Journal of Rehabilitation Practices and Research Volume 4 (2022), Article ID: JRPR-142
Special Issue: Physical Therapy: From Acute Care to Rehabilitation and Wellness
https://doi.org/10.33790/jrpr1100142Special Issue
Change Over Time in the Structural Equation Model of Factors Related to Depression in Stroke Survivors Living at Home Since Discharge from the Acute Phase
Takashi Kimura1,2
1Department of Physical Therapy, ASO Rehabilitation College, Fukuoka, Japan.
2Department of Rehabilitation Medicine, Saga University Hospital, Saga, Japan.
Corresponding Author Details: Takashi Kimura, PhD, RPT, Department of Physical Therapy, ASO Rehabilitation College, Fukuoka, Japan.
Received date: 21th February, 2023
Accepted date: 10th March, 2023
Published date: 13th March, 2023
Citation: Kimura, T., (2023). Change Over Time in the Structural Equation Model of Factors Related to Depression in Stroke Survivors Living at Home Since Discharge from the Acute Phase. J Rehab Pract Res, 4(1):142.
Copyright: ©2023, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Purpose: To clarify the changes in causal factors related to depression over time among stroke survivors living at home since discharge using structural equation model analysis.
Methods: This was a longitudinal study using secondary data from the Stroke Recovery in Underserved Population database. This study used the Functional Independence Measure and the Center for Epidemiologic Studies Depression Scale to assess based on data collected at three time points: at discharge, 3 months after discharge, and 12 months after discharge. Structural equation modelling analysis was performed after conducting exploratory factorial and confirmatory factorial analysis.
Results: The results of factorial and structural equation modelling analysis showed that all factor loadings and model fits were acceptable. Upon comparing the three times points, the structural equation model related to depression changed. At discharge, depressive symptoms were mainly impacted by personal relationships; at three months after discharge, they were mainly impacted by physical symptoms; at 12 months after discharge, they were mainly impacted by living activities and cognitive function. Locomotion and three items related to depressive symptoms commonly appeared at all three time points.
Conclusion: The factors related to depression symptoms change over time post discharge. Therefore, this study suggests that an effective rehabilitation for ensuring that stroke survivors living at home can experience high quality of life should focus, at the physical level, on activities with gaps between performance and capability for, for example, locomotion and sphincter control; at the psychological level, on ensuring that stroke survivors have access to appropriate personal relationships without stress, and intervenors should consider survivors’ depressive symptoms at each relevant time point.
Key words: Depression; Stroke; FIM; CESD; Home dwelling
Introduction
Post-stroke depression (PSD) is a common symptom in people with stroke (stroke survivors). Moreover, depression is one of the many factors that contribute to suicide risk [1-4]; indeed, it has the highest suicide rate within one year from the onset of PSD [5,6]. In the addition, symptoms of PSD have a high incidence and are related to factors such as the unexpected development of stroke, the stress of cerebrovascular accident itself, and the reduction in the survivors’ quality of life (QOL) [7,8]. Age, gender, cultural background, and dysfunction after stroke are some of the many factors related to PSD [8,9]. Age, especially being 65 years old or over, has a stronger relationship with depression in stroke survivors compared to gender differences [10,11]. This age, of 65 years old or over, is in the range of age of stroke onset; additionally, the onset of PSD, which is frequently between two weeks and three months after stroke, overlaps with the stroke recovery period [12,13]. Furthermore, depression symptoms can be related to instrumental activities of daily living (IADL), that is the tasks required for independent living [14,15]. The goal of rehabilitation, in and of itself, can also contribute to the development of depression sometimes [16].
Compared with stroke survivors without PSD, those with PSD have a higher mortality rate and lower QOL [17]. They need more time to be independent compared to those without PSD [18,19]. In addition, PSD impacts the functions of recovery related to home living [20,21]. Stroke survivors have a high rate of returning to home living after discharge, living for a long-time after post stroke. Still, many stroke survivors experience the losses or changes of their previous roles in their homes, and this tends to contribute to PSD. Stroke is also associated with a high risk of falls at home, and reports show that survivors who experienced more than two falls had symptoms of depression [22-25].
As aforementioned, there is many of evidence showing that various factors contribute to the risk of developing PSD [17,18]. Thus, for stroke survivors to stay at home for a long-time and maintain a high QOL, it is important to consider PSD and how early preventative measures in stroke rehabilitation and continued treatment after discharge can be incorporated. This brings forth the need to investigate and consider social interaction and physical function in the home living environment. However, to our knowledge, few longitudinal studies have investigated causal relationships among the factors related to PSD in dwelling after discharge. This study, thus, aimed to clarify and comprehend the changing causal factors related to PSD among home-living stroke survivors using the structural equation model (SEM) analysis. The research also aimed to consider potential influential factors that can help maintain the QOL of home-living stroke survivors at high levels.
Participants and Methods
Participants and Data Collection
The Stroke Recovery in Underserved Populations 2005-2006 (SRUP) study collected data on patients with stroke that were treated at inpatient rehabilitation services, at medical facilities across the United States during 2005 and 2006 [28]. The present study is a secondary data from the SRUP. The data set underwent a confidentiality review and was altered when necessary to limit the risk of disclosure. Therefore, the need for informed consent was waived owing to a) the retrospective and observational design of the study and b) the use of secondary data. The SRUP's aimed to investigate the contribution of emotional and social factors to stroke recovery. An observational follow-up study with the cooperation of eleven medical facilities was performed to clarify the interaction of social and emotional factors with physical recovery after stroke. The data was collected at 3 time points: discharge, 3 months after discharge, and 12 months after discharge. Incomplete data (i.e., did not include information for all three time points) was excluded, resulting in data from 504 participants were included in the analyses. The data regarding the following factors was extracted as general information: age (55 or above over), sex, stroke diagnosis, stroke type, length of stay in hospital, number of comorbidities, number of stroke symptoms, home setting, and information regarding household members.
