Journal of CAM Research Progress Volume 1 (2022), Article ID: JCRP-102
https://doi.org/10.33790/jcrp1100102Research Article
Network Pharmacology-Based Validation of Traditional Therapeutic Claim of Momordica Charantiain Alleviating Diabetic Nephropathy
Gaurav Gautam
Senior Researcher, Department of Pharmacognosy and Phytochemistry, School of Pharmaceutical Education and Research, JamiaHamdard, New Delhi, India.
Corresponding Author Details: Gaurav Gautam, Senior Researcher, Department of Pharmacognosy and Phytochemistry, School of Pharmaceutical Education and Research, JamiaHamdard, New Delhi, India. E-mail: evamam03@ucm.es
Received date: 14th January, 2022
Accepted date: 13th April, 2022
Published date: 16th April, 2022
Citation: Gautam, G. (2022). Network Pharmacology-Basedvalidation of Traditional Therapeutic Claim of Momordica Charantiain Alleviating Diabetic Nephropathy. J CAM Research Progress, 1(1): 102.
Copyright: ©2022, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited. Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
For centuries, medicinal plants have been playing an important role in the alleviation of various diseases, traditionally. Momordica charantia L. (M. charantia) is a folk medicinal herb belong to the Cucurbitaceae family, used as the folk medicinal regime for the treatment of diabetes or diabetic nephropathy (DN), traditionally. Due to the lack of scientific evidence based on its molecular mechanism for treating DN, the study is aimed to investigate the molecular mechanism of M. charantia metabolites using a network pharmacology approach. Furthermore, ADME analysis was performed to determine the lipophilicity and the drug-likeness response of the metabolites. The network pharmacology results showed a multi-mechanistic and therapeutic role of the metabolites present M. charantia by regulating several genomes involved in the pathophysiology of DN. Mean while, M. charantia ameliorates endothelial dysfunction, fatty liver disease, diabetes mellitus, acute kidney injury, fibrosis, hypertensive disease, obesity, etc. furthermore, it was also found that the targets potentially play an essential role in the regulation of oxidative stress, inflammation, and oxidative stress-induced inflammation. In ADME analysis, each selected molecule of M. charantia exhibited good gastrointestinal (GI) absorption, lipophilicity and bioavailability response. Hence, it can be demonstrated that M. charantiapossesses several metabolites including polyphenols which exhibit an important role in the treatment of DN via regulation of several genomes such as AKTs, CASPs, MAPKs, ILs, NOs, etc, responsible for its pathophysiology. Furthermore, the generated evidence validates the traditional claim of M. charantia for alleviating DN.
Keywords: Momordica charantiaL., Polyphenols, Network pharmacology, ADME analysis
Introduction
For centuries, medicinal plants have been playing an important role in the alleviation of various diseases, traditionally, because of their complexity in phytochemicals and multi-mechanistic and therapeutic effects. However, from the last decades, exponential growth has been seen in the utilization of medicinal plants and their derived products due to their least side effects, easy availability, and economic thus maintaining sustainable development inthe healthcare system and the economy of the country [1,2]. Furthermore, medicinal plants are acknowledged as the main source for new drug discovery and development as more than 50 percent of the existing drugs are derived from natural resources. Taking a look into their uncountable use in the healthcare system, quality, safety, efficacy-based assessment, and generating scientific evidence includes a big contribution for their regulatory aspects [3,4]. Based on the pharmacological perspective, it is not easy to determine the exact principle of phytochemicals that are being exhibited in the therapeutic response for the treatment of targeted or un-targeted body ailments. Bioassay-guided fractions or pharmacological assessment on individual phytochemicals from the targeted plant matrix provide us the factual information about the therapeutic response exhibited by the specific phytochemicals in the plant matrix [5-7].
In-silico computational techniques are playing an essential role in drug design and development via evaluating interaction affinity of a newly developed molecule or isolated from natural resources, with the targeted proteins or genomes involved in the diseases [8,9]. Out of several computational techniques used to determine the biological effect of the compounds, network pharmacology in one of the advanced and newly admirable techniques is being used exponentially to evaluate pharmacological aspects of the drug-based interaction with a diversity of targeted or untargeted genomes. It is the most used technique to screen the pharmacologically active phytochemicals from a big data of plant matrix such as metabolomic and generating the molecular-based pharmacological evidence for their further applicability [10,11].
