Current Research in Materials Chemistry Volume 1 (2019), Article ID: CRMC-103
https://doi.org/10.33790/crmc1100103Review Article
The size fractionation of graphene oxide induced by Brownian motion
Yaming Hao, Zhipeng Deng, Xiaomin Zhao, Yunjie Liu, Danqing Chen, Guohua Chen*
College of Materials Science and Engineering, Huaqiao University, Xiamen, 361021, P.R.China
Corresponding Author Details: College of Materials Science and Engineering, Huaqiao University, Xiamen, 361021, P.R.China E-mail: hdcgh@hqu.edu.cn
Received date: 15th October, 2019
Accepted date: 11th October, 2019
Published date: 13th October, 2019
Citation: Hao Y, Deng Z, Zhao X, Liu Y, Chen D, Chen G (2019) The size fractionation of graphene oxide induced by Brownian motion Cur Res Mater Chem 1: 103.
Copyright:©2019, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
The size fractionation of graphene oxide (GO) sheets is essential for its performances. Based on the classical “Brownian motion” theory of microscopic particles suspended in a fluid, a novel, cost-effective and eco-friendly method was developed for the size fractionation of graphene oxide (GO) sheets. Owing to the more intensive Brownian motion of small-size sheets compared to the large-size ones, the small-size GO sheets passed through a self-designed wire-mesh nylon filter and were separated successfully. The size of separated GO sheets were mostly (>90%) smaller than 6 μm, and they showed narrower size distribution compared with original GO sheets before size fractionation.
Keywords: Carbon materials; Graphene oxide; Size fractionation; Diffusion
Introduction
Graphene, a monolayer of sp2-bonded carbon atoms with twodimensional (2D) honeycomb lattice [1], has been widely applied in various fields, such as composites, electronics, transistors and biosensors due to its excellent electrical, optical, thermal and mechanical properties [2-6]. The lateral dimensions of graphene sheets play a key role in controlling their properties and applications [7-10]. Therefore, it is essential to produce graphene with homogeneous size to meet practical applications. It’s known that the most widely used methods to massively prepare graphene with homogeneous size are the reduction of graphene oxide (GO) [11,12]. The size of graphene sheets are mainly depended on the GO sheets. However, the prepared GO sheets always possess wide size distribution inevitably. Hence, the size fractionation of GO sheets is necessary for the reduced graphene oxide (RGO). In recent years, tremendous efforts have been devoted to investigate the size fractionation of GO sheets [13-17]. For example, pH-assisted selective sedimentation [9], the density gradient ultracentrifugation technique (DGU) [13], centrifugation [14], the polar solvent-selective natural deposition method [11], the filtration through using track-etched membranes [15], electrophoresis [16], and directional freezing [17]. However, these methods are difficult to scale up in controlling the size of GO sheets for practical applications, owing to the complicated operating procedures, high centrifugation rate and high cost. Therefore, it is highly desired to develop a facile, scalable and effective method for the size fractionation of GO sheets.
To address aforementioned issues, we originally reported a facile and economical method for separating the small-size GO sheets utilizing the Brownian motion of GO sheets in aqueous solution. GO sheets were prepared by modified Hummer’s method. A wire-mesh nylon setup was self-designed for the size fractionation. The GO sheets (<10 μm) evidently passed through the filter into the pure water, while the GO sheets (>10 μm) were kept in the original solution. Such can be supported by the Tyndall effect of the upper solution of GO samples that underwent 12 h stirring the bottom solution. The separated GO sheets (<6 μm) shows narrow size distribution. The size-fractionation mechanism of this method was described in detail in view of Brownian motion theory.
We designed a setup to separate GO sheets, and the small-size GO sheets can be observed by a transparent optical path appeared when we using the laser irradiation treated the upper solution. Interestingly, after fractionation, the small-size GO sheets possessed uniform size distribution. Furthermore, this method can be used easily to produce GO sheets with size mostly smaller than 6 μm.
Materials and methods
Materials
Graphite powder with a mesh of 8000 and a purity of 99.9% was obtained from Xia'men Kano Graphene Technology Co., Ltd. Sulfuric acid (H2SO4, 98%), hydrochloric acid (HCl), hydrogen peroxide (H2O2, 30%), potassium permanganate (KMnO4), were all analytical grade from Sinopharm Chemical Reagent Co., Ltd. All these materials and chemicals were used as it is without further purifications.