The Functional Independence Measure (FIM) was also used as an outcome and physical recovery measure and the Center for Epidemiologic Studies - Depression (CESD) scale to evaluate depressive symptoms [29]. The FIM consists of 13 motor function and 5 cognitive function items, and is widely used to evaluate the disability level of stroke survivors [30]. Scores range from 0 to 126, with a higher score indicating a higher recovery level. The CESD scale has 20 items measuring 4 positive and 16 negative emotions. Scores range from 0 to 60 and lower score indicating a lower level of depressive symptoms. The cut-off score in the CESD scale for depression symptoms is 16 or above.
Statistical Analysis
First, correlation analysis was performed to evaluate the relationship between datasets. The differences between data then collected at each of the three time points were assessed using the Friedman test to evaluate their characteristics and, if the results were significant, a Conover’s post-hoc test was conducted. This was followed by an exploratory factorial analysis (EFA) to select the appropriate number of minimum latent variables from the data. The EFA method was applied and maximum likelihood estimation and promax rotation were used based on the assumption that there were correlations among factors.
The EFA was screened using the Kaiser-Myer-Olkin (KMO) measure of sample adequacy and Bartlett’s test of sphericity for factorability. The exclusion condition on the factor loading was set at < 0.4. The number of factors that needed to be retained was based on the Kaiser rule and a scree plot. This was followed by a confirmatory factorial analysis (CFA) to evaluate the appropriate explanatory model fit of the extracted data from the results of the EFA. The additional fit measures were used comparative fit index (CFI; range 0 - 1, recommended values ≥ 0.95), goodness-of-fit index (GFI; range 0 - 1, recommended values ≥ 0.90), root-mean-square error of approximation (RMSEA; range 0 - 1, recommended values ≤ 0.06), and the standardized RMS residual (SRMR; range 0 - 1, recommended values ≤ 0.08) were used as additional fit measures [31,32]. A poor model fit is indicated when more than three indexes showed disagreement values.
Finally, structural equation modeling (SEM) analysis was preformed to assess causal relationships between latent variables based on the result of the CFA. The modification indices (mi) value was set over 10 as the modification condition of the analyzed SEM. The same measures used with the CFA method were used to assess the adequacy of the model fit. The statistical significance level was set at < 0.05. Statistical analyses were conducted using Jeffreys's Amazing Statistics Program (JASP, version 0.16.1) created by a group of researchers at the University of Amsterdam.
Results
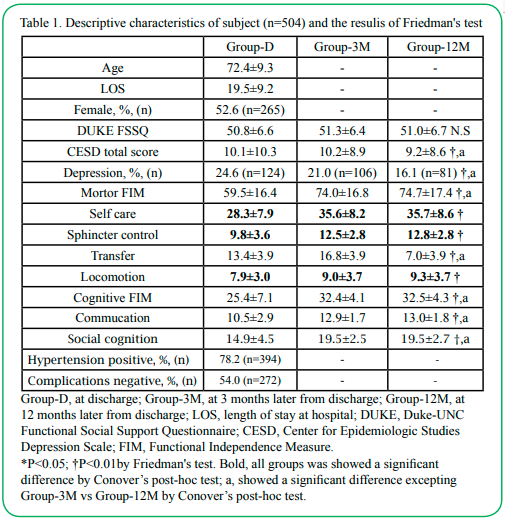
The study included data from 504 patients aged 55 years or more who were admitted to an inpatient medical rehabilitation facility and for whom there was complete information at each of the 3 times point (Group-D; at discharge, Group-3M; 3 months after discharge, Group-12M; 12 months after discharge). The gender division in the data was almost equal, with slightly more female participants (52%; Fig. 1). 75.3% of the participants identified their race as white, and 98.1% were living at home before at admission for stroke treatment. The most prevalent type of stroke was ischemic (74.6%), followed by hemorrhagic (16.5%), and hemiplegia (80%). The type of hemiplegia was same as both right and left (40%). Stroke patients with complications accounted for 56% of the group, and the number of complications were mainly two or three. The percentages of depression score measured to be above 16 in Group-D, Group-3M, and Group-12M were 24.6%, 21%, and 16.1%, respectively. At each of the three time points, the average of total CESD score was 9 to 10. Friedman's test found significant results for each item in multiple group comparison. However, Conover’s post-hoc test found that it was not significant in comparing between Group-3M and Group-12M were not significant, except for the three items of three items, self-care, sphincter control, and locomotion.
Exploratory Factorial Analysis and Confirmatory Factorial Analysis
The extracted latent variables were extracted at discharge were named “depressive symptoms” (DS), “personal relationships” (PR), and “living activities” (LA). Extracted latent variables at 3 months later from discharge were similar to the model of at discharge; however, physical symptoms (PS) were added. Extracted latent variables at 12 months were “interaction function” (IF) and LA.