Momordica charantia L. (M. charantia) is a folk medicinal herb belonging to the Cucurbitaceae family. It is widely distributed throughout the world in tropical and subtropical regions. Traditionally, it is used as the folk medicinal regime for the treatment of diabetes mellitus, and its fruit has been used as a vegetable. M. charantia hasa wide diversity of phytochemicals belonging to the class of steroids, triterpenes, polyphenols, polysaccharides, protein, etc. Furthermore, based on the current evidence various biological activities of M. charantia have been reported, such as antiviral, antitumor, antihyperglycemic, antibacterial, antimutagenic, antiulcer, antioxidant, antidiabetic, antifertility, anthelmintic, antilipolytic, anticancer, anti-inflammatory immunomodulation and hepatoprotective activities [12,13].
Based on the above facts and their applicability to the molecular-based pharmacological assessment of phytochemicals, the present study is associated to explore the molecular mechanistic role of M. charantia phytochemicals in the treatment of diabetes and diabetic nephropathy (DN) and generating scientific evidence for M. charantia, which would laid a stable foundation for further research on exploring its pharmacological mechanisms in treating DN.
1. Material and methods
1.1. Selection of compounds
Some reported chemical constituents M. charantia were selected from different databases. The screened compounds were linalool, quercetin, gallic acid, apiole, ferulic acid, caffeic acid, limonene and catechin [14,15].
1.2. Selection of Potential DN Targets
Several gene targets were selected from the gene card platform. The keywords like diabetic nephropathy or diabetes were inputted in the GeneCards (https://www.genecards.org/), a human gene compendium with information about genomics, proteomics, and transcriptomics, and UniPort gene database (https://www.uniprot. org/), a comprehensive platform including one of the largest publicly accessible collections of genes, to search for DN-associated targets [16,17].
1.3. Screening Compound-Disease potential interacted Targets and active components network construction analysis
Potential interacted target genes were screened through the integration analysis of the compound-gene network. The screened compound targets were imported into the STRING platform (https://string-db.org/), a software used mainly for functional enrichment and interaction network analysis of genes. Further, the integration analysis was performed using Cytoscape version 3.8.2. Protein-protein interactions (PPI) network and compound-proteins interactions were constructed and interaction information based on the number of nodes, the number of edges, average node degree, average local clustering coefficient were determined for constructed PPI. The analysis covered all the nearly functional interactions among the expressed proteins-proteins and compound-proteins network [10,18].
1.4. Gene Ontology (GO) analysis
Gene Ontology (GO) analysis was performed to evaluate multiple pathophysiological of genes in DN. Metascape (metascape.org) AND network analyst (https://www.networkanalyst.ca/) tool were used to perform the analysis. The analysis involved the genes which were found to interact with the active metabolites. The common abbreviation of each gene was inputted in the search toolbar at the platform of metascape and network analysis. In this analysis, top enrichment outcomes were added as the outcome of the study [19-21].
1.5. ADME analysis
ADME (absorption, distribution, metabolism, and excretion) and toxicological analysis will be performed for selected metabolites through “SwissADME (http://www.swissadme.ch/index.php)” and ProTox-II-Prediction tool of toxicity of chemicals (https://tox-new. charite.de/protox_II/index.php?site=home). TPSA (Topological Polar Surface Area (TPSA) for drug integrity, Consensus Log Po/w for drug lipophilicity, Log Kp (skin permeation) and drug-likeness were predicted as the standard parameters for selected the ADME response of [22].
