 0.05 for all tests. Repeated measures analysis of variance (ANOVA) compared peak torque and average peak torque to body weight between the sEMG-triggered NMES test session and MVIC-only test sessions. Repeated measures ANOVA assessed peak torque across contraction repetitions within each individual test session. Univariate ANOVA compared time to peak torque between contraction repetitions that elicited the sEMG-triggered NMES and contraction repetitions that did not elicit the NMES.
Results
Twenty-two qualifying participants completed the two test sessions with no attrition while three persons were excluded because of history of knee injury. Table 1 shows the descriptive statistics and characteristics of the participants. The degree of skewness was analyzed and determined all data found to be normally distributed.
Knee Extensor Isometric Peak Torque
Table 2 illustrates descriptive statistics of knee extensor peak torque and average peak torque to body weight. ANOVA results revealed the knee extensor peak torque differences were statistically significant between the sEMG-triggered NMES test and the MVIC-only test (F = 8.47, p = 0.008, power = 0.79, effect size =0.29). Participants produced greater isometric peak torque with the sEMG-triggered NMES than MVIC alone.
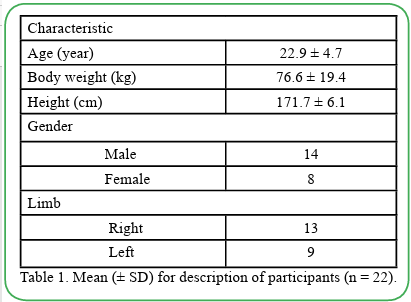
Table 2. Mean (± SD) and 95% confidence interval for knee extensor isometric peak torque and average isometric peak torque to body weight ratio at 60° knee flexion.
During the sEMG-triggered NMES test session, participants reached their predetermined sEMG threshold to elicit the NMES 60% of the time (133 repetitions of the total 220 attempted). The remaining attempts were not capable of producing 90% of the MVIC required to initiate the NMES. ANOVA results of only the contraction repetitions that elicited the sEMG-triggered NMES displayed an increase (185.8 Nm to 195.5 Nm) in peak torque across ten repetitions (F = 1.173, p= 0.35) (Figure 2). Analysis of all contraction repetitions, regardless of whether the sEMG-triggered NMES was elicited, during the sEMG-triggered NMES test session showed a peak torque decrease (180.4 Nm to 172.1 Nm) across ten contractions (F = 1.47, p = 0.23). Analysis revealed a decrease (149.8 Nm to 144.3 Nm) across all ten contractions during the MVIC-only test session (F = 0.805, p = 0.5).
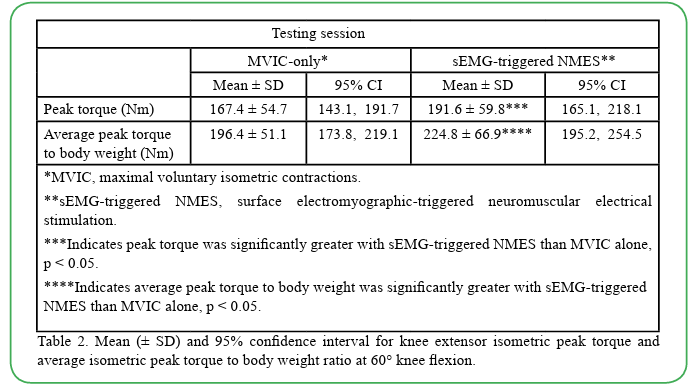
Figure 2. Knee extensor isometric peak torque (Mean ± SD) across contraction repetitions by test session. *sEMG-triggered NMES, surface electromyographic-triggered neuromuscular stimulation. **MVIC, maximal voluntary isometric contraction.
Knee Extensor Average Isometric Peak Torque to Body Weight
ANOVA results revealed statistically significant differences between the sEMG-triggered NMES test and the MVIC-only test (F = 6.72, p = 0.017, power = 0.70, effect size = 0.24 (Table 2). Participants produced greater average isometric peak torque to body weight with the sEMG-triggered NMES than MVIC alone.