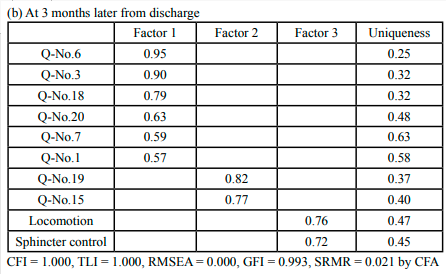
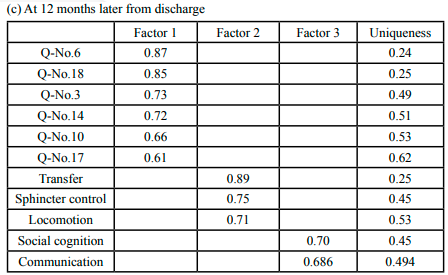
CFA was conducted to assess the validity of the latent variables and each item based on the results of EFA at three times point. The results of CFA based on each EFA showed that all the factor loadings and the model fits were acceptable: at time of discharge, CFI = 1.000, TLI = 1.000, RMSEA = 0.000, GFI = 0.993, SRMR = 0.021 (Table 2-a); at 3 months after discharge, CFI = 0.972, TLI = 0.957, RMSEA = 0.063, GFI = 0.994, SRMR = 0.033 (Table 2-b); at 12 months after discharge, CFI = 0.986, TLI = 0.977, RMSEA = 0.065, GFI = 0.999, SRMR = 0.036 (Table 2-c).
Structural Equation Modeling Analysis
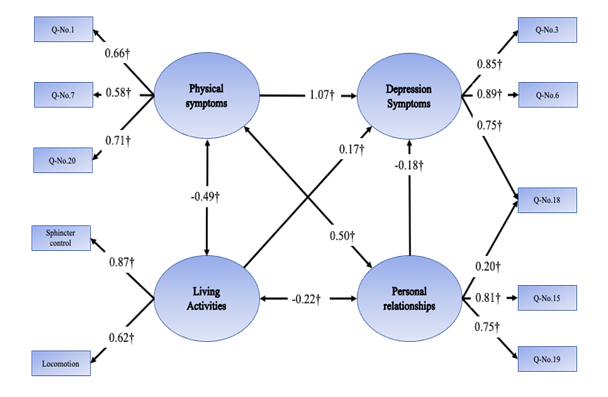
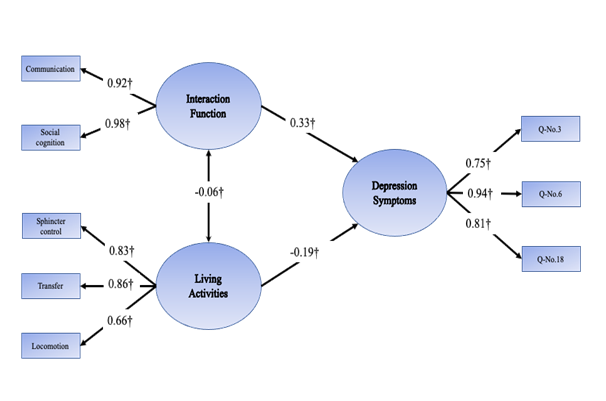
The model fit of SEM at discharge (SEM-D) had three latent factors; “depressive symptoms” (DS-D), “personal relationships” (PR-D), “living activities” (LA-D). The fit of the model was good, considering the following values: CFI = 1.000, TLI = 1.000, RMSEA = 0.000, GFI = 0.992, SRMR = 0.018 (Fig. 2-a). For the SEM at 3 months later (SEM-3), there were four latent factors: “depressive symptoms” (DS-3M), “personal relationships” (PR-3M), “living activities” (LA-3M), “physical symptoms” (PS-3M). The model fit of SEM-3 was as follows: CFI = 0.972, TLI = 0.957, RMSEA = 0.063, GFI = 0.944, SRMR = 0.033. For the SEM at 12 months later (SEM-12), there were three latent factors: “depressive symptoms” (DS-12M), “living activities” (LA-12M) “interaction function” (IF-12M). The model fit of SEM-12 was as follows: CFI = 0.986, TLI = 0.989, RMSEA = 0.065, GFI = 0.999, SRMR = 0.036.
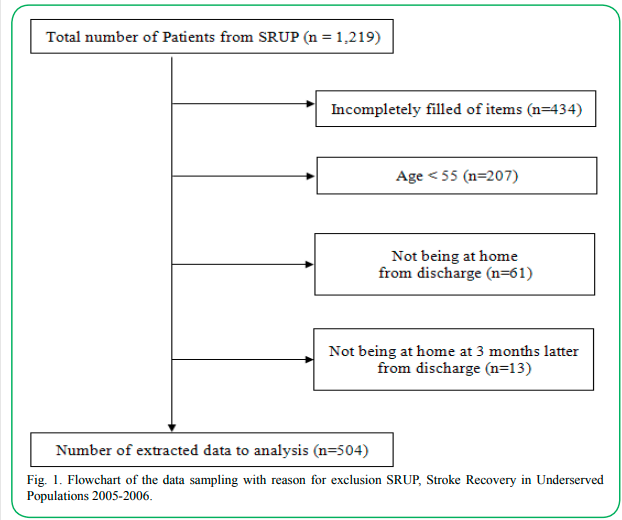
Fig 1. Flowchart of the data sampling with reason for exclusion SRUP, Stroke Recovery in Underserved Populations 2005-2006.