2. Results
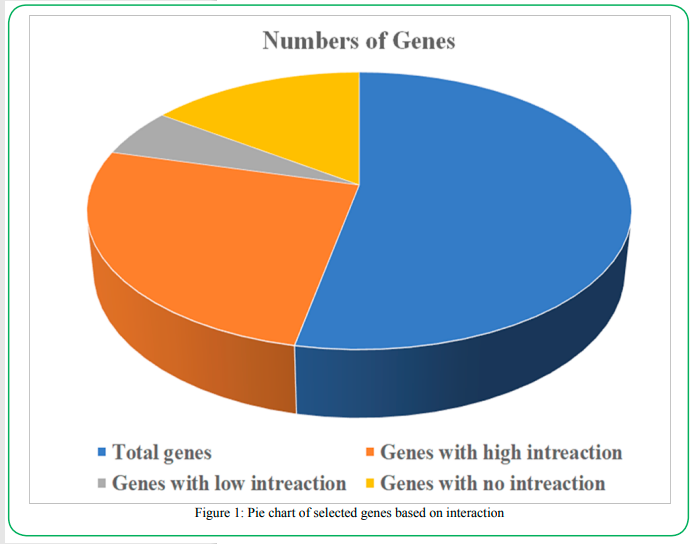
Eight active major metabolites were selected for the network pharmacology analysis based on their interaction and ligation efficacy with the screened genes. In the pre-screening analysis of potential targeted genes, 41 genes were selected which were even partially interconnected with the active metabolite and other genes. The analysis was performed based on the interaction efficacy of each target. The analysis showed that out of 84 genes, 41 genes were found to highly interact with the active metabolites and each target while 9 and 24 genes were found with the least even no interaction between the active metabolites and targeted genes. The genes summary has been summarized in Figure 1.
2.1. Screening Compound-Disease potential interacted Targets and active components network construction analysis
2.1.1. Common target network (protein-protein and protein-disease interaction
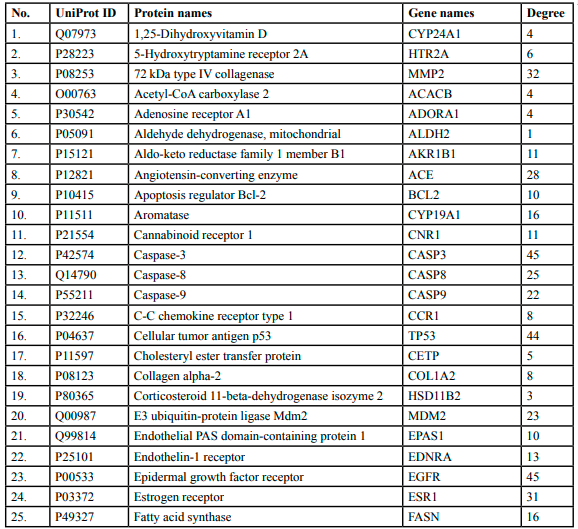
Forty-one putative target genes interconnected in pathophysiology in DN were further analyzed for protein-protein interactions and protein-disease interaction. The analysis was conducted through the STRING database and Network analyst (https://www.networkanalyst.ca/). In PPI analysis, the gene-gene interaction network was established with a medium confidence score of 0.400. The established network embodied number of nodes:41, number of edges: 508, average node degree: 24.8, average local clustering coefficient: 0.844, expected number of edges: 182 and PPI enrichment p-value: < 1.0e-16. In addition, the established PPIwas found significantly with more interactions than expected. The significant interaction of geneswas based on proteins of similar size selected from the genome database and characterized as at least partially biologically connected during DN. In the figure, the edges characterize the interaction between sets of potential targets, while the nodes characterize the targeted genes. The PPI network has been summarized in Figure 2.

Figure 2: PPI network generated through Cytoscape and STRING tool. Figure (A) represents the PPI generated by cytoscape tool based on experimental databased of the genomes, the pink line represents the interaction of the edges with nodes screened from experimental sources. Figure (B) represents the PPI network generated by STRING database.
2.1.2. Active metabolites target genes network
Active metabolites and target gene associated network was constructed and the outcome of the study revealed that each selected metabolite of M. charantia was found to potentially interact with each gene. In this analysis, edges that directly connected with the active metabolites were remained to appear, whereas the edges which were disappeared interconnected with each target (Figure 3). Quercetin was found to have strong interaction with targets such as NOS, ILs, CASPs, MAPKs, etc which were found to play an essential role in oxidative stress, inflammation, or oxidative stress-induced inflammation. Ferulic acid was found to have interaction with G6PD, MAPK1, MAPK3, etc. Caffeic acid was found to have interaction with MAPKs, MMPs, and G6PD. Catechin was found to have interaction with NOS, PTGS2, PON1, ILs, etc. Gallic acid was found to have interaction with CASPs, AKT1, JUN, MMPs, etc. Apiole was found to have interaction with TP53 while limonene and linalool were found to have interaction with NOS, PPARG, TP53, etc. The constructed network of active metabolites and targets has been represented in Figure 3.