Time to Peak Torque
Descriptive statistics of time to peak torque are reported in Table 3. During the sEMG-triggered NMES test session, the differences in time to peak torque exhibited statistical significance for contraction repetitions that elicited the sEMG-triggered NMES and repetitions that did not elicit the NMES (F = 10.966, p = 0.001, power = 0.91, effect size =0.05). For both the sEMG-triggered NMES and MVIC only test sessions, the differences in time to peak torque revealed statistical significance for contraction repetitions that elicited the sEMG-triggered NMES and all other repetitions (F = 5.518, p = 0.019, power = 0.65, effect size =0.01).
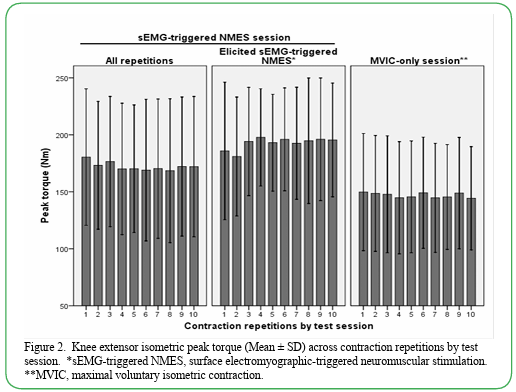
Table 3. Mean (± SD) and 95% confidence interval of time to peak torque for elicited and non-elicited sEMG-triggered NMES repetitions of maximal knee extensor isometric contractions at 60° knee flexion.
Discussion
Knee Extensor Torque
The sEMG-triggered NMES produced significantly greater QF isometric peak torque and average peak torque to body weight than MVIC alone. The isometric peak torque with the sEMG-triggered NMES in this study (191.6 Nm) was higher than the results of healthy similar aged individuals (147.9 Nm and 175.75 Nm) using only NMES at 60° knee flexion [16,19]. The larger results of this study may be attributed to the addition of the sEMG to the NMES.
Despite the fact that only 60% of the attempted repetitions eclipsed 90% MVIC required to trigger the NMES, participants in this study produced significantly greater torque with the sEMG-triggered NMES application due, in part, to the influence of the sEMG biofeedback. Descending pathways are known to influence reflex activation through spinal interneurons pre-synaptic inhibition [37]. The sEMG biofeedback, including change in muscle length, muscle tension, and tactile sensation, influences the descending pathways either increasing the frequency of active motor units discharge or the number of motor units recruited.
The effects of sEMG biofeedback include learning and motivation enhancing enthusiasm and compliance. Biofeedback can increase the person’s muscle awareness by providing additional information on the quality and magnitude of contraction. The sEMG feedback provides an immediate, precise, and concurrent source of information as a result of muscle effort. This increased awareness may improve recruitment effort and subsequent strength gains. When a patient is attempting to relearn a motor response to regain voluntary control over an inhibited muscle, sEMG can provide the additional information required and reaffirm that the correct motor response has occurred, enhancing motivation [7]. This type of feedback is critical in the acquisition of motor skills [38].
The reflexive AMI often present in the QF musculature prevents quadriceps activation and also likely reduces afferent activity arising from the muscle [37]. Motor learning and changes at the neural level have been postulated to occur faster with sEMG induced isometric exercise due to additional recruitment of motor units, causing a reorganization of facilitation patterns thus affecting afferent activity that contribute to AMI. Through sEMG feedback, afferent stimuli excite cerebral areas that normally receive proprioceptive information enabling the central nervous system to reestablish sensory motor loops and facilitate the firing and recruitment of new motor units thus influencing recruitment [5]. The sEMG-triggered NMES intervention also mimics the central activation ratio procedures where motor units that are not recruited during voluntary contraction are contracted with supramaximal electrical stimulation of the muscle fibers [37]. Exercising with sEMG-triggered NMES may produce a subsequent greater central activation ratio potentially disinhibiting AMI.