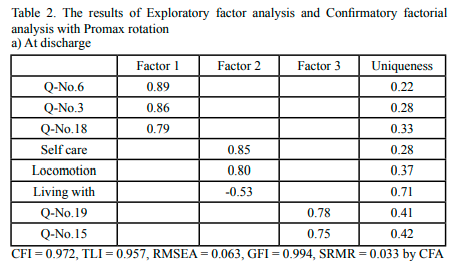
Table 2.(a) The results of Exploratory factor analysis and Confirmatory factorial analysis with Promax rotation
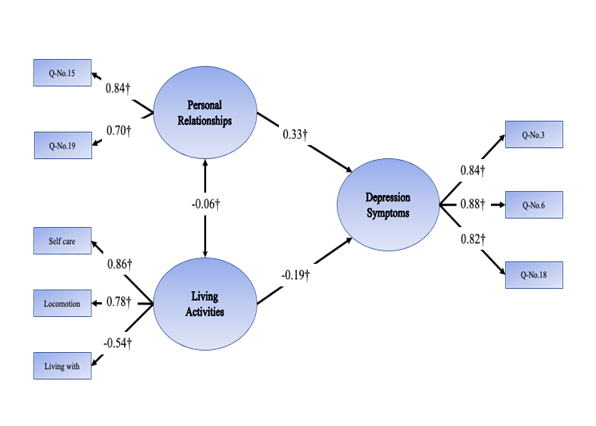
Figure 2. The structural model of factors related to depression at each time point (standardized path coefficients) †P<0.01. Q-No.1, I was bothered by things that usually don’t bother me; Q-No.3, I felt that I could not shake off the blues even with help from my family & friends; Q-No.6, I felt depressed; Q-No.7, I felt that everything I did was an effort; Q-No.15, People were unfriendly; Q-No.17, I had crying spells; Q-No.18, I felt sad; Q-No.19, I felt that people disliked me; Q-No.20, I could not get going.
(a) At Discharge
Nonetheless, another SEM indicated the possibility to improve the adequacy of the model fit, except for SEM-D. Particularly, the item “I felt sad” was related with PR-3M, and then SEM-3M was recalculated; the results are as follows: CFI = 0.995, TLI = 0.992, RMSEA = 0.027, GFI = 0.998, SRMR = 0.021. These results led for the previous SEM-3M to be replaced by the recalculated model (Fig. 2-b).
SEM-12M was recalculated after it made a correlation between the factors of transfer and locomotion; the results for the SEM-12M were slightly improved by this procedure, giving the following values: CFI = 0.993, TLI = 0.989, RMSEA = 0.046, GFI = 0.999, SRMR = 0.025. Therefore, the recalculated SEM-12M was applied (Fig. 2-c)
Discussion
This study aimed to clarify the causal factors related to PSD in stroke survivors using SEM analysis at three time points (at discharge, 3 months after discharge, and 12 months after discharge). These time points were compared to investigate how the SEM changed overtime. Three central findings emerged. First, the LA factor, which comprised three items (sphincter control, transfer, and locomotion), impacted the DS factor at all three time points. Second, SEM in this study changed over time. At discharge, “personal relationships” were an important factor; 3 months later, “physical symptoms” were influential,”; and 12 months later, “living activities” and “cognitive function” were important. Finally, the sub-items of the DS factor were the same across all time points; that is, “I felt that I could not shake off the blues even with the help of my family and friends,” “I felt depressed,” and “I felt sad”.
Sub-items of the DS factor that were included in the SEM analysis were relevant to depression, and the same three sub-items were related to the DS factor at all time points, although the CESD scale has 14 negative items. Moreover, these items had a high relevance. Thus, the extracted three items are likely important for PSD prevention, and intervening in them (i.e., “I felt that I could not shake off the blues even with the help of my family and friends,” “I felt depressed,” and “I felt sad,”) may be one of the strategy to prevent from PSD.
The SEM-D comprised three factors: PR-D, LA-D and DS-D. Studies suggest that the human and environmental resources (e.g., caregiving capacity and the home environment) in the home setting, which are insufficient when compared with the corresponding resources provided in the hospital [33], can make it difficult for people with stroke to live at home. Thus, stroke patients make feeling of loss and compromises because of these limitations. Furthermore, caregivers often experience both physical and mental fatigue because of the long-term care that stroke survivors require [34,35]. Stroke survivors may also often face greater difficulties to perform activities of daily living at home compared with at the hospital, and they may feel a sense of embarrassment because of this. Family caregivers may also lack the necessary knowledge, skills, or experience to offer relevant support. Therefore, both stroke survivors and their caregivers/families often feel as if they are burdening each other, and they may need to restructure their lifestyle and home environment after the patient is discharged. Consequently, to maintain stroke survivors living at home, it may be important to consider what stroke survivors feel ever since the onset of the period after discharge and their staying at home; "People were unfriendly" and "I felt that people disliked me" in PR-D factor, from the early phases of staying at home post discharge.
Additionally, the level of FIM scores in SEM-D was lower compared with other time points. Locomotion, which appears in the LA-3D factor, was an activity that had a gap between performance and capability in ADL [36,37]. Moreover, the LA-D factor showed a negative relation to living with other household members. At the point of discharge, stroke survivors’ functional level tends to be low and they tend to depend on family caregivers. Thus, the LA-D factor might be lower if stroke survivors have too-great a level of dependence on their caregivers. Stroke survivors with a lower FIM require extensive caregiving, which can affect a stroke survivor’s sense of personal dignity and self-esteem. Then, in an attempt to avoid negative emotions, stroke survivors may find it difficult to ask for the care they need. Additionally, stroke survivors may experience different forms of psychological pressure (e.g., a sense that one keeps making troubles for or placing high loads on caregivers) within the context of their long-term caregiving relationships, which could then lead to the development of inappropriate personal relationships. As the LA-D factor impacted both the DS-D and PR-D factors, it is suggested that these other two factors are preventable or relievable by interventions (e.g., by avoiding excessive care given to survivors and encouraging their independence) focused on the LA-D factor at the onset of survivors’ lives at their homes after discharge.