Figure 3: compound and protein interaction (CPI) network represents the active components with targets while quercetin showed the most prominent interaction.
2.1.3. Gene Ontology (GO) analysis
GO analysis was performed to evaluate the multiple physiological roles of each gene for the regulation of DN. The observation of enriched terms across input genes and enrichment gene-disease network analysis suggests their physiological role in the management of DN. The results showed multiple physiological roles of each target in the regulation of DN via ameliorating endothelial dysfunction, fatty liver disease, diabetes mellitus, acute kidney injury, fibrosis, hypertensive disease, obesity, etc. furthermore, it was also found that the targets potentially play an essential role in the regulation of oxidative stress, inflammation, and oxidative stress-induced inflammation. The outcomes of the analysis have been summarized in Figure 4.
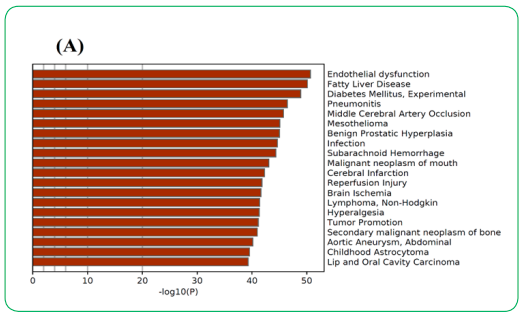
Figure 4 (A): Gene ontology (GO) and gene-disease association analysis of potential genes were found to interact with the active compounds of M. charantia. Figure (A) represents the GO analysis while figure (B) represents the gene-disease association network.
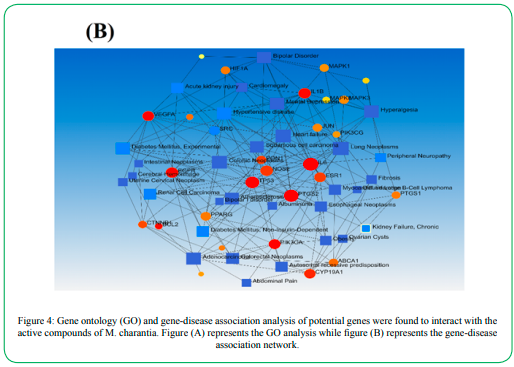
Figure 4 (B): Gene ontology (GO) and gene-disease association analysis of potential genes were found to interact with the active compounds of M. charantia. Figure (A) represents the GO analysis while figure (B) represents the gene-disease association network.
2.2. ADME analysis
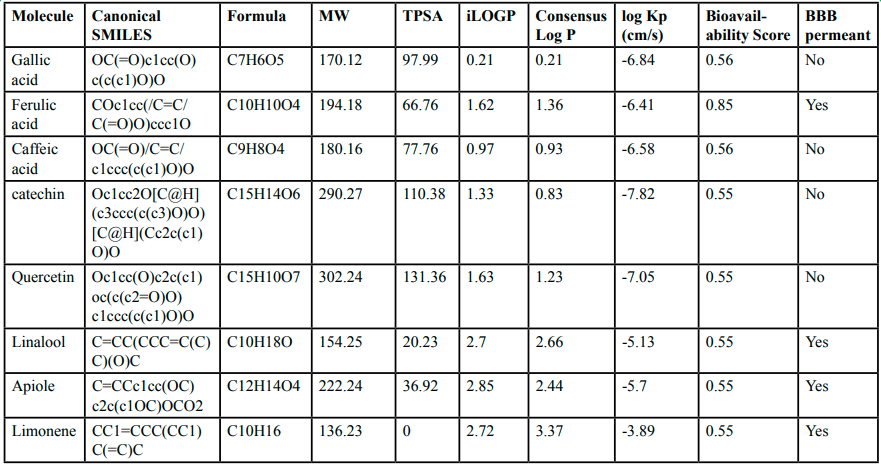
ADME analysis of selected metabolites was performed successfully using the computational tool “SwissADME”. The parameters such as TPSA, consensus Log Po/w, ESOL Log S values, GI absorption, BBB permeant and log Kp (cm/s) (skin permeation) were predicted to determine the ADME, lipophilicity and the drug-likeness response of the metabolites. The polar surface area (PSA) of metabolites was calculated using the fragmental technique TPSA. It acts as a useful descriptor in many models and rules to estimate some ADME properties, especially concerning absorption and brain access [23]. The consensus log Po/w is characterized as the arithmetic mean of five proposed methods of lipophilicity which represents the lipophilicity of anticipated molecules. The classical descriptor for lipophilicity is generally the partition coefficient between n-octanol and water (log Po/w). SwissADME dedicated this section due to the critical importance for the assessment of physicochemical properties for pharmacokinetic drug discovery using computational tools. The models accelerate the prediction accuracy for the physicochemical properties through consensus log Po/w [24]. The outcomes of the study revealed that a negative value of logP for each metabolite means that the compound poses a high-affinity hydrophilic nature, while a positive value of logP represents the lipophilicity of the molecule. Similarly, Potts and Guy provided a model to predict skin permeability using the skin permeability coefficient (Kp). The more negative the log Kp (with Kp in cm/s), the less skin permeant the molecule [22]. Our findings suggest that each metabolite possesses high skin permeability, as the log Kp values for these molecules were less than -8.00. The outcomes of ADME analysis are summarized in Tables 2 and Figure 5.
