Current Research in Materials Chemistry Volume 1 (2019), Article ID: CRMC-104
https://doi.org/10.33790/crmc1100104Review Article
Conformational Polymorphism in Organic Crystals: Structural and Functional aspects - A Review
Bidisha Sengupta1,3*,Pradeep K. Sengupta2,Romans Grant3,Matthew Beasley1,Benjamin Mason1,Tanesha L. Love3,Larissa Barroso4,Mariela Alvarado4,M. S. Zaman4,5
1Department of Chemistry and Biochemistry, Stephen F. Austin State University, Nacogdoches, TX, USA.
2Department of Biophysics, Molecular Biology and Bioinformatics, University of Calcutta, India.
3Former Associate Professor of Chemistry at Tougaloo College, 500 West County Line Road, Tougaloo, MS, USA.
4Department of Biology, South Texas College, McAllen, TX, USA.
5Department of Biological Sciences, Alcorn State University, Lorman, MS, USA.
Corresponding Author Details: Bidisha Sengupta, Department of Chemistry and Biochemistry, Stephen F. Austin State University, PO Box 13006, SFA Station, Nacogdoches, TX 75962, United States. E-mail: bidisha.sengupta@sfasu.edu
Received date: 30th October, 2019
Accepted date: 19th November, 2019
Published date: 22nd November, 2019
Citation: Sengupta B, Sengupta PK, Grant R, Beasley M, Mason B, Love TL, Barroso L, Alvarado M, Zaman MS (2019) Conformational Polymorphism in Organic Crystals: Structural and Functional aspects - A Review. Cur Res Mater Chem 1: 104.
Copyright:©2019, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Polymorphism in organic crystals involves the formation of isomeric molecular identities. It is dependent on the structural arrangement due to inter-atomic interactions, as well as external stimuli, which include temperature, visible and UV radiation. Conformational polymorphism of organic crystalline molecules is often the result of isomerism due to the twisting and turning of angular bonds. The arrangement of the atoms supports different types of bonding mechanisms (which include hydrogen bonding) within the same compound. This, in turn, results in the formation of cis/trans configurational isomers or a proton transfer species (tautomer), having different functional properties. The conformers support the flexibility of bond angles in an attempt to reduce strains, thereby leading to the occurrence of different structural isomers resulting in polymorphism. The challenge of predicting a crystalline structure from chemical formula (connectivity of atoms in the molecule) is overcome by the recent advances in molecular mechanics simulations. The useful applications of this methodology in the field of pharmaceutical development has played a vital role in understanding the function and dynamics of the thermodynamically most stable organic crystal polymorph landscape.
Introduction
Organic crystal polymorphs are compounds which exhibit more than one crystalline form of the same molecule, depending on the conformation of their constituent molecules. Polymorphism is the result of different types of bonds and packing existing in the conformers of the same compound which, interestingly, can result in chromatic fragments. This mechanism follows the self-assembly pathway of the organic compound during synthesis and is often referred to as crystal nucleation [1,2]. The growth of the crystal to macroscopic visualization level is dependent on the inter- and intramolecular interactions in the compound. This, in turn, results in the most stable arrangement of atoms which is dependent on the position of each individual atom or molecule and their packing mode. The occurrence of specific structural rearrangements results in various crystalline polymorphs.
Representative examples of structural polymorphs in organic crystals: molecular conformation, packing mode and related aspects
First we refer to the interesting case of 5-Methyl-2-[(2-nitrophenyl) amino]-3-thiophenecarbonitrile which exists as six solventfree polymorphs [3]. Figure 1 presents such compounds where differences in bond arrangements and molecular interactions lead to differences in the overall crystal morphologies. The structural differences among the polymorphs is dependent on the mode of packing and conformation. Yu et al. [3] discovered that the different conformers are dependent on the torsional strain existing between thiophene and the o-nitroaniline fragment which results indifferent crystals colors (yellow, orange, and red). 'Further studies have shown that the most thermodynamically stable conformer is favored. For instance, formation of the polymorphs of bicifadine hydrochloride is dependent on molecular conformation as well as mode of packing. Interestingly, upon heating, a second polymorph is formed, slightly below the melting point [4]. The thermodynamic stability introduces external factors such as temperature-controlled movements resulting in locomotion (walking, rolling and jumping) in the crystals [5]. Also, the structure-property relationship is vital in predicting the different forms a single compound can exhibit. In this context Amplified Spontaneous Emission (ASE) has come into much recent prominence as a powerful technique for characterizing individual crystalline forms of organic compounds [6]. This method is especially effective in predicting conformational polymorphs (isomers). Remarkably, it provides evidence indicating that manipulating the polymorphic structure of organic compounds can reveal their functional properties, and developing structures of polymorphs are solely dependent on the atomic arrangements.