The SEM-3M had four factors (PS-3M, LA-3M, PR-3M and DS-3M), with the PS-3M and LA-3M factors having had a positive impact and the PR-3M factor a negative impact. This suggests that stroke survivors developed PSD symptoms as their living activities and personal relationships were improving and as physical symptoms could be observed. Moreover, responses to “I felt sad” were related to the PR-3M factor. From the point of view of physical function, survivors had improved their status at three months after discharge compared with that at discharge. This finding of this study is similar to that in prior studies, as stroke recovery improves during this period after discharge [38-40]. Thus, the results of the score of each FIM item in SEM-3M in this study suggested moving toward recovery when compared with the score in SEM-D. Normally, improvements in FIM scores would mean a positive impact in stroke survivors’ psychological health. However, as aforementioned, improvements in the LA-3M factor in this particular case made DS-3M scores go up, thus negatively impacting the DS-3M factor. Sphincter control, as a sub-item in the LA-3M factor, is an activity with gaps similar to those for locomotion, and could be considered as a highly personal and private issue. Moreover, sphincter control was higher relation than locomotion to LA-3M factor, and it was considered to be a serious problem for stroke survivors. When dealing with sphincter control issues, which is a common problem among them after their hospital discharge and may even require continued care despite the overall recovery, stroke survivors may experience a sense of embarrassment and mental distress. Stroke survivors realize that ongoing issues with sphincter control can greatly increase caregiver burden and consequently, they blame themselves, which negatively affects their self-esteem [35]. This makes stroke survivors realize that their situation will remain unchanged, which increases their stress, though LA-3M factor improved.
In general, it is believed that improvements in personal relationships positively impacts people’s psychological health. However, as with the LA-3M factor, improvements in the PR-3M factor negatively impacted the DS-3M factor. In the PR-3M factor, the sub-item “I felt sad” was included, something that did not occur in the SEM-D. Researchers have described that the role of sadness is primarily to promote self-protection and avoid unpleasant experiences [41]. The PR-3M factor was related to the PS-3M factor, suggesting that survivors experienced some level of stress or sadness since discharge, causing them to have reduced motivation and leading to a negative psychological status. Thus, stroke survivors might have experienced an annoying physical symptom related to the PR-3M factors, which means that improving the PR-3M factor provides some sort of load to the minds of stroke survivors, indicating that improvements in the PR-3M factor causes their stress. In response to these findings, it would be important to intervene in activities that have gaps between performance and capability, as well as make arrangements to try and ensure that stroke survivors have access to appropriate personal relationships, so that they do not experience much stress as of 3 months after discharge.
Three factors emerged in SEM-12M: LA-12M, IF-12M and DS-12M factor. Compared with at 3 months later, at 12 months after discharge, it was assumed that stroke survivors and their caregivers had gained a better understanding of the care needed and had developed better relationships. Thus, it was assumed that PR-12M would not emerge as central in SEM-12M. Functional recovery was mostly unchanged compared with at three months after discharge. Stroke recovery tends to improve sharply until three months from stroke onset, and only moderately thereafter [38-40].
The results of the current study correspond to these prior assertions, as stroke survivors in this study seemingly fully realized the progress of recovery until six months after discharge; then, the progress became slower and it was assumed that they could not realize much. The results of the current study correspond to these prior assertions, as stroke survivors in this study seemingly fully realized the progress of recovery until six months after discharge; then, the progress became slower and it was assumed that they could not realize much. This slowdown could make patients less hopeful about further recovery and lead to negative thought patterns. Furthermore, compared with healthy older adults, stroke survivors may find it harder to remain active in their community and experience more limitations in their social interactions [42-44]. Thus, in general, stroke survivors’ social cognition score has been found to be lower. Moreover, it has been reported that stroke survivors who live in their community for more than one year after discharge from an acute ward experience limitations in their social interactions [45-47]. Social interaction is related to QOL and poor QOL can lead to depression symptoms. Actually, prior researchers have described that providing support for people to engage in social interaction is a form of depression prevention [48]. Other scholars further demonstrated that limited communication leads stroke survivors to experience a reduced self-esteem [49], and limited activities could cause QOL reductions among stroke patients [50,51]. Together, the findings of this study and past research suggest that it could be necessary to consider approaches to improving the motor and interaction functions of stroke survivors as of 12 months after discharge.
This study had some limitations. First, this study could not reflect the influences of tertiary items on depression symptoms, as it was based on a secondary item in the FIM. Moreover, the impact of hobbies could not be considered for the SEM because the start time and continuation of hobbies are both unclear. However, hobby activities may impact QOL and interventions on individual self-care could contribute to stroke survivors to continue living at their homes. Therefore, future research should investigate the modeling between depression and these factors.
Conclusion
This study compared three time points in stroke recovery using SEM analysis and clarified the changing relationships between causal factors and depressive symptoms for stroke patients living at home. When comparing the three time points, the causal factors related to depressive symptoms changed overtime. Personal relationships weremainly impacted upon discharge. While physical symptoms were influential 3 months later, living activities and cognitive function were important at 12 months later. Locomotion and three items related to the DS factor were found to be common at all three time points. Therefore, this study suggests that an effective rehabilitation should focus, at the physical level, on activities with gaps between performance and capacity ability, like locomotion and sphincter control ; at the psychological level, on ensuring that stroke survivors have access to appropriate personal relationships without stress, and should consider stroke survivors’ depressive symptoms at each relevant time point. Further research should comprehensively consider the association between depression, hobby activities, and individual self-care.