Surface EMG biofeedback techniques have been shown to be effective by facilitating voluntary initiated contractions in restoring both neural and muscular elements. Comparison of sEMG and NMES on knee extensor torque following Anterior cruciate ligament (ACL) reconstruction with volitional exercises revealed sEMG produced greater torque than NMES [39]. The authors concluded using the sEMG required participants to make greater work effort, recruit, and maintain muscle contraction while the NMES had only artificial assistance. An anticipatory motor response is initiated by the use of the sEMG, providing a more normal neuromuscular control feedback loop than by NMES alone. The addition of sEMG to NMES in this study augmented this physiological feed-forward system of motor control.
The results of this study contrast a previous study assessing postoperative isometric knee extensor peak torque using a portable sEMG-triggered NMES unit with similar settings [32]. Range of motion improved but torque production with portable sEMG-triggered NMES with exercise did not differ from exercise alone. Snyder-Mackler et al. [36] found patients following ACL reconstruction who trained with a clinical stimulator, such as the unit in our study design, had greater QF recovery, torque production, and functional recovery than those training with portable stimulators. The authors attributed this to the amount of sensory discomfort associated with the current and the amount of muscular soreness produced from portable stimulators. The high frequency of clinical stimulator driven Russian current, such as used in this study, allows more current to stimulate the nerve at a lower intensity due to decreased skin impedance and stimulates the deeper motor nerves while obstructing superficial sensory nerves.
Several authors have demonstrated that the electrical frequencies to elicit near maximal contractions are lower with concurrent voluntary contractions than with electrical stimulation alone [40,41]. The difference is attributed to voluntary asynchronous motor unit recruitment versus electrically induced synchronous motor unit recruitment. During voluntary contraction, the central nervous system can vary and modulate the motor units utilized to maintain a level of force. Electrically induced contractions do not have this capability and contribute to the increased fatigue associated with electrical stimulation. During normal voluntary muscle contractions, smaller motor units composed of primarily type I, fatigue-resistant muscle fibers are recruited before type II fibers. The order of recruitment begins with the most fatigue-resistant fibers at low level contractions and more fatigable motor units are recruited with stronger contractions [41]. During motor level electrical stimulation, large diameter fast twitch type II muscles fibers with low impedance and excitation thresholds are recruited first [17,42,43]. With the sEMG-triggered NMES intervention in this study, the initial sEMG induced volitional contraction may diminish the predominance of electrically induced Type II fiber recruitment and assist in producing a more normal neurophysiological biased maximal contraction.
Strength gains with electrical stimulation occur via overload produced by the stimulation combined with an increased recruitment of type II muscle fibers [44]. Type II fibers contract sooner due to the depolarization of large diameter nerves first but also fatigue first [42,43]. In this study, negligible decreases in torque over the course of the ten MVIC contractions were observed during both testing sessions while demonstrating an increase in torque for only contractions that elicited sEMG-triggered NMES (Figure 2). The increase in this study is in contrast to the findings of Vanderthommen et al. [37] who found no difference in QF torque between electrostimulated, active, and passive recovery strategies after fatiguing exercise. Results of this s tudy are also in contrast Hicks et al. [45] who found progressive decreases in strength over ten repeated MVIC’s. The authors attributed this to the susceptibility of type II fibers to fatigue and overwhelmed their greater capacity for potentiation after repeated MVIC’s. The addition of sEMG-triggered NMES to the QF in this study appears to resist progressive fatigue and sustain potentiation after repeated contractions. This pattern can be attributed to the sEMG that promotes feedback loops to elicit maximal volitional contractions that hinder electrically induced Type II fiber fatigue. This neuromuscular benefit can also be attributed to the Russian electrical stimulation and duty cycle parameters which allowed the muscle to recover between contractions as well as the current ramp setting that permits a natural increase in tension with minimized pain influence [46]. While this study’s protocol replicates clinical therapeutic exercise prescription, the procedures did not lead to exhaustion; therefore, the resultant fatigue effects with the sEMG to the NMES are limited to brief exercise bouts.