Figure 1.A. The chemical structure of Ritonavir [7]: blue circles denote torsion angles. B. The chemical structure of (E)- 2-benzamido-3-(pyridin-2-yl)acrylic acid [8]. C and D shows the structure of N-hydroxy-N-(5-(iminomethyl)furan-2-yl) hydroxylamine [9].
Role of hydrogen bonds on crystal structure and related physicochemical properties
Inter- as well as intra molecular hydrogen bonds in organic compounds have a crucial role on their crystal structure and physicochemical properties [8]. This is exemplified by the isomers (E)-2-benzamido- 3-(pyridine-2-yl) acrylic acid and (E)-2-benzamido-3-(pyridine-4- yl) acrylic acid (shown in Figure 2), where the isomerization process involves a rearrangement of a pyridinyl ring supporting hydrogen bonding.These two molecules contain a planar benzene ring and are chemically stable.The presence of the intramolecular H-bonding (IHB) in the first molecule, and its absence in the latter, causes remarkable differences in their physiochemical behaviors including solubility, pKa, and optical properties. The crystal structure of the first isomer (see Figure 2, left) shows a seven-membered ring intra-molecular hydrogen bond, which is absent in the other isomer (Figure 2, right). These two isomers show distinctly different physicochemical properties, such as solubility and pKa. The π-conjugation between the pyridinyl and acrylate moieties is extended by IHB, leading to a strong electronic absorption maximum at ca. 340 nm. The O-H…N IHB facilitates occurrence of a photoinduced excited state intramolecular proton transfer (ESIPT), which is a fundamental and extensively studied reaction in organic photochemistry [10,11]. Here, the ESIPT reaction results in ‘two color’ fluorescence with emission peaks at ca. 420 nm and 490 nm in the polar aprotic solvent acetonitrile, and furthermore, the IHB supports the strength of the organic crystals [8].

Figure 2. Rearrangement of a pyridinyl ring supporting intramolecular proton transfer [8].
Intramolecular H-bonding in organic crystals in a donor-acceptor relationship usually ranges between 2.50 ≤ d(O---O) ≤ 2.65 and bonds shorter than 2.50 Å (d(O---O) ≤ 2.50) exhibits very strong H-bonding [12]. Additional bonding in compounds can also affect the angular arrangements of atoms in the compound. These arrangements due to intramolecular forces of attraction and repulsion can also lead to additional bonding or bonding between the same atoms at different angles. This is demonstrated in Figure 2 where it is seen that hydrogen can be directly attached to the nitrogen of the pyridinyl ring. This results in a positive charge on the nitrogen (Figure 2) as it becomes protonated by the dissociated hydrogen from the acrylate part of the molecule. The hydrogen atom, in turn, is partially attracted to the negatively charged oxygen resulting in a different angular shape of the molecule (O(3)-H(12)-N(2) angle is 171.3°).
X-ray diffraction is a common technique used for determining space between atoms in a molecule. X-ray diffraction studies on the compound shown in Figure 2 revealed the distance between O(3)-H(12) to be 1.345 Å, which is 0.425 Å greater than the theoretical length for hydroxyl (OH) in carboxylic acids. This length is also longer than the usual length of N(2)-H(12) (1.145 Å) indicating that H(12) is bonded closer to N(2) of the pyridine than O(3) of the acrylate [8]. The tautomer in Figure 2 is stabilized by the delocalization of the electrons across O(2), and -C=O making it to mainly exist as the proton transferred species (tautomer).
In a study on the analysis of the structure of conformational polymorph, Nangia et. al. [13,14] indicated that X-Ray diffracted distance-angle scatter plots of O-H---O and C-H---O based on the data collected from Cambridge Structural Database implies that polymorphs with larger number of symmetry-independent molecules usually have better interactions than compounds with lower number of symmetry-dependent molecules. This study was done using crystal packing software to demonstrate the overlay of the rings in a molecule supporting the claim that bonding angle and length may result in a fully different crystal. A representation of the polymorphs is provided in Figure 3.
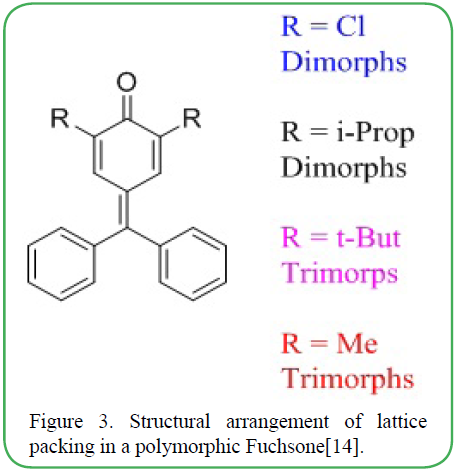
Figure 3. Structural arrangement of lattice packing in a polymorphic Fuchsone[14].