Experiment
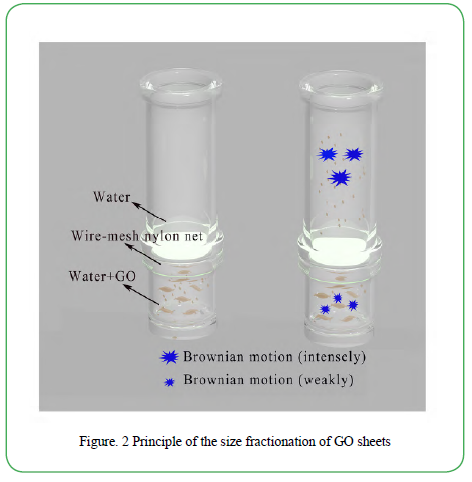
The GO was prepared by modified Hummers’ method. As-prepared GO was kept as a honey-like colloidal without being dried. The Brownian diffusion device was self-designed as shown in Figure. 2. One glass column was connected vertically to a glass container sealing by two silicone O-rings. A wire-mesh nylon net (≤18 μm) was put between the two O-rings carefully. Experimentally, GO aqueous colloidal (1 ml) was poured into the bottom glass container and stirred magnetically. Then, 150 ml deionized water was gradually added, followed by 12 h stirring. After that, the separated GO sheet were obtained by collecting the upper solution using pipet, and remained to be studied.
Characterization
The size of nylon net was observed by using an optical microscope (59 XD-PC, Shanghai optical instrument factory). Powder X-ray diffraction (XRD) patterns were collected on a Rigaku SmartLab 3KW diffractometer using Cu Kα radiation. A Field emission scanning electron microscope (FESEM) was used to characterize the size and morphology of GO sheets (JSM-6700, operating voltage is 5 kV).
Results and discussion
Figure. 1 showed the XRD spectra of graphite and GO. Graphite showed a sharp [002] peak at 26.6º as shown in Figure. 1, which is attributed to the highly organized lay structure. A new diffraction [001] peak appeared at around 10º in Figure. 1, which was caused by the insertion of oxygen functional groups. It showed that GO was synthesized by a modified Hummers’ method successfully.
Figure. 2 described the size fractionation principle based on the Brownian motion of GO sheets. According to the Stokes-Einstein equation [18] (1), where D is diffusion coefficient, KB is Boltzmann’s constant, T is the temperature, β=3πμd, μ is fluid viscosity, d is Brownian particle diameter, Brownian diffusion is more extensive as the particle size increases under condition of constant temperature. D =KBT (1)
As is known, GO sheets possess abundant hydroxyl (-OH) and epoxy groups on the layer planes, together with carboxylic groups at the edges of the lamellae. Therefore, GO sheets have the good dispensability in aqueous solution. In the GO solution, random Brownian motion of GO sheets naturally takes place. Based on the equation 1, the Brownian motion of small-size GO sheets is more extensive than that of large-size ones. In the self-designed size fractionation setup, it’s easier for the small-size GO sheets to diffuse through the wire-mesh nylon net into the upper pure water than the large-size ones, when stirring the bottom solution. Consequently, small-size GO sheets have high priority to be collected in the upper solution during the stirring.
In Figure. 3, the size fractionation of GO sheets was carried out by a self-designed wire-mesh nylon filter. GO solution was formed by stirring in the bottom (Figure. 3a). At first, Tyndall effect was not shown when the upper water was irradiated by a laser pen (Figure. 3b). After 12 h stirring, a transparent optical path appeared in the upper solution when the solution was irradiated using a laser pen at room temperature (Figure. 3c). This phenomenon was a typical Tyndall effect. It indicated that some GO sheets passed through the nylon net into the upper water.

Figure. 3 The GO colloidal dispersion in the bottom (a); The upper water before stirring the bottom solution, using laser pen with a constant temperature (b); Tyndall effect of the upper solution that underwent 12 h stirring the bottom solution (c).
The size of GO sheets in the upper solution and original GO solution were studied by the Field-emission scanning electron microscope (FESEM). The SEM images and corresponding size fractionation histograms of GO sheets were showed in Figure. 4a and b. Before fractionation, the size of synthesized GO sheets ranged from 1 to 35 μm and the distribution was wide (Figure. 4a). After 12 h stirring, the size of GO sheets diffused into the upper water mainly ranged from 1 to 10 μm (Figure. 4b). Above 90% of them were smaller than 6 μm. The size distribution was narrower than that before the size fractionation. Furthermore, the Gauss fits of the GO sheets before and after fractionation indicated that their maximum distributions of GO sizes were 4 μm and 3 μm, respectively. These results indicated that the GO sheets were separated successfully according to Brownian motion of GO sheets. It was consistent with the principle discussed above.
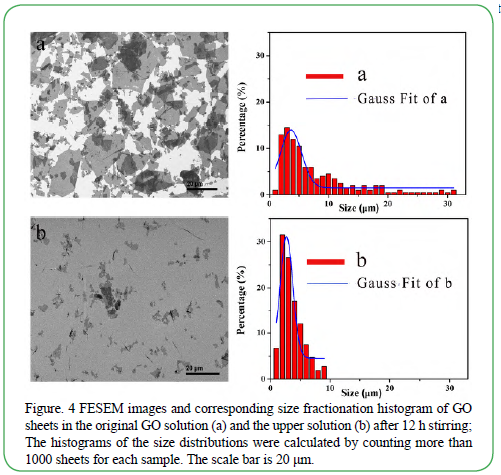
Figure. 4 FESEM images and corresponding size fractionation histogram of GO sheets in the original GO solution (a) and the upper solution (b) after 12 h stirring; The histograms of the size distributions were calculated by counting more than 1000 sheets for each sample. The scale bar is 20 μm.