The blood-brain barrier (BBB) permeant affinity of the molecules depends on two physicochemical descriptors only (consensus log Po/w and TPSA) which represent lipophilicity and apparent polarity. In case, If the egg-shaped molecular classification plot covers the yolk, it means that the molecule exhibits physicochemical space for highly probable BBB permeation, while it remains within the range of the white which represents the physicochemical space for highly probable HIA absorption. In addition, both compartments are not mutually exclusive by the molecule and remain outside the gray region, representing the molecules implying low absorption and limited brain penetration [22]. The outcomes of our study suggest that ferulic acid, apiole, linalool and limonene exhibited high BBB permeant affinity, excluding other compounds. The boiled egg plot of ADME analysis is summarized in Figure 6.

Figure 5: ADME analysis of selected compounds in M. charantia. Figure 1 represents the chemical structure and ADME radar plot of Gallic acid, Figure 2 represents the chemical structure and ADME radar plot of Ferulic acid, Figure 3 represents the chemical structure and ADME radar plot of Caffeic acid, Figure 4 represents the chemical structure and ADME radar plot of catechin, Figure 5 represents the chemical structure and ADME radar plot of Quercetin, Figure 6 represents the chemical structure and ADME radar plot of Linalool, Figure 7 represents the chemical structure and ADME radar plot of Apiole and Figure 8 represents the chemical structure and ADME radar plot of Limonene.

Figure 6: ADME Boiled egg plot of M. charantia constituents such as Gallic acid, Ferulic acid, Caffeic acid, catechin, Quercetin, Linalool, Apiole and Limonene.
3. Discussion
Forcenturies, medicinal plants have been playing an important role in alleviating several acute and chronic disorders due to their multi-mechanistic approach to cure even treating the ailments. M. charantia is an Indian herb used to treat various disorders including diabetes, traditionally. M. charantia possesses several varieties of phytoconstituents which belong to the class of terpenoids, steroids, polyphenols, etc., and reported for treating diabetes, the present study is associated for validation of traditional therapeutic claim M. charantia in alleviating diabetes or DN using network pharmacology approach.
In this study, several genes were selected from different gene databases and explored for their multi-mechanistic role in alleviating DN. The study was performed on some selected metabolites such as quercetin, gallic acid, apiole, ferulic acid, caffeic acid, etc and the outcomes of the analysis revealed that, among eight metabolites, quercetin was the most active compound of M. charantia, which were found to have interaction with several genes such as nitric oxide synthases (NOS), caspases (CASPs), mitogen-activated protein kinase (MAPKs), matrix metalloproteinase (MMPs), in DN. NOS, CASPs, and MAPKs play an important role in the regulation of inflammation [25,1]. Metalloproteinases (MMPs) play an essential role in diabetic wound healing by removal of damaged extracellular matrix (ECM) during the inflammatory phase, breakdown of the capillary basement membrane for angiogenesis and cell migration during the proliferation phase, as well as in construction and remodeling of tissue [26]. AKT is responsible for the regulation of glucose uptake by mediating insulin-induced translocation of GLUT4 but endogenous AKT is likely to play a significant physiological role in insulin-stimulated glucose uptake in insulin targets such as muscle and adipose tissue [27].