Polymorphism in pharmaceutically relevant compounds
Polymorphs of pharmaceutical drugs are particularly important due to the changes in their characteristics that may occur from one morph to the next. It is predicted that more than 50% of pharmaceuticals have multiple polymorphic forms [15,16]. Some potential drug characteristics that may be affected by polymorphic forms include efficacy, bioavailability, and toxicity [17].
Bioavailability is a major component of drug effectiveness; polymorphs of a drug may affect the solubility of the active ingredient, leading to an increase or decrease in the physiological concentration. The corticosteroid Methylprednisolone is typically used in therapeutic applications for its anti-inflammatory capabilities. Methylprednisolone (Figure 4) has two crystalline polymorphic forms, each of which exhibit different dissolution rates. During in vivo testing it was determined that Form II of methylprednisolone has a 1.2 times greater dissolution rate than the more stable Form I of the drug [18]. A higher dissolution rate can be advantageous when considering the speed at which a drug will start taking effect and the levels to which it will be present.
Another common drug that shows polymorphic forms is acetaminophen (Paracetamol in Europe) (Figure 4). Acetaminophen has three known forms I, II, III [19]. Its unique structure makes it useful in conjugating with caffeine, morphine, phenatanyl and many other opioids of various strengths and delivering to the target [20]. Crystal structure prediction methodology (CSP) [21] is in high prominence for predicting the forms, and stability of many organic crystals and their polymorphs from known organic formulas. This will allow to determine the thermodynamically favorable polymorphs and their ability to bond to a specific substrate (e.g. opioid drugs)for a particular physiological target, and utilize more polymorphs in the pharmaceutical and medical field.
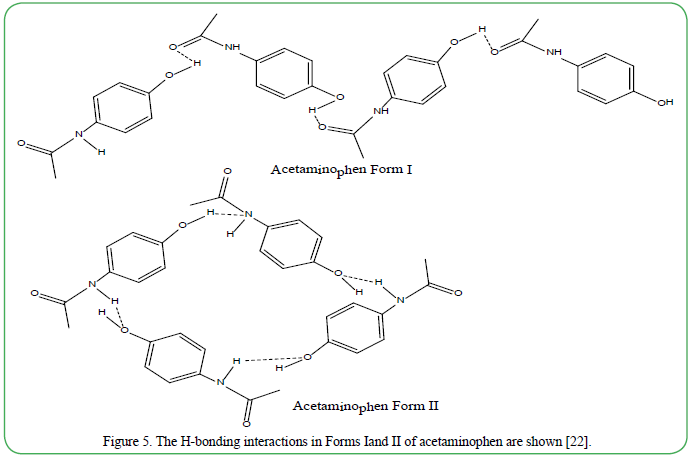
Figure 5. The H-bonding interactions in Forms Iand II of acetaminophen are shown [22].
The most common and stable form I [22,23] (Figure 5) of acetaminophen is a sheet of acetaminophen with a herringbone packing Form I forming zig-zag shapes via (01)O–H-----(02)O=C hydrogen bonds. Form II takes a flatter, plate-like morphology that is more organized than form I due to (01)N–H----O (02) and (01)O–H- ----O(2) intermolecular hydrogen bonds. The resulting structure has better tableting property. Form III is produced as needles organized into distinctive islands bonded much like form II, only orientated vertically [22]. Figure 5 shows the intermolecular hydrogen bonding in forms I and II.
Complex molecules that can form metal-ligand interaction are also affected by modification due to the docking of the metal ion [24]. Isonicotinic acid, for example, can stack together with different divalent metal ions which in turn changes the dimension of the molecular structure of the compound and also results in conformational polymorphism with different neutral layered arrays [24]. The new contorted molecule is now strengthened by additional hydrogen bonding interactions which stabilizes the structure of the compound. Another interesting example of polymorphism include the influence of a chlorine substituent in an aromatic system, such as that in the potential anticancer drug, 4-chloro-2-phenylanthranilic acid (CPAA) (Figure 6). Three polymorphs of CPAA have been discovered. The highly electronegative chlorine can participate in hydrogen bonding, halogen bonding [25] and London dispersion forces [26]. This facilitates the rotation of the benzene ring making the interactions more accessible which thereby tightly pack the structure to global minima [27,28]. The hydrogen bond existing between the acidic portion of the compound 2-((3-chloro-2-methylphenyl)amino) benzoic acid) is shown in Figure 6. Due to twisting and turning of the aniline (Figure 6), H-bond strength varies. The resulting crystal will, therefore, be stronger though it may cause bending in the overall structure. Bending can result in mechanical stress which can be used to predict the mechanical response.