Acknowledgments
I would like to deeply thank Dr. Glenn V. Ostir and Inter-university Consortium for Political and Social Research for their approval of the use of the data sets that were analyzed in this study.
Conflict of interest:
The author declare that they have no competing interest.
References
Pompili, M., Venturini, P., Lamis, D. A., Giordano, G., Serafini, G., Belvederi Murri, M., Amore, M. (2015). Suicide in stroke survivors: epidemiology and prevention. Drugs & aging, 32(1), 21–29. https://doi.org/10.1007/s40266-014-0233-xView
Medeiros, G. C., Roy, D., Kontos, N., Beach, S. R. (2020). Post-stroke depression: A 2020 updated review. General hospital psychiatry, 66, 70–80. https://doi.org/10.1016/j. genhosppsych.2020.06.011View
Towfighi, A., Ovbiagele, B., El Husseini, N., Hackett, M. L., Jorge, R. E., Kissela, B. M., Mitchell, P. H. (2017). Poststroke Depression: A Scientific Statement for Healthcare Professionals From the American Heart Association/American Stroke Association. Stroke, 48(2), e30–e43. https://doi.org/10.1161/ STR.0000000000000113View
Starkstein, S. E., Mizrahi, R., Power, B. D. (2008). Antidepressant therapy in post-stroke depression. Expert opinion on pharmacotherapy, 9(8), 1291–1298. https://doi. org/10.1517/14656566.9.8.1291View
Chun, H. Y., Ford, A., Kutlubaev, M. A., Almeida, O. P., Mead, G. E. (2022). Depression, Anxiety, and Suicide After Stroke: A Narrative Review of the Best Available Evidence. Stroke, 53(4), 1402–1410. https://doi.org/10.1161/ STROKEAHA.121.035499.View
Choi, H. L., Yang, K., Han, K., Kim, B., Chang, W. H., Kwon, S., Jung, W. (2023). Increased Risk of Developing Depression in Disability after Stroke: A Korean Nationwide Study. International journal of environmental research and public health, 20(1), 842. https://doi.org/10.3390/ijerph20010842View
Zhao, F. Y., Yue, Y. Y., Li, L., Lang, S. Y., Wang, M. W., Du, X. D., Deng, Y. L. (2018). Clinical practice guidelines for post-stroke depression in China. Revista brasileira de psiquiatria (Sao Paulo, Brazil : 1999), 40(3), 325–334. https://doi. org/10.1590/1516-4446-2017-2343View
Verdelho, A., Hénon, H., Lebert, F., Pasquier, F., Leys, D. (2004). Depressive symptoms after stroke and relationship with dementia: A three-year follow-up study. Neurology, 62(6), 905– 911. https://doi.org/10.1212/01.wnl.0000115107.66957.8cView
Franco-Urbano, M. S., Rodríguez-Martínez, M. D. C., & García-Pérez, P. (2022). The Impact of Depression on the Functional Outcome of the Elderly Stroke Victim from a Gender Perspective: A Systematic Review. Healthcare (Basel, Switzerland), 10(10), 2110. https://doi.org/10.3390/healthcare10102110View
Almeida O. P. (2014). Prevention of depression in older age. Maturitas, 79(2), 136–141. https://doi.org/10.1016/j. maturitas.2014.03.005View
Salk, R. H., Hyde, J. S., Abramson, L. Y. (2017). Gender differences in depression in representative national samples: Meta-analyses of diagnoses and symptoms. Psychological bulletin, 143(8), 783–822. https://doi.org/10.1037/bul0000102View
Douven, E., Köhler, S., Rodriguez, M. M. F., Staals, J., Verhey, F. R. J., Aalten, P. (2017). Imaging Markers of Post-Stroke Depression and Apathy: a Systematic Review and Meta-Analysis. Neuropsychology review, 27(3), 202–219. https://doi. org/10.1007/s11065-017-9356-2View
Yang, L., Zhang, Z., Sun, D., Xu, Z., Zhang, X., Li, L. (2010). The serum interleukin-18 is a potential marker for development of post-stroke depression. Neurological research, 32(4), 340– 346. https://doi.org/10.1179/016164110X12656393665080View
Kiosses, D. N., & Alexopoulos, G. S. (2005). IADL functions, cognitive deficits, and severity of depression: a preliminary study. The American journal of geriatric psychiatry : official journal of the American Association for Geriatric Psychiatry, 13(3), 244–249. https://doi.org/10.1176/appi.ajgp.13.3.244View
Ghaffari, A., Rostami, H. R., Akbarfahimi, M. (2021). Predictors of Instrumental Activities of Daily Living Performance in Patients with Stroke. Occupational therapy international, 2021, 6675680. https://doi.org/10.1155/2021/6675680View
Zhang, W. N., Pan, Y. H., Wang, X. Y., Zhao, Y. (2013). A prospective study of the incidence and correlated factors of post-stroke depression in China. PloS one, 8(11), e78981. https://doi. org/10.1371/journal.pone.0078981View
Ezema, C. I., Akusoba, P. C., Nweke, M. C., Uchewoke, C. U., Agono, J., Usoro, G. (2019). Influence of Post-Stroke Depression on Functional Independence in Activities of Daily Living. Ethiopian journal of health sciences, 29(1), 841–846. https://doi.org/10.4314/ejhs.v29i1.5View
Ghaffari, A., Asadi, B., Zareian, A., Akbarfahimi, M., Raissi, G. R., Fathali Lavasani, F. (2022). The Effects of Vestibular Rehabilitation on Poststroke Fatigue: A Randomized Controlled Trial Study. Stroke research and treatment, 2022, 3155437. https://doi.org/10.1155/2022/3155437View