Several studies have demonstrated that phenols exhibit potential anti-inflammatory action by suppression of IL-4, TNF, NF-κB, MAPKs activation, and expression of the Na+/Ca2+ exchanger [28]. Quercetin down regulates the expression of IL-1β, IL-6, IFN-γ, and TNF-α secretion and thus regulates the inflammatory stress [29]. PONs proteins play an important role in dyslipidemia-induced CKD. Quercetin exhibits potential interaction with PONs and thus regulating dyslipidemia-induced CKD [30]. Gallic acid exerts anti-inflammatory, anti-oxidative stress, and nephroprotective effects by regulation of TNF, IL-1B, AKT, CASP3, and STAT/JUN [1,31,32]. Ferulic acid exhibits a protective effect against diabetic nephropathy by attenuating oxidative insult, inflammation, and autophagy. The underlying mechanism of ferulic acid was established and revealed AGEs, NF-κB, MAPKs CASPs, JNK, and ERK are the most regulated proteins in renal pathophysiology [33,34].
It is reported that catechins exert a protective effect against inflammation, diabetes-induced renal oxidative damage, fibrosis, and albuminuria via the regulation of several genomes involved in oxidative and inflammatory stress. Furthermore, it is reported that catechins are the most prominent active against IL-6, inducible nitric oxide synthase, and nitrite and reducing oxidative stress-induced inflammation.
4. Conclusion
Based on the above facts, it can be demonstrated thatM. charantia possesses several metabolites including polyphenols which exhibit an important role in the treatment of DN via regulation of several genomes such as AKTs, CASPs, MAPKs, ILs, NOs, etc, responsible for its pathophysiology. Furthermore, the generated evidence validates the traditional claim of M. charantia for alleviating DN.
Conflict of interest:
The author declares no conflict of interest.
Acknowledgment:
I wish thanks to Jamia Hamdard for providing the facilities to conduct the study.
Source of funding:
Nil
References
G. Gautam, B. Parveen, M. Umar Khan, I. Sharma, A. Kumar Sharma, R. Parveen, S. Ahmad, (2021). A systematic review on nephron protective AYUSH drugs as constituents of NEERI-KFT (A traditional Indian polyherbal formulation) for the management of chronic kidney disease, Saudi J. Biol. Sci. https://doi.org/10.1016/J.SJBS.2021.07.008.View
Gaurav, S. Zahiruddin, B. Parveen, M. Ibrahim, I. Sharma, S. Sharma, A.K. Sharma, R. Parveen, S. Ahmad, (2020). TLC-MS Bioautography-Based Identification of Free-Radical Scavenging, α-Amylase, and α-Glucosidase Inhibitor Compounds of Antidiabetic Tablet BGR-34, ACS Omega. https://doi.org/10.1021/acsomega.0c02995.View
N.M.A. Rasheed, V.C. Gupta, Standardization of a compound Unani herbal formulation “qurs-e- Luk” with modern techniques, Pharmacognosy Res. (2010). https://doi.org/10.4103/0974- 8490.69115.View
M.N. Mallick, M. Singh, R. Parveen, W. Khan, S. Ahmad, M. Zeeshan Najm, S.A. Husain, (2015). HPTLC Analysis of Bioactivity Guided Anticancer Enriched Fraction of Hydroalcoholic Extract of Picrorhiza kurroa, Biomed Res. Int. https://doi.org/10.1155/2015/513875.View
Y. Yang, L. Gu, Y. Xiao, Q. Liu, H. Hu, Z. Wang, K. Chen, (2015). Rapid identification of α-glucosidase inhibitors from phlomis tuberosa by sepbox chromatography and thin-layer chromatography bioautography, PLoS One. https://doi. org/10.1371/journal.pone.0116922.