The aliphatic calorie-free sweetener acesulfame (6-methyl-1,2,3- oxathiazin-4(3H)-one 2,2-dioxide) is commonly used in food products, beverages, and in pharmaceutical formulations. Velaga and colleagues [29] discovered that acesulfame exist in two crystalline forms. One of the forms shows mechanical stress bending in crystalline studies. This form is more stable in ambient conditions making acesulfame the first aliphatic organic compound to exhibit stabilized bending phenomenon. Additionally, bond bending and twisting can occur simultaneously, according to Bond, A. et. al., as in the case of aspirin [30]. This may result in particles growing into each other. The crystalline arrangements are very similar such that aspirin crystals intergrow within itself forming inter growth structures [30].
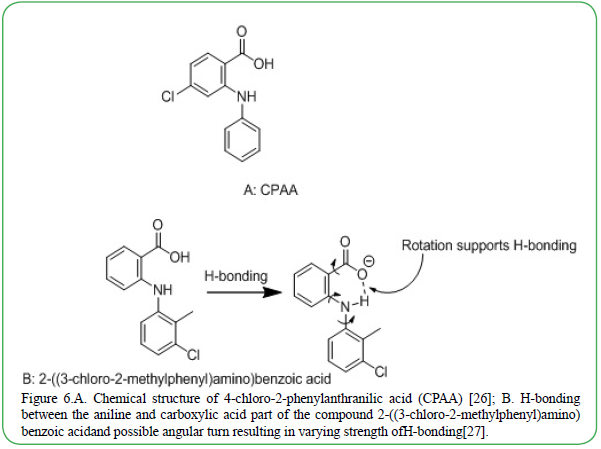
Figure 6.A. Chemical structure of 4-chloro-2-phenylanthranilic acid (CPAA) [26]; B. H-bonding between the aniline and carboxylic acid part of the compound 2-((3-chloro-2-methylphenyl)amino) benzoic acidand possible angular turn resulting in varying strength ofH-bonding[27].
Polymorphism can also occur in engineered cocrystals that may result from intergrowth [31]. In such case, calculated X-Ray powder diffractometric data for single crystal shows a valid relationship between the position of the peak and reflections. Error in measured data arises, however, when the intensity is increased. The difference in the structure of polymorphic organic crystals is determined by several different computational and analytical method including X-ray crystallography [32], Neutron Diffraction [33], BIOVIA Materials Studio Polymorph Predictor [34], and Cambridge Structural Database [35] for retrieving compounds.
Experimental Tools for determining crystal structures
X-ray Crystallography and powder diffraction techniques
Diffraction of X-rays and neutrons provide powerful and complementary tools for determining the structures of organic crystals. X-ray crystallography [32] is a highly reliable method for determining the atomic and molecular structure of organic crystals. This is possible because the atoms can diffract X-ray beams in different directions, generating a pattern which reveals the arrangement of atoms within a crystal structure, thus providing precise information on 3-dimensional structure, including accurate data on bond lengths and bond angles. However, very often, if the crystals are not large enough to permit single crystal diffraction studies, the sample can be analyzed by an alternative (though less informative) technique, namely X-ray powder diffraction (XRPD)[32,36]. A beam of X-rays striking a sample containing randomly oriented micro-crystals (powders) produces a pattern of rings, which is useful for establishing the identity of the solid substance, and as a fingerprint for a particular crystalline phase.
Neutron Diffraction
In view of wave/particle duality, neutrons can be diffracted like X-rays. Unlike X-rays, neutrons do not interact with electron shells, and instead, they interact with the atomic nuclei. Therefore, using neutron diffraction, specific and accurate data can be collected on hydrogen bonds because of the depth of the penetrability of the radiation and its interaction with the atomic nuclei [33]. The quantum interactions and emanation of diffracted particles are measured in small units (Å, or even 10-15m.) making it suitable to measure the interactions of H-bonding and, thus, detecting the hardness of the crystal [33,38]. Use of the neutron diffraction technique increases the likelihood of validating conformational polymorphism. Thus X-ray and neutron diffraction serve as complementary techniques for in depth structural studies on organic crystals.