Time to Peak Torque
The application of sEMG-triggered NMES to the QF produced significantly faster recruitment time to peak torque versus all other volitional contractions that did not elicit sEMG-triggered NMES and the contractions of the MVIC-only test session. While NMES has independently proven to be effective for improving QF strength, the addition of sEMG to the NMES increases the recruitment time of the QF and can decrease the influence of AMI. Arthrogenic muscle inhibition of the QF results from the inhibitory process of the muscle being inactivated to prevent further injury despite injury to the muscle itself. One mechanism to limit AMI is to limit or modify the afferent input that is proposed to initiate and sustain AMI, specifically the change in afferent activity induced by pain and swelling [4]. The initial volitional contraction required with the sEMG-triggered NMES aids in maintaining normal neurophysiological homeostasis thereby limiting abnormal afferent receptor discharge to interneurons and the spinal cord that increase AMI. The quicker time to reach peak torque using sEMG-triggered NMES also potentially did not allow adequate time for the Golgi tendon organ to provide its inhibitory protection to override the developing musculotendinous unit tension due to the quickly triggered NMES and differential electrically induced type II fiber firing.
With injury and pathology, activation of normal motor units is altered by the disruption of the sensory feedback [47]. As motor unit activation is reduced, it is postulated that motor units with higher excitation thresholds may be differentially affected than more easily excited motor units. The authors suggest this trauma may selectively inhibit Type II muscle fibers as a result. Type II fiber atrophy has also been shown to be a result of injury and disuse atrophy [48]. The greatest decrease in muscle activity was seen in the rectus femoris, which contains the largest proportion of type II fibers in the QF muscle. The results of this study indicate sEMG-triggered NMES has the ability to recruit motor units faster than volitional contractions by influencing type II fiber latency while maintaining normal physiological contractions. The sEMG can increase sensory feedback and the NMES can disinhibit selective type II fibers thus increasing QF activation especially the rectus femoris.
Electrically induced reflex excitatory components have been shown to precede the inhibitory components in the cutaneous reflex [49]. With sEMG-triggered NMES, the short time duration to elicit the electrical stimulation may facilitate this excitatory cutaneous reflex component through noxious and tactile stimulation of the skin. The NMES also impedes the effects of QF reflex inhibition as electrical activation of the motor units augments the volitional utilization of the same units [17].
The results of this study support the hypothesis that sEMG-triggered NMES is more effective for improving isometric torque and time to peak torque versus voluntary contraction alone. To our knowledge, no other study has been conducted on the effectiveness of sEMG- triggered NMES intervention on torque of the QF muscle.
Limitations of this study include heterogeneous participant NMES pain tolerance and not quantifying the exact NMES dosing to the QF. The Russian NMES administered in our study was composed of a continuous and tetonic stimulation with a regular current intensity increase creating a visible and palpable muscle contraction in accordance with the programs often used and recommended in sports and rehabilitation to facilitate QF activation. Inaccurate location of the QF motor points may have influenced the findings although the methods are common clinical procedure. While the electrode location may not have provided optimal surface EMG readings, placements were limited by sites that require the dual capability of reading EMG signals as well as transduction of the NMES current. In addition, the potential for stimulus conduction to other adjacent muscle tissue via skin or other biologic tissue may confound the results readings and may influence muscle contractility. While time to peak torque measures rate of QF force development, the true rate between the two testing sessions was unable to analyzed to determine if it changed once the participant achieved their 90% MVIC to elicit the NMES. Additional limitations include instrumentation inability to collect exact sEMG data, electrode-skin contact, skin temperature, skin preparation, skin hydration, subcutaneous tissue thickness [50] and the inherent variability of sEMG. While this study warrants the use of sEMG-triggered NMES for increased muscle torque and recruitment, we can only generalize our results to this population but shows potential for conditions, injury, pathology, and postoperative applications that are impacted by weakness and delayed recruitment.