Zhang, Y., Xiong, Y., Yu, Q., Shen, S., Chen, L., Lei, X. (2021). The activity of daily living (ADL) subgroups and health impairment among Chinese elderly: a latent profile analysis. BMC geriatrics, 21(1), 30. https://doi.org/10.1186/s12877-020- 01986-xView
Allida, S., Cox, K. L., Hsieh, C. F., House, A., Hackett, M. L. (2020). Pharmacological, psychological and non-invasive brain stimulation interventions for preventing depression after stroke. The Cochrane database of systematic reviews, 5(5), CD003689. https://doi.org/10.1002/14651858.CD003689.pub4View
Gilsanz, P., Kubzansky, L. D., Tchetgen Tchetgen, E. J., Wang, Q., Kawachi, I., Patton, K. K., Fitzpatrick, A. L. (2017). Changes in Depressive Symptoms and Subsequent Risk of Stroke in the Cardiovascular Health Study. Stroke, 48(1), 43–48. https://doi. org/10.1161/STROKEAHA.116.013554View
Forster, A., & Young, J. (1995). Incidence and consequences of falls due to stroke: a systematic inquiry. BMJ (Clinical research ed.), 311(6997), 83–86. https://doi.org/10.1136/ bmj.311.6997.83View
Demakakos, P., Cooper, R., Hamer, M., de Oliveira, C., Hardy, R., & Breeze, E. (2013). The bidirectional association between depressive symptoms and gait speed: evidence from the English Longitudinal Study of Ageing (ELSA). PloS one, 8(7), e68632. https://doi.org/10.1371/journal.pone.0068632View
Choi, N. G., Zhou, Y., Marti, C. N., Kunik, M. E. (2022). Associations Between Changes in Depression/Anxiety Symptoms and Fall Worry Among Community-Dwelling Older Adults. Journal of applied gerontology : the official journal of the Southern Gerontological Society, 41(12), 2520–2531. https://doi.org/10.1177/07334648221119464View
Gambaro, E., Gramaglia, C., Azzolina, D., Campani, D., Molin, A. D., Zeppegno, P. (2022). The complex associations between late life depression, fear of falling and risk of falls. A systematic review and meta-analysis. Ageing research reviews, 73, 101532. https://doi.org/10.1016/j.arr.2021.101532View
Medeiros, G. C., Roy, D., Kontos, N., Beach, S. R. (2020). Post-stroke depression: A 2020 updated review. General hospital psychiatry, 66, 70–80. https://doi.org/10.1016/j. genhosppsych.2020.06.011View
Pearce, M., Garcia, L., Abbas, A., Strain, T., Schuch, F. B., Golubic, R., Kelly, P. (2022). Association Between Physical Activity and Risk of Depression: A Systematic Review and Meta-analysis. JAMA psychiatry, 79(6), 550–559. https://doi. org/10.1001/jamapsychiatry.2022.0609View
Ostir, Glenn, Ottenbacher, Kenneth, and Kuo, Yong Fang. Stroke Recovery in Underserved Populations 2005-2006 [United States]. Inter-university Consortium for Political and Social Research [distributor], 2016-05-03. https://doi.org/10.3886/ ICPSR36422.v1View
Pan, C., Li, G., Sun, W., Miao, J., Qiu, X., Lan, Y., Wang, Y. (2022). Neural Substrates of Poststroke Depression: Current Opinions and Methodology Trends. Frontiers in neuroscience, 16, 812410. https://doi.org/10.3389/fnins.2022.812410View
Bravo, G., Richards, C. L., Corriveau, H., Trottier, L. (2018). Converting Functional Autonomy Measurement System Scores of Patients Post-Stroke to FIM Scores. Physiotherapy Canada. Physiotherapie Canada, 70(4), 349–355. https://doi. org/10.3138/ptc.2017-82View
Ek, A., Sorjonen, K., Eli, K., Lindberg, L., Nyman, J., Marcus, C., Nowicka, P. (2016). Associations between Parental Concerns about Preschoolers' Weight and Eating and Parental Feeding Practices: Results from Analyses of the Child Eating Behavior Questionnaire, the Child Feeding Questionnaire, and the Lifestyle Behavior Checklist. PloS one, 11(1), e0147257. https://doi.org/10.1371/journal.pone.0147257View
Steiger, J. H. (2007). Understanding the limitations of global fit assessment in structural equation modeling. Personality and Individual Differences, 42(5), 893–898. https://doi. org/10.1016/j.paid.2006.09.017View
Chevalley, O., Truijen, S., Saeys, W., Opsommer, E. (2022). Socio-environmental predictive factors for discharge destination after inpatient rehabilitation in patients with stroke: a systematic review and meta-analysis. Disability and rehabilitation, 44(18), 4974–4985. https://doi.org/10.1080/09638288.2021.1923838View
Jaracz, K., Grabowska-Fudala, B., Górna, K., Jaracz, J., Moczko, J., Kozubski, W. (2015). Burden in caregivers of long-term stroke survivors: Prevalence and determinants at 6 months and 5 years after stroke. Patient education and counseling, 98(8), 1011–1016. https://doi.org/10.1016/j.pec.2015.04.008View
Park, E. Y., & Kim, J. H. (2021). Interaction of Socio-demographic Characteristics on Acceptance of Disability Among Individuals With Physical Disabilities. Frontiers in psychiatry, 12, 597817. https://doi.org/10.3389/fpsyt.2021.597817View