Solid state NMR and Electron Diffraction techniques
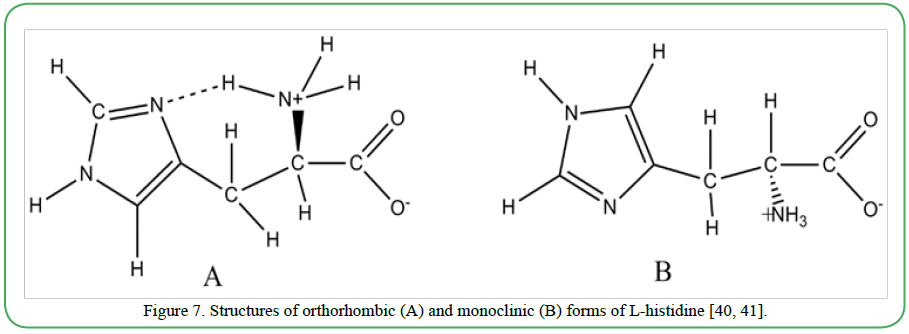
For pharmaceuticals, where samples are frequently available in powder form with micro to submicrometer sized grains, detailed structural analysis is limited by X-ray crystallography and neutron diffraction. Solid state NMR along with electron diffraction (ED) appear to be promising tools with emerging potential in these cases. In a recent study, Oikawa et al. [39] demonstrated the combined use of multidimensional ssNMR and ED to discriminate (in finger print patterns) three crystalline polymorphs of L-histidine namely orthorhombic (form A) and monoclinic (form B) samples of L-histidine (shown in Figure 7), and L-histidine •HCl•H2O. Since intense electron beam induces degradation in organic molecules, very mild level of irradiation was used, to avoid such degradation effects.
BIOVIA Materials Studio Polymorph Predictor
BIOVIA Material studio [34,42] is a computer simulation and modeling software that is useful for predicting potential polymorphs of a compound directly from its molecular structure. Users have a visual image to fully understand and compare polymorphic structures. Analyzing the simulation requires understanding of surface interactions, intra-molecular bonding and bond rotations.
This method of polymorph prediction is applicable to pigments, drug candidates, scale inhibition, and explosives. This is one of the easiest methods of analyzing the polymorphic structures and can be used by almost anyone. The user would simply select the molecule needed and the computer simulates the possible structure arrangement based on the bonding arrangement, reactivity and conformity making it an easy way to detect the varying structure of the crystal. The software also uses molecular packing arrangement space of low-lying minima in lattice energy of the organic crystal [43].
Cambridge Structural Database
The Cambridge Structural Database (CSD) is a large repository of experimentally determined organic and metal-organic crystal structures, based on X-ray and neutron diffraction studies. These data are readily accessible for chemists mainly for experimental purposes in computational chemistry. It is also the most widely used resource for conformational polymorphs of organic crystals. Originally created in 1965, the CSD is being continuously updated by new entries which are constantly been uploaded in the system. The structures are processed computationally by expert structural chemists prior to including in the database [44-46]. Scientists world wide use this system to view three-dimensional structures of conformational polymorphs of organic crystals.
Polymorphic structures are retrievable and attainable in molecular data management systems. These data are validated by numerous experts in computational chemistry to ensure accuracy for other users. Polymorphs can further be validated using X-ray crystallography and neutron diffraction.
Role of external stimuli, and self-healing properties
In response to external stimuli crystal structure rearranges in different forms exhibiting characteristics such as walking, bending, rolling, etc. This characteristic also allows molecular interaction to undergo reverse phase transition: the same locomotion on the opposite form or direction [5,42]. Size of crystals can also affect the movement: Thicker crystals exhibit slow bending and straightening while thinner crystals are able to exhibit these changes at a faster rate. The symmetry of the crystal can also affect mobility of unsymmetrical crystals, and are more capable to promote rolling due to a slanted structure resulting in easier tilting to one side than the other. Thus, the crystal is more probable to roll to the tilted side when the crystal is exposed to temperature changes [5,42]. Furthermore, adjusting the temperature can result in crawling crystals. These crystals are capable of undergoing repeated recrystallization and melting under temperature fluctuations. Fluctuations in temperature may results in different atomic and molecular rearrangements. This results in different polymorphic structures. Moreover, thermodynamic manipulation can weaken or strengthen atomic and molecular interaction in the crystal. The shape of the crystal may deform when heated and reform when cooled. The change in shape and direct position of the temperature change result in overall mobility [5,42].
