Current Research in Materials Chemistry Volume 3 (2021), Article ID: CRMC-109
https://doi.org/10.33790/crmc1100109Research Article
Paper-Based Portable Sensor and Nanosensor For Sulfur Dioxide Detection
Thuy Le1, Samantha Macchi1, Amanda Jalihal1, Sylvia Szwedo1, and Noureen Siraj1*
1Department of Chemistry, University of Arkansas at Little Rock, 2801 S. University Ave., Little Rock AR 72204, USA.
Corresponding Author Details: Noureen Siraj, Department of Chemistry, University of Arkansas at Little Rock, 2801 S. University Ave., Little Rock AR 72204, USA. E-mail: nxsiraj@ualr.edu
Received date: 08th March, 2021
Accepted date: 06th April, 2021
Published date: 08th April, 2021
Citation: Le, T., Macchi, S., Jalihal, A., Szwedo, S., & Siraj N. (2021). Paper-Based Portable Sensor and Nanosensor For Sulfur Dioxide Detection. Cur Res Mater Chem 3(1):109.
Copyright:©2021, This is an open-access article distributed under the terms of the Creative Commons Attribution License 4.0, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited
Abstract
Sulfur dioxide (SO2) pollution has become an increasing issue world-wide as it is produced both naturally and as industrial waste. Thus, it is critical to develop a sensor and detection methods to analyze SO2 in the atmosphere. In order to design and generate an effective sensor that detects low levels of SO2, fuchsine dyes have been used as a potential sensor material. New hydrophobic derivatives of Pararosaniline hydrochloride (pR-HCl) is developed to further improve the sensitivity of fuchsine dyes towards SO2 gas. It has been shown that these dyes can provide an economic and efficient colorimetric detection of SO2. In this work, (pR-HCl) is converted into an ionic material (IM) via a facile ion exchange reaction with bis (trifluoromethane) sulfonamide (NTF2) counterion. The new, hydrophobic derivative, pararosaniline bis (trifluoromethane) sulfonamide (pR-NTF2) IM was converted into stable aqueous ionic nanomaterials (INMs) by a reprecipitation method. Examination of absorption spectra results revealed that pR-NTF2 IM exhibits enhanced molar absorptivity in comparison to the parent dye (pR-HCl). The improved photophysical properties allowed a framework for a highly sensitive nanosensor for detection of SO2. A paper based portable SO2 sensor was also developed and tested for its ability to colorimetric detection of SO2. The cost effective and stable paper-based sensor exhibited the rapid response to decolorize the fuchsine dyes in few seconds as compared to their parent compound.
Keywords: SO2 Detection, Portable and Low-cost Sensor, Nanosensor.
Introduction
Air pollution has contributed many issues to the global warming and human health. Among those air pollutants, sulfur dioxide (SO2) is a very toxic gas that has a sharp odor. SO2 is released in the process of generating electricity from sulfur-containing non-renewal energy sources (coal, oil, and gas), the fuel combustion in vehicles, and the extraction of metal ores [1]. SO2is emitted from natural processes such as volcanic eruptions, pollen grains [2] and natural decays [3]. The presence of SO2 gas in the atmosphere also cause acid rain when the rain water reacts with SO2 gas in air [4]. Since SO2 is highly soluble in water, it easily absorbs into respiratory tract and eyes of human where it is converted into sulfurous acid (H2SO3). In addition, a short-term exposure to SO2 causes irritation in nose, throat, lungs and respiratory tract in human. The U.S. Environmental Protection Agency (EPA) has listed SO2 as one of the six common air pollutants that needed to set National Ambient Air Quality Standards (NAAQS) to protect human health and environment [1]. According to U.S. Department of the Interior National Park Services [5], SO2 becomes toxic to human health when its concentration exceeds from 5 parts per million (ppm) in the duration of 15 minutes [5]. It suggests that a very low concentration of SO2 can cause many adverse health and environmental effects. For this reason, monitoring SO2 at low concentrations has gained attention to protect environment, living organisms as well as human health.
Various methods have been developed for monitoring SO2 such as acidimetry, conductimetry, colorimetry, flame photometry, potentiometry and coulometry [6]. The instruments such as gas chromatography, Fourier Transform Infrared (FTIR), Ozone Monitoring Instrument (OMI), etc. was also used to detect the presence of SO2. However, these instruments are very expensive, and are difficult to use in the field to monitor SO2. Developing an inexpensive portable sensor that is highly selective and sensitive (at ppm level) to SO2 is necessary for the prevention of the adverse effects of SO2 on human, environment, and other living organisms. Fuchsine dyes like pararosaniline hydrochloride (pR-HCl) and rosaniline hydrochloride (R-HCl) have been widely used as colorimetric sensor molecules for the detection of atmospheric SO2 gas pollution [7,8]. These dyes play a significant role in the regulation of SO2 gas by providing an economical and relatively efficient colorimetric method for determining SO2. However, further improvement in these dyes can enhance the sensitivity of fuchsine dyes towards SO2 detection.