Iwai, N., Aoyagi, Y., Tokuhisa, K., Yamamoto, J., Shimada, T. (2011). The gaps between capability ADL and performance ADL of stroke patients in a convalescent rehabilitation ward -based on the functional independence measure-. Journal of Physical Therapy Science, 23(2), 333-338. https://doi.org/10.1589/ jpts.23.333View
Nakanishi, K., Yamaga, T., Ikeya, M. (2022). Gaps between Activities of Daily Living Performance and Capacity in People with Mild Dementia. International journal of environmental research and public health, 19(23), 15949. https://doi. org/10.3390/ijerph192315949View
Swayne, O. B., Rothwell, J. C., Ward, N. S., Greenwood, R. J. (2008). Stages of motor output reorganization after hemispheric stroke suggested by longitudinal studies of cortical physiology. Cerebral cortex (New York, N.Y. : 1991), 18(8), 1909–1922. https://doi.org/10.1093/cercor/bhm218View
Uzair, M., Arshad, M., Abualait, T., Al-Harbi, Z. T., Al-Harbi, T. M., Fahad, R. B., Arshad, A. (2022). Neurobiology of amphetamine use in stroke recovery combined with rehabilitative training and brain stimulation. CNS & neurological disorders drug targets, 10.2174/1871527321666220512155811. Advance online publication. https://doi.org/10.2174/1871527321666220 512155811View
Lang, C. E., Waddell, K. J., Barth, J., Holleran, C. L., Strube, M. J., Bland, M. D. (2021). Upper Limb Performance in Daily Life Approaches Plateau Around Three to Six Weeks Post-stroke. Neurorehabilitation and neural repair, 35(10), 903–914. https:// doi.org/10.1177/15459683211041302View
Albert, J., Lopez-Martin, S., and Carretie, L. (2010). Emotional context modulates response inhibition: neural and behavioral data. Neuroimage 49, 914–921. doi: 10.1016/j. neuroimage.2009.08.045View
Moore, S. A., Hallsworth, K., Plötz, T., Ford, G. A., Rochester, L., Trenell, M. I. (2013). Physical activity, sedentary behaviour and metabolic control following stroke: a cross-sectional and longitudinal study. PloS one, 8(1), e55263. https://doi. org/10.1371/journal.pone.0055263View
Nijsse, B., Spikman, J. M., Visser-Meily, J. M., de Kort, P. L., van Heugten, C. M. (2019). Social Cognition Impairments in the Long Term Post Stroke. Archives of physical medicine and rehabilitation, 100(7), 1300–1307. https://doi.org/10.1016/j. apmr.2019.01.023View
Nijsse, B., Spikman, J. M., Visser-Meily, J. M. A., de Kort, P. L. M., van Heugten, C. M. (2019). Social cognition impairments are associated with behavioural changes in the long term after stroke. PloS one, 14(3), e0213725. https://doi.org/10.1371/ journal.pone.0213725View
Mudzi, W., Stewart, A., Musenge, E. (2013). Community participation of patients 12 months post-stroke in Johannesburg, South Africa. African Journal of Primary Health Care & Family Medicine, 5(1), 426. https://doi.org/10.4102/phcfm.v5i1.426View
Mountain, A., Patrice Lindsay, M., Teasell, R., Salbach, N. M., de Jong, A., Foley, N., Bhogal, S. (2020). Canadian Stroke Best Practice Recommendations: Rehabilitation, Recovery, and Community Participation following Stroke. Part Two: Transitions and Community Participation Following Stroke. International journal of stroke : official journal of the International Stroke Society, 15(7), 789–806. https://doi. org/10.1177/1747493019897847View
Yoon, J. A., Park, S. G., Roh, H. L. (2015). Comparisons of social interaction and activities of daily living between long-term care facility and community-dwelling stroke patients. Journal of physical therapy science, 27(10), 3127–3131. https:// doi.org/10.1589/jpts.27.3127View
Lewin, A., Jöbges, M., Werheid, K. (2013). The influence of self-efficacy, pre-stroke depression and perceived social support on self-reported depressive symptoms during stroke rehabilitation. Neuropsychological rehabilitation, 23(4), 546–562. https://doi. org/10.1080/09602011.2013.794742View
Tough, H., Brinkhof, M. W. G., Fekete, C. (2022). Untangling the role of social relationships in the association between caregiver burden and caregiver health: an observational study exploring three coping models of the stress process paradigm. BMC public health, 22(1), 1737. https://doi.org/10.1186/ s12889-022-14127-3View
Ryszewska-Łabędzka, D., Tobis, S., Kropińska, S., Wieczorowska-Tobis, K., Talarska, D. (2022). The Association of Self-Esteem with the Level of Independent Functioning and the Primary Demographic Factors in Persons over 60 Years of Age. International journal of environmental research and public health, 19(4), 1996. https://doi.org/10.3390/ijerph19041996View
Fulk, G. D., He, Y., Boyne, P., Dunning, K. (2017). Predicting Home and Community Walking Activity Poststroke. Stroke, 48(2), 406–411. https://doi.org/10.1161/ STROKEAHA.116.015309View