Recently, Gupta et al. [47] have presented a novel example of smart crystalline material (namely, the 2:1 cocrystal of probenecid and 4,4’-azopyridine) which exhibit reversible response to a variety of external stimuli (UV light, heat, and mechanical pressure), by bending, twisting and elastic deformation. The materials are capable of self-healing on thermal manipulation, involving heating and cooling. These properties are based on reversible trans-cis isomerization and crystal to crystal phase transitions. The importance of self-healing in determining polymorphic structures in a single crystal is receiving increasing attention. Soft crystals also have specific molecular packing. It is, therefore, crucial to find a polymer that can keep its integrity (via intermolecular interactions such as hydrogen bonding, hydrophobic interaction, or interactions between aromatic rings)during heating/cooling transitions [47]. The ability to self-heal allows the structure to regain and alter the shape during both heating and cooling periods or even at room temperature. This is opening the door to manufacturing micro-robotics [5,42,47-49], simulating natural organisms in the biosphere, many of which possess self-healing properties [50]. The mobility of organic crystals under heating or cooling is called inchworm walking or bending. The recorded temperature for bending and twisting in organic crystals ranges between 140 0C and -140 0C. Though there is a correlation between the shape of the crystal and the direction in which it moves, the reason for this relation is not yet established. Latest studies by Taniguchi et al. [42] on “photo-triggered phase transition” is rather interesting and worthy of note. These authors examined photochromic, chiral salicylideneamine crystals. The material exhibits temperature induced single crystal-single crystal phase transition which shows reversibility on heating and cooling. Interestingly, it transforms to the identical phase upon photo-irradiation at temperatures below the thermal transition temperature. The phototriggered phase transition has been attributed to the strain resulting from enol-keto photoisomerization which is facilitated by the small energy barrier associated with alterations in the crystal structures. Taniguchi et al. further reports that the photo-triggered phase is metastable and reverts back to the original stable phase, by a step involving back isomerization from the trans-keto to enol form [42].
Summary
Literature data suggests that one-third of organic compounds and about 80% of marketed pharmaceuticals show some kind of polymorphism where the initial species converts to subsequent crystal polymorphs depending on the crystallization conditions. Thus crystal structure prediction (CSP) plays a vital role in understanding the crystal energy landscape and screening polymorphs. X-Ray diffraction and neutron diffraction can be used to verify the conformational characteristics of the organic polymorphic crystals. The more accurate of the two experimental methods is neutron diffraction due to the in-depth penetrability of radiation in the molecular structure detecting specific bond angles and length especially in hydrogen exchange (H/D). The ability to predict possible polymorphs, as well as anticipate its stability, opens the possibility of conceptualizing the feasible application of the hypothetical polymorphs. Thermodynamics has subtle interplay between the molecular conformations and crystal packing, to transition organic crystals into more than one sister polymorphs. Polymorphic forms of therapeutic drugs may lead to increased performance in some applications. Future research into drug crystalline polymorphs has the potential to unlock hidden characteristics of common pharmaceuticals.
Acknowledgements
This publication was made possible by an Institutional Development Award (IDeA) from the National Institute of General Medical Sciences of the National Institutes of Health under Grant #P20GM103476. BS acknowledges NSF Research Initiation Award under HBCU-UP program: Award number (FAIN): 1800732 and NSF Target Infusion Award under HBCU-UP program: Award number (FAIN): 1818528 for the summer and student support at Tougaloo College. BS also thanks the Department of Chemistry and Biochemistry at Stephen F. Austin University for the student support.
References