The successful size fractionation was attributed to the following reasons: (i) the good dispensability of GO sheets; (ii) the different ability of Brownian motion between small GO sheets and large GO sheets; (iii) the pore selectivity of wire-mesh nylon net.
Conclusions
In summary, we have demonstrated a facile, low cost method for the size fractionation of GO sheets based on Brownian motion. Smallsize GO sheets have stronger Brownian diffusion than large-size ones, when the temperature is constant. Hence, the diffusion ability of GO sheets varied from their size dimension in a solution. Owing to this phenomenon, the small-size of GO sheets priorly diffused into the upper water zone and was separated instead of large-size ones in a self-designed separator. The results showed that the smallsize GO sheets collected from the upper solution had narrow size distribution, which was mostly smaller than 6 μm (>90%). The novel size fractionation method in this work shows potential prospect on the preparation of GO with different lateral size.
Acknowledgements
This work was financially supported by Natural Science Foundation of China (51373059), Science and technology plan projects in Fujian province (2018H6012, 2017H2001, 2018H0019), Science and technology innovation team of Huaqiao University (Z14X0046), and The Research Center of Graphene Powder & Composites of Fujian Province.
Conflicts of Interest (COI) Statement
The author declare that there is no conflict of interest regarding the publication of this paper.
References
Stankovich, S., Dikin, D., Dommett, G., Kohlhaas, K.,& Zimney, E. (2006). Graphene-based composite materials. Nature 442: 282.View
Bonaccorso, F., Sun, Z., Hasan, T.,& Ferrari, A. (2010). Graphene photonics and optoelectronics. Nature photonics 4: 611.View
Clochard, M., Melilli, G., Rizza, G., Madon, B.,& Alves, M. (2016). Large area fabrication of self-standing nanoporous grapheneon- PMMA substrate. Materials Letters 184: 47-51.View
Chen, D., Feng, H.,& Li, J. (2012). Graphene oxide: preparation, functionalization, and electrochemical applications. Chemical reviews 112: 6027-6053.View
Wan, S., Bi, H., Zhou, Y., Xie, X.,& Su, S. (2017). Graphene oxide as high-performance dielectric materials for capacitive pressure sensors. Carbon 114: 209-216.View
Deka, M., Baruah, U.,& Chowdhury, D. (2015). Insight into electrical conductivity of graphene and functionalized graphene: Role of lateral dimension of graphene sheet. Mater Chem Physics 163: 236-244.View
Pattananuwat, P.,& Aht-Ong, D. (2016). One-step method to fabricate the highly porous layer of poly (pyrrole/ (3,4-ethylenedioxythiophene)/) wrapped graphene hydrogel composite electrode for the flexibile supercapacitor. Mater Letters 184: 60-64.View
Olowojoba, G., Eslava, S., Gutierrez, E., Kinloch, A.,& Mattevi, C. (2016). In situ thermally reduced graphene oxide/epoxy composites: thermal and mechanical properties. Applied Nanoscience 6: 1015-1022.View
Wang, X., Bai, H.,& Shi, G. (2011). Size fractionation of graphene oxide sheets by pH-assisted selective sedimentation. J Am Chem Soc 133: 6338-6342.View
Zhang, Y., Chen, Z., Wang, B., Wu, Y.,& Jin, Z. (2013). Controllable growth of millimeter-size graphene domains on Cufoil. Mater Letters 96: 149-151.
Zhang, W., Zou, X., Li, H., Hou, J.,& Zhao, J.(2015). Size fractionation of graphene oxide sheets by the polar solventselective natural deposition method. RSC Advances 5: 146-152.
Nia, Z., Chen, J., Tang, B., Yuan, B.,& Wang, X. (2017). Optimizing the free radical content of graphene oxide by controlling its reduction. Carbon 116: 703-712.View
Sun, X., Luo, D., Liu, J.,& Evans, DG. (2010). Monodisperse chemically modified graphene obtained by density gradient ultracentrifugal rate separation. Acs Nano 4: 3381-3389.View
Özçakır, E.,& Eskizeybek, V. (2016). A Facile and Effective Method for Size Sorting of Large Flake Graphene Oxide. In Proceedings of the World Congress on Recent Advances in Nanotechnology (RAN’16)View
Chen, J., Li, Y., Huang, L., Jia, N.,& Li, C. (2015). Size Fractionation of Graphene Oxide Sheets via Filtration through Track Etched Membranes. Adv mater 27: 3654-3660.View
Cui, C., Huang, J., Huang, J.,& Chen, G. (2017). Size separation of mechanically exfoliated graphene sheets by electrophoresis. Electrochimica Acta 258: 793-799.
Geng, H., Yao, B., Zhou, J., Liu, K., Bai,& G. (2017). Size fractionation of graphene oxide nanosheets via controlled directional freezing. J Am Chem Soc 139: 12517-12523.View
Maragó, O., Bonaccorso, F., Saija, R., Privitera, G.,& Gucciardi, P. (2010). Brownian motion of graphene. ACS nano 4: 7515-7523.View