A. Nugroho, H. Heryani, J.S. Choi, H.J. Park, (2017). Identification and quantification of flavonoids in Carica papaya leaf and peroxynitrite-scavenging activity, Asian Pac. J. Trop. Biomed. https://doi.org/10.1016/j.apjtb.2016.12.009.View
S.K. Bhattacharya, D. Bhattacharya, K. Sairam, S. Ghosal, (2002). Effect of bioactive tannoid principles of Emblica officinalis on ischemia-reperfusion-induced oxidative stress in rat heart, Phytomedicine. https://doi.org/10.1078/0944-7113- 00090.View
K. Chandra, W. Khan, S. Jetley, S. Ahmad, S.K. Jain, (2018). Antidiabetic, toxicological, and metabolomic profiling of aqueous extract of Cichorium intybus seeds, Pharmacogn. Mag. https://doi.org/10.4103/pm.pm_583_17.View
M.R. Islam, A. Zaman, I. Jahan, R. Chakravorty, S. Chakraborty, (2013). In silico QSAR analysis of quercetin reveals its potential as therapeutic drug for Alzheimer’s disease, J. Young Pharm. https://doi.org/10.1016/j.jyp.2013.11.005.View
F. Yi, L. Li, L. jia Xu, H. Meng, Y. mao Dong, H. bo Liu, P. gen Xiao, (2018). In silico approach in reveal traditional medicine plants pharmacological material basis, Chinese Med. (United Kingdom). https://doi.org/10.1186/s13020-018-0190-0.
J. Zhang, C. Zheng, S. Yuan, X. Dong, L. Wang, Y. Wang, W. Wang, K. Gao, J. Liu, (2019). Uncovering the Pharmacological Mechanism of Chaibei Zhixian Decoction on Epilepsy by Network Pharmacology Analysis, Evidence-Based Complement. Altern. Med. https://doi.org/10.1155/2019/3104741.View
A.M.L. Dans, M.V.C. Villarruz, C.A. Jimeno, M.A.U. Javelosa, J. Chua, R. Bautista, G.G.B. Velez, (2007). The effect of Momordica charantia capsule preparation on glycemic control in Type 2 Diabetes Mellitus needs further studies, J. Clin. Epidemiol. https://doi.org/10.1016/j.jclinepi.2006.07.009.View
S. Jia, M. Shen, F. Zhang, J. Xie, (2017). Recent advances in momordica charantia: Functional components and biological activities, Int. J. Mol. Sci. https://doi.org/10.3390/ijms18122555.View
M.A. Alam, R. Uddin, N. Subhan, M.M. Rahman, P. Jain, H.M. Reza, (2015). Beneficial Role of Bitter Melon Supplementation in Obesity and Related Complications in Metabolic Syndrome, J. Lipids. https://doi.org/10.1155/2015/496169. View
J. Ahamad, S. Amin, S. R. Mir, (2017). Momordica charantia Linn. (Cucurbitaceae): Review on Phytochemistry and Pharmacology, Res. J. Phytochem. https://doi.org/10.3923/ rjphyto.2017.53.65.View
R. Zhang, X. Zhu, H. Bai, K. Ning, (2019). Network pharmacology databases for traditional Chinese medicine: Review and assessment, Front. Pharmacol. https://doi. org/10.3389/fphar.2019.00123. View
A.I. Casas, A.A. Hassan, S.J. Larsen, V. Gomez-Rangel, M. Elbatreek, P.W.M. Kleikers, E. Guney, J. Egea, M.G. López, J. Baumbach, H.H.H.W. Schmidt, (2019). From single drug targets to synergistic network pharmacology in ischemic stroke, Proc. Natl. Acad. Sci. U. S. A. https://doi.org/10.1073/ pnas.1820799116.View
Y. Li, L. Wang, B. Xu, L. Zhao, L. Li, K. Xu, A. Tang, S. Zhou, L. Song, X. Zhang, H. Zhan, (2021). Based on Network Pharmacology Tools to Investigate the Molecular Mechanism of Cordyceps sinensis on the Treatment of Diabetic Nephropathy, J. Diabetes Res. https://doi.org/10.1155/2021/8891093. View
Q. Tao, J. Du, X. Li, J. Zeng, B. Tan, J. Xu, W. Lin, X. lin Chen, (2020). Network pharmacology and molecular docking analysis on molecular targets and mechanisms of Huashi Baidu formula in the treatment of COVID-19, Drug Dev. Ind. Pharm. https:// doi.org/10.1080/03639045.2020.1788070.View