Davey, RJ., Schroeder, SL.,& ter Horst, JH. (2013). Nucleation of Organic Crystals—A Molecular Perspective. Angew. Chem Int Ed 52: 2166-2179.View
Back, KR., Davey, RJ., Grecu, T., Hunter, CA.,& Taylor, LS. (2012). Molecular Conformation and Crystallization: The Case of Ethenzamide. Crystal Growth & Design 12: 6110-6117.View
Yu, L., Stephenson, GA., Mitchell, CA., Bunnell, CA.,& Snorek, SV. (2000). Thermochemistry and Conformational Polymorphism of a Hexamorphic Crystal System. J Am Chem Soc 122: 585-591.View
McArdle, P., Gilligan, K., Cunningham, D., Dark, R.,& Mahon, MA. (2004) method for the prediction of the crystal structure of ionic organic compounds—the crystal structures of o-toluidinium chloride and bromide and polymorphism of bicifadine hydrochloride, Cryst Eng Comm 6: 303-309.View
Taniguchi, T., Sugiyama, H., Uekusa, H., Shiro, M.,& Asahi, T. (2016). Walking and rolling of crystals induced thermally by phase transition, Nature Commun. 9: 538 (1-8).View
Wang, K., Zhang, H., Chen, S., Yang, G.,& Zhang, J. (2014). Organic Polymorphs: One-Compound-Based Crystals with Molecular-Conformation and Packing-Dependent Luminescent Properties. Adv Mater 26: 6168-6173.View
Bauer, J., Spanton, S., Henry, R., Quick, J.,& Dziki, W. (2001). An Extraordinary Example of Conformational Polymorphism. Pharmaceutical Res 18: 859-66.View
Pesyan, NN. (2012). Crystal Structures of Organic Compounds, chapter 9, Recent Advances in Crystallography, Intech Open 191-242.View
Pogoda, D., Janczak, J.,& Videnova-Adrabinska, V. (2016). New Polymorphs of an Old Drug: Conformational and Synthon Polymorphism of 5-Nitrofurazone, Acta Crystallogr B Struct Sci CrystEng Mater 72: 263-273.View
Sengupta, B.,& Sengupta, PK. (2002). The interaction of quercetin with human serum albumin: a fluorescence spectroscopic study. Biochem Biophys Res Commun 299: 400-403.View
Sengupta, PK.,& Kasha, M. (1979). Excited state proton-transfer spectroscopy of 3-hydroxyflavone and quercetin.Chem. Phys Lett 68: 382-385.View
Gilli, P., Bertolasi, V., Pretto, L., Ferretti, V.,& Gilli, G. (2004). Covalent versus Electrostatic Nature of the Strong Hydrogen Bond: Discrimination among Single, Double, and Asymmetric Single-Well Hydrogen Bonds by Variable-Temperature X-ray Crystallographic Methods in β-Diketone Enol RAHB Systems, J Am Chem Soc 126: 3845-3855.View
Chandran, SK., Nath, NK., Roy, S.,& Nangia, A. (2008). Polymorphism in Fuchsones. Crystal Growth & Design 8: 140-154.View
Nangia, A. (2008). Conformational Polymorphism in Organic Crystals. Accounts Chem Res 41: 595-604 .View
Raza, K., Kumar, P., Ratan, S., Malik, R.,& Arora, S.(2014) Polymorphism: The Phenomenon Affecting the Performance of Drugs. SOJ Pharm Pharm Sci 1: 10.View
Haleblian, J.,& Walter, M. (1969). Pharmaceutical applications of polymorphism. J Pharm Sci 58.View
Lee, EH. (2014). A practical guide to pharmaceutical polymorph screening & selection. Asian J Pharma Sci 9: 163–175.View
Corvis, Y., Négrier, P., Soulestin, J.,& Espeau, P. (2016). New Melting Data of the Two Polymorphs of Prednisolone. J Phys Chem B 120: 10839-10843.View
Ehmann, HMA.,& Werzer, O. (2014). Surface Mediated Structures: Stabilization of Metastable Polymorphs on the Example of Paracetamol, Crystal Growth & Design 14: 3680–3684.View
Raffa, RB., Pergolizzi, JV., Segarnick, DJ.,& Tallarida, RJ. (2010). Oxycodone combinations for pain relief, Drugs Today (Barc) 46: 379–398.View
Pawanpreet, S.,& Chadha, R. (2017). Crystal Structure Prediction In The Context Of Pharmaceutical Polymorph Screening And Putative Polymorphs Of Ciprofloxacin, Int J Pharm Pharma Sci 9: 1.View
Du, JJ., Lai, F., Váradi, L., Williams, PA.,& Groundwater, PW.(2018). Monoclinic Paracetamol vs. Paracetamol-4,40- Bipyridine Co-Crystal; What Is the Difference? A Charge Density Study Crys 8: 46.View
Peterson, ML., Morissette, SL., McNulty, C., Goldsweig, A.,& Shaw, P.(2002). Iterative High-Throughput Polymorphism Studies on Acetaminophen and an Experimentally Derived Structure for Form III. J Am Chem Soc 124: 10958–10959.View
Chen, XD., Wu, HF., Zhao, XH., Zhao, XJ.,& Du, M. (2007). Metal−Organic Coordination Architectures with Thiazole- Spaced Pyridinecarboxylates: Conformational Polymorphism, Structural Adjustment, and Ligand Flexibility. Crys Growth Design 7: 124-131.View
Szell, PMJ., Zablotny, S.,& Bryce, DL. (2019). Halogen bonding as a supramolecular dynamics catalyst, Nature Comm 10: 916.View