Conclusions
The results support the use of sEMG-triggered NMES to impact QF recruitment, supported by excellent power and large effect size (η2) for torque production and excellent power and small to medium effect sizes for time to peak torque [51]. Based on this study’s findings, it can be recommended to use sEMG-triggered NMES as an adjunct to increase torque and the time frame to attain peak torque while reducing fatigue. The combination of NMES to sEMG has significant summative benefits on torque and recruitment timing which warrants further investigation. Patient populations such as patellofemoral pathologies, osteoarthritis, and inflammatory conditions may benefit from sEMG-triggered NMES. The sEMG triggered NMES also has the potential to limit QF reflex inhibition caused by arthrogenic pain and edema especially in post surgical populations.
Recommendations for future study include comparing sEMG triggered NMES to sEMG and NMES independently and administering sEMG-triggered NMES to the pathological and postoperative population for neuromuscular reeducation and disuse atrophy. The long-term effectiveness of sEMG-triggered NMES should also be examined to determine its sustaining effect on muscular hypertrophy and other functional outcome measures.
Competing interest:
The author declare that they have no competing interest.
References
Palmieri, R., Ingersoll, C., Edwards, J., Hoffman, M., Stone, M., Babington, J., …Krause, B. (2003). Arthrogenic muscle inhibition is not present in the limb contralateral to a simulated knee joint effusion. Am J Phys Med Rehabil, 82:910-916.View
Snyder-Mackler, L., Delitto, A., Stralka, S., Bailey, S. (1994). Use of electrical stimulation to enhance recovery of quadriceps femoris muscle force production in patients following anterior cruciate ligament reconstruction. Phys Ther, 74(10):901-907.View
Suter, E., Herzog, W., De Souza, K., Bray, R. (1998). Inhibition of the quadriceps muscles in patients with anterior knee pain. J Appl Biomech, 14(4):360-373.View
Hopkins, J., & Ingersoll, C. (2000). Arthrogenic muscle inhibition: A limiting factor in joint rehabilitation. J Sport Rehabil, 9:135-159.View
Croce, R. (1986). The effects of EMG biofeedback on strength acquisition. Biofeedback Self Regul, 11(4):299-310.View
Lucca & Recchiuti, (1983). Effect of electromyographic biofeedback on an isometric strengthening program. Physical therapy, DOI:10.1093/PTJ/63.2.200.View
Draper, V. (1990). Electromyographic biofeedback and recovery of quadriceps femoris muscle function following anterior cruciate ligament reconstruction. Phys Ther, 70(1):11-17.View
Ingersoll, C., & Knight, K. (1991). Patellar location changes following EMG biofeedback or progressive resistive exercises. Med Sci Sports Exerc, 23(10):1122-1127.View
Levitt, R., Deisinger, J., Remondet Wall, J., Ford, L., Cassisi J. (1995). EMG feedback-assisted postoperative rehabilitation of minor arthroscopic knee surgeries. J Sports Med Phys Fitness, 35(3):218-223.View
Wise, H., Fiebert, I., Kates, J. (1984). EMG biofeedback as treatment for patellofemoral pain syndrome. J Orthop Sports Phys Ther, 6(2):95-103.View
Silkman, C., & McKeon, J. (2010). The Effectiveness of Electromyographic Biofeedback Supplementation During Knee Rehabilitation After Injury. J Sport Rehabil, 19,343-351.View
Wasielewski, N., Parker, T., Kotsko, K. (2011). Evaluation of Electromyographic Biofeedback for the Quadriceps Femoris: A Systematic Review J Athl Train, 46(5):543–554.View
Dursun, N., Dursun, E., Kilic, Z. (2001). Electromyographic biofeedback-controlled exercise versus conservative care for patellofemoral pain syndrome. Arch Phys Med Rehabil, 2001;82(12):1692-1695.View
Delitto, A., Rose, S., McKowen, J., Lehman, R., Thomas, J., Shively, R. (1988). Electrical stimulation versus voluntary exercise in strengthening thigh musculature after anterior cruciate ligament surgery. Phys Ther, 68(5):660-663.View