L. Cheng, F. Wang, S.B. Zhang, Q.Y. You, (2021). Network Pharmacology Integrated Molecular Docking Reveals the Anti-COVID-19 and SARS Mechanism of Fufang Banlangen Keli, Nat. Prod. Commun. https://doi.org/10.1177/1934578X20988420. View
Q.D. Xia, Y. Xun, J.L. Lu, Y.C. Lu, Y.Y. Yang, P. Zhou, J. Hu, C. Li, S.G. Wang, (2020). Network pharmacology and molecular docking analyses on Lianhua Qingwen capsule indicate Akt1 is a potential target to treat and prevent COVID-19, Cell Prolif. https://doi.org/10.1111/cpr.12949.View
A. Daina, O. Michielin, V. Zoete, (2017). SwissADME: A free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules, Sci. Rep. https://doi.org/10.1038/srep42717.View
A. Daina, V. Zoete, (2016). A BOILED-Egg To Predict Gastrointestinal Absorption and Brain Penetration of Small Molecules, ChemMedChem. https://doi.org/10.1002/ cmdc.201600182.View
R. Mannhold, G.I. Poda, C. Ostermann, I. V. Tetko, (2009). Calculation of molecular lipophilicity: State-of-the-art and comparison of log P methods on more than 96,000 compounds, J. Pharm. Sci. https://doi.org/10.1002/jps.21494. View
J.P. S, S. Evan Prince, (2018). Diclofenac-induced renal toxicity in female Wistar albino rats is protected by the pre-treatment of aqueous leaves extract of Madhuca longifolia through suppression of inflammation, oxidative stress and cytokine formation, Biomed. Pharmacother. https://doi.org/10.1016/j. biopha.2017.12.028.View
S.M. Ayuk, H. Abrahamse, N.N. Houreld, (2016). The Role of Matrix Metalloproteinases in Diabetic Wound Healing in relation to Photobiomodulation, J. Diabetes Res. https://doi. org/10.1155/2016/2897656.View
L.N. Cong, H. Chen, Y. Li, L. Zhou, M.A. McGibbon, S.I. Taylor, M.J. Quon, (1997). Physiological role of AKT in insulin-stimulated translocation of GLUT4 in transfected rat adipose cells, Mol. Endocrinol. https://doi.org/10.1210/ mend.11.13.0027.View
S. Shukla, S. Gupta, (2010). Apigenin: A promising molecule for cancer prevention, Pharm. Res. https://doi.org/10.1007/ s11095-010-0089-7.View
S. Chen, H. Jiang, X. Wu, J. Fang, (2016). Therapeutic Effects of Quercetin on Inflammation, Obesity, and Type 2 Diabetes, Mediators Inflamm. https://doi.org/10.1155/2016/9340637.View
M. Solati, H.R. Mahboobi, (2012). Paraoxonase enzyme activity and dyslipidemia in chronic kidney disease patients, J. Nephropathol. https://doi.org/10.5812/nephropathol.8106.View
X. Lin, G. Wang, P. Liu, L. Han, T. Wang, K. Chen, Y. Gao, (2021). Gallic acid suppresses colon cancer proliferation by inhibiting SRC and EGFR phosphorylation, Exp. Ther. Med. https://doi.org/10.3892/etm.2021.10070.View
A. Nouri, F. Heibati, E. Heidarian, (2021). Gallic acid exerts anti-inflammatory, anti-oxidative stress, and nephroprotective effects against paraquat-induced renal injury in male rats, Naunyn. Schmiedebergs. Arch. Pharmacol. https://doi. org/10.1007/s00210-020-01931-0.View
S. Chowdhury, S. Ghosh, A.K. Das, P.C. Sil, (2019). Ferulic acid protects hyperglycemia-induced kidney damage by regulating oxidative insult, inflammation and autophagy, Front. Pharmacol. https://doi.org/10.3389/fphar.2019.00027.View
E. Bami, O.B. Ozakpınar, Z.N. Ozdemir-Kumral, K. Köroglu, F. Ercan, Z. Cirakli, T. Sekerler, F.V. Izzettin, M. Sancar, B. Okuyan, (2017). Protective effect of ferulic acid on cisplatin induced nephrotoxicity in rats, Environ. Toxicol. Pharmacol. https://doi.org/10.1016/j.etap.2017.06.026.View