Liu, M., Yin, C., Chen, P., Zhang, M.,& Parkin, S.(2017). sp2- --Cl Hydrogen Bond in the Conformational Polymorphism of 4-chloro-phenylanthranilic acid. Crys Eng Comm 19: 4345.View
Uzoh, OG., Cruz-Cabeza, AJ.,& Price, SL. (2012). Is the Fenamate Group a Polymorphophore? Contrasting the Crystal Energy Landscapes of Fenamic and Tolfenamic Acids. Crys Growth Design 12: 4230-4239.View
Gilli, P., Bertolasi, V., Ferreti, V.,& Gilli, G. (1994). Covalent Nature of the Strong Homonuclear Hydrogen Bond. Study of the O–H O System by Crystal Structure Correlation Methods, J Am Chem Soc 116: 909.View
Velaga, SP., Vangala, VR., Basavoju, S.,& Boström, D. (2010). Polymorphism in Acesulfame Sweetener: Structure-Property and Stability Relationships of Bending and Brittle Crystals. The Royal Soc Chem 46: 3562-3564.View
Bond, A., Boese, R.,& Desiraju, G. (2007). On the Polymorphism of Aspirin: Crystalline Aspirin as Intergrowths of Two “Polymorphic” Domains. Angewandte Chemie Int Ed 46: 618- 622.View
Zerkowski, JA., MacDonald, JC.,& Whitesides, GM. (1997). Polymorphic Packing Arrangements in a Class of Eng Organic Crys Chem Mater 9: 1933-1941.View
Taylor, R.,& Kennard, O. (1982). Crystallographic Evidence for the Existence of C-H…O, CH…N, and C-H…Cl Hydrogen Bonds, J Am Chem Soc 104: 5063.View
Vishweshwar, P., Babu, NJ., Nangia, A., Mason, SA.,& Puschmann, H.(2004). Variable Temperature Neutron Diffraction Analysis of a Very Short O-H O Hydrogen Bond in 2,3,5,6-Pyrazinetetracarboxylic Acid Dihydrate: Synthon- Assisted Short Oacid-H····Owater Hydrogen Bonds in a Multicenter Array, J Phys Chem A 108: 9406-9416.View
Day, GM. (2011). Current approaches to predicting molecular organic crystal structures, Crystallo Rev 17: 1, 3-52.View
Allen, FH. (2002). The Cambridge Structural Database: A Quarter of a Million Crystal Structures and Rising. Acta Crystallogr B58: 380–388.View
Day, GM., Streek, J., Bonnet, A., Burley, JC.,& Jones, W. (2006). Polymorphism of Scyllo-Inositol: Joining Crystal Structure Prediction with Experiment to Elucidate the Structures of Two Polymorphs, Crys Growth Design 0: 1-7.View
Ni, L.,& Yong, G. (2018). Crystal Structures, Phosphorescent and Magnetic Properties of Novel 1,2-dihyroisoquinoline radicals. J Mol Struct 1171: 614-618.View
Fucke, K.,& Steed, JW. (2010). X-ray and Neutron Diffraction in the Study of Organic Crystalline Hydrate, Water 2: 333-350.View
Oikawa, T., Okumura, M., Kimura, T.,& Nishiyama, Y. (2017). Solid-state NMR meets electron diffraction: determination of crystalline polymorphs of small organic microcrystalline samples, Acta Cryst C 73: 219–228.View
Madden, JJ., McGandy, EL.,& Seeman, NC. (1972). The crystal structure of the orthorhombic form of L-(+)-histidine, Acta Cryst B 28: 2377-2382.View
CMadden, JJ., McGandy, EL., Seeman, NC., Harding, MM.,& Hoy, A.(1972). The crystal structure of the monoclinic form of L-histidine, Acta Cryst B 28: 2382-2389.
Taniguchi, T., Sato, H., Hagiwara, Y., Asahi, T.,& Koshima, H. (2019). Photo-triggered phase transition of a crystal, Comm Chem 2: 19.View
Lutker, KM., Quiñones, R., Xu, J., Ramamoorthy, A.,& Matzger, AJ. (2011). Polymorphs and Hydrates of Acyclovir. J Pharmaceu Sci 100: 949-963.View
Price, SL. (2009). Computed Crystal Energy Landscapes for Understanding and Predicting Organic Crystal Structures and Polymorphism. Accounts Chem Res 42: 117-126.View
Bruno, IJ., Cole, JC., Edgington, PR., Kessler, M.,& Macrae, CF. (2002). New software for searching the Cambridge Structural Database and visualizing crystal structures, Acta Cryst B 58: 389-397.View
Groom, CR., Bruno, IJ., Lightfoot, MP.,& Ward, SC. (2016). The Cambridge structural database, Acta Cryst B 72: 171–179.View
Gupta, P., Karothu, DP., Ahmed, E., Naumov, P.,& Nath, NK. (2018). Thermally Twistable, Photobendable, Elastically Deformable, and Self-Healable Soft Crystals. Angewandte Chemie 57: 8498-8502.View
Zhang, L., Bailey, JB., Subramanian, RH., Groisman, A.,& Tezcan, FA. (2018). Hyperexpandable, self-healing macromolecular crystals with integrated polymer networks. Nature 557: 86–91.View
Guangfeng, L., Ye, X., Liu, J.,& Nie, L. (2016). Self-Healing Behavior in a Thermo-Mechanically Responsive Cocrystal during a Reversible Phase Transition,Angewandte Chemie Int Ed 56.View
Karothu, DP., Weston, J., Desta, IT.,& Naumov, P. (2016). Shape- Memory and Self-Healing Effects in Mechanosalient Molecular Crystals, J Am Chem Soc 138: 13298-13306.View