Lewek, M., Stevens, J., Snyder-Mackler, L. (2001). The Use of Electrical Stimulation to Increase Quadriceps Femoris Muscle Force in an Elderly Patient Following a Total Knee Arthroplasty. Phys Ther, 81(9):1565-1571.View
Selkowitz, D. (1985). Improvement in isometric strength of the quadriceps femoris muscle after training with electrical stimulation. Phys Ther, 65(2):186-196.View
Snyder-Mackler, L., Ladin, Z., Schepsis, A., Young, J. (1991). Electrical stimulation of the thigh muscles after reconstruction of the anterior cruciate ligament. Effects of electrically elicited contraction of the quadriceps femoris and hamstring muscles on gait and on strength of the thigh muscles. J Bone Joint Surg, 73(7):1025-1036.View
Snyder-Mackler, L., Delitto, A., Bailey, S., Stralka, S. (1995). Strength of the quadriceps femoris muscle and functional recovery after reconstruction of the anterior cruciate ligament. A prospective, randomized clinical trial of electrical stimulation. J Bone Joint Surg, 77(8):1166-1173.View
Soo, C., Currier, D., Threlkeld, A. (1988). Augmenting voluntary torque of healthy muscle by optimization of electrical stimulation. Phys Ther, 68(3):333-337.View
Kim, K., Croy, T., Hertel, J., Saliba, S. (2010). Effects of neuromuscular electrical stimulation after anterior cruciate ligament reconstruction on quadriceps strength, function, and patient-oriented outcomes: a systematic review. J Orthop Sports Phys Ther, 40(7):383-391.View
Logerstedt, D., Snyder-Mackler, L., Ritter, R., Axe, M. (2010a). Knee Pain and Mobility Impairments: Meniscal and Articular Cartilage Lesions. J Orthop Sports Phys Ther, 40(6):A1-A6.View
Logerstedt, D., Snyder-Mackler, L., Ritter, R., Axe, M., Godges, J., Altman, R, …Torburn L. (2010b). Knee Stability and Movement Coordination Impairments: Knee Ligament Sprain. J Orthop Sports Phys Ther, 40(4):A1-A37.View
Currier, D., Mann, R. (1983). Muscular strength development by electrical stimulation in healthy individuals. Phys Ther, 63(6):915-921.View
Paternostro-Sluga, T., Fialka, C., Alacamliogliu, Y., Saradeth, T., Fialka-Moser, V. (1999). Neuromuscular electrical stimulation after anterior cruciate ligament surgery. Clin Orthop Relat Res, 368:166-175.View
Sisk, T., Stralka, S., Deering, M., Griffin, J. (1987). Effect of electrical stimulation on quadriceps strength after reconstructive surgery of the anterior cruciate ligament. Am J Sports Med, 15(3):215-220.View
Bolton, D., Cauraugh, J., Hausenblas, H. (2004). Electromyogram-triggered neuromuscular stimulation and stroke motor recovery of arm/hand functions: a meta-analysis. Journal of the neurological sciences, 223(2):121-127.View
Cauraugh, J., & Kim, S. (2002). Two coupled motor recovery protocols are better than one: electromyogram-triggered neuromuscular stimulation and bilateral movements. Stroke, 33(6):1589-1594.View
Fields, R. (1987). Electromyographically triggered electric muscle stimulation for chronic hemiplegia. Arch Phys Med Rehabil, 68(7):407-414.View
Francisco, G., Chae, J., Chawla, H., Kirshblum, S., Zorowitz, R., Lewis, G., Pang, S. (1998). Electromyogram-triggered neuromuscular stimulation for improving the arm function of acute stroke survivors: a randomized pilot study. Arch Phys Med Rehabil, 79(5):570-575.View
Kraft, G., Fitts, S., Hammond, M. (1992). Techniques to improve function of the arm and hand in chronic hemiplegia. Arch Phys Med Rehabil, (3):220-227.View
Rakoš, M., Freudenschuβ, B., Girsch, W., Hofer, C., Kaus, J., Meiners, T., Paternostro, T., Mayr, W. (1999). Electromyogram Controlled Functional Electrical Stimulation for Treatment of the Paralyzed Upper Extremity. Artificial Organs, 23(5):466-469.View
Boucher, T., Wang, S., Trudelle-Jackson, E., Olson, S. (2009). Effectiveness of surface electromyographic biofeedback triggered neuromuscular electrical stimulation on knee rehabilitation. N Am J Sports Phys Ther, 4(3):100-109.View
Warfel, J. (1993). The extremities: muscles and motor points. 6th ed. Philadelphia: Lea & Febiger.
Kollmitzer, J., Ebenbichler, G., Kopf, A. (1999). Reliability of surface electromyographic measurements. Clin Neurophysiol, 110(4):725-734.View
Larsson, B., Karlsson, S., Eriksson, M., Gerdle, B. (2003). Test retest reliability of EMG and peak torque during repetitive maximum concentric knee extensions. J Electromyogr Kinesiol, 13(3):281-287.View
Snyder-Mackler L, De Luca ,P.F., Williams PR, Eastlack ME, Bartolozzi AR, (1994). 3. Reflex inhibition of the quadriceps femoris muscle after injury or reconstruction of the anterior cruciate ligament. The Journal of Bone And Joint Surgery. 76(4):555-560.View
Vanderthommen, M., Makrof, S., Demoulin, C. (2010). Comparison of active and electrostimulated recovery strategies after fatiguing exercise. J Sports Sci Med, 9, 164-169.View
Mulder, T., & Hulstijn, W. (1986). EMG feedback and the restoration of motor control: a controlled group study of 12 hemiparetic patients. Am J Sports Med, 65(4):173-188.View
Draper, V., & Ballard, L. (1991). Electrical stimulation versus electromyographic biofeedback in the recovery of quadriceps femoris muscle function following anterior cruciate ligament surgery. Phys Ther, 71(6):455-464.View
Binder-Macleod, S., & McDermond, L. (1992). Changes in the force-frequency relationship of the human quadriceps femoris muscle following electrically and voluntarily induced fatigue. Phys Ther, 72(2):95-104.View
Binder-Macleod, S., & Snyder-Mackler, L. (1993). Muscle fatigue: clinical implications for fatigue assessment and neuromuscular electrical stimulation. Phys Ther, 73(12):902-910.View
Sinacore, D., Delitto, A., King, D., Rose, S. (1990). Type II fiber activation with electrical stimulation: a preliminary report. Phys Ther, 70(7):416-422.View
Trimble, M., & Enoka R. (1991). Mechanisms underlying the training effects associated with neuromuscular electrical stimulation. Phys Ther, 71(4):273-280.View
Delitto, A., & Snyder-Mackler, L. (1990). Two theories of muscle strength augmentation using percutaneous electrical stimulation. Phys Ther, 1990;70(3):158-64.View
Hicks, A., Cupido, C., Martin, J., & Dent, J. (1991). Twitch potentiation during fatiguing exercise in the elderly: the effects of training. Eur J Appl Physiol, 63, 278–281.View
Balogun, J., Onilari, O., Akeju, O., Marzouk, D. (1993). High voltage electrical stimulation in the augmentation of muscle strength: Effects of pulse frequency. Arch Phys Med Rehabil, 74:910.View
Baratta,R., Solomonow, M., Zhou, B., Letson, D., Chuinard, R., D'Ambrosia, R. (1988). Muscular coactivation: The role of the antagonist musculature in maintaining knee stability. Am J Sports Med, 16(2):113-22.View
Elmqvist, L., Lorentzon, R., Johansson, C., Fugl-Meyer, A. (1988). Does a torn anterior cruciate ligament lead to change in the central nervous drive of the knee extensors? Eur J Appl Physiol Occup Physiol, 58(1-2):203-7View
Khan, S., & Burne, J. (2009). Afferents contributing to autogenic inhibition of gastrocnemius following electrical stimulation of its tendon. Brain Research, 1282:28-37.View
Park, J., & Ty Hopkins, J. (2011). Quadriceps activation normative values and the effect of subcutaneous tissue thickness. J Electromyogr Kinesiol, 21(1):136-140.View
Cohen, J. (1988). Statistical Power Analysis for the Behavioral Sciences. 2nd ed. Hillsdale, New Jersey: Lawrence Erlbaum Associates.View