Ionic materials (IMs) have been attracted great attention due to their unique tunable photophysical property, high thermal stability, environmentally friendly nature and economic synthesis approach [9]. Therefore, many fluorescent [10], magnetic [11], colorimetric [12], hydrophobic [13]. IMs have been developed for variety of applications such as sensor [12], solar cells [14], organic light emitting diodes (OLEDs) [15], cancer therapy [16]. IMs with extended π-conjugated system can shift the absorption or emission wavelength maxima of the molecule [17]. Simply by changing the counterion of the IMs, improved photophysical and electronic property can be attained [18,19]. Moreover, hydrophobic characteristics can be introduced to molecules which aids to develop stable nanostructures termed ionic nanomaterials (INMs) via simple methodology such as reprecipitation [20-23].
In the recent year, nanoparticles are getting tremendous interest of many researchers due to its amazing characteristics such as optical, electrical, and magnetic property [24,25]. Several methods have been explored to design the nanomaterials [22,26]. The size, shape, and surface charge of nanoparticles play an important role in many different applications such as biomedical, industrial, pharmaceutical, environment, electronics, textiles, energy, and sensing use [10,23,27]. It has been shown that nanoparticle-based sensors have several advantages over others such as greater sensitivity and faster response time [28]. However, the typical nanostructures used are metal based (gold, silver, etc.) which are expensive and not environmentally friendly [29]. Thus, implementing nanostructures based on organic molecules could prove to be an economic way to introduce effective sensors of pollutants.
In this project, a new hydrophobic derivative of pR-HCl is synthesized by combining pR cation with bis (trifluoromethane) sulfonamide (NTF2) anion via ion exchange and applied as a stable nanosensor. The INMs were developed using pR-NTF2 in water. The nanoparticle-based sensor for detection of SO2 using fuchsine dyes is sought out for the first time. Photophysical characterization of parent compound, IMs and INMs are studied in detail to investigate the colorimetric sensor performance of pR based IMs and INMs. Herein, a new, simple, and inexpensive approach is introduced to tune the hydrophobicity and photophysical properties of fuchsine dye which can impact the sensitivity of the dyes towards SO2. Moreover, the potential application to use the hydrophobic dye to develop an inexpensive, stable and portable paper-based sensor for prompt detection of SO2 is presented. This study can be used to developed highly efficient nanoparticles or hydrophobic potable, stable sensor. Quantitative analysis will be performed in future.
Materials and methods
Materials
pR-HCl, R-HCl and sodium bis (trifluoromethanesulfonyl) imide (NaNTF2) were purchased from Sigma-Aldrich and their chemical structures are presented in Figure 1. The 18 MΩ-cm triple deionized ultrapure distilled (DI) water was used. Sulfuric acid (H2 SO4), ethanol (EtOH), dichloromethane (DCM) and copper (Cu) were purchased from VWR. Plastic cuvet was used for Dynamic Light Scattering (DLS). 150 mesh copper grids with a formvar/carbon coating (EMS cat# FCF150-Cu) were used for transmission electron microscopy (TEM) imaging.
Synthesis of new hydrophobic derivatives of fuchsine dyes
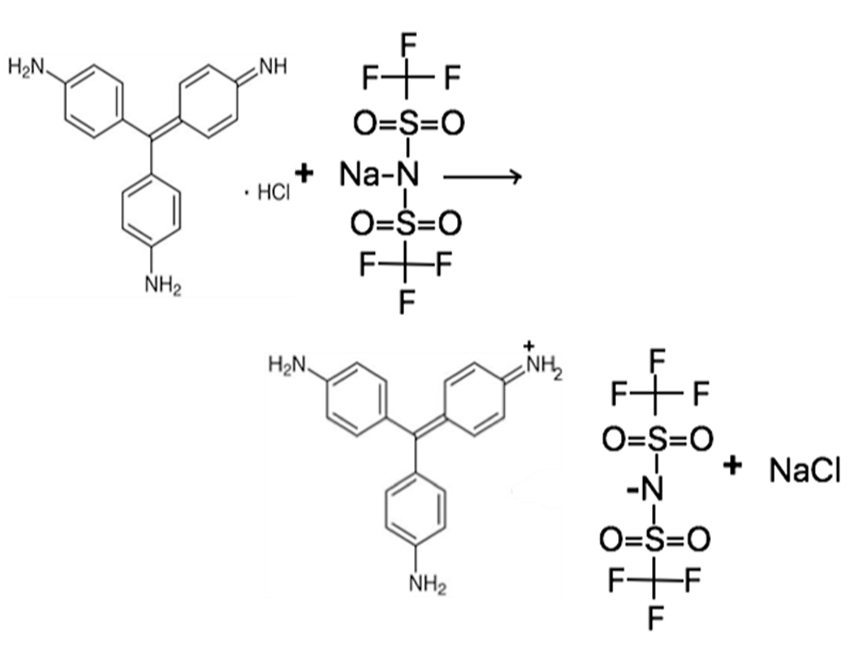
Hydrophobic compound was synthesized using a simple and single step ion-exchange method. A 1:1 ratio of parent dyes (pR-HCl) to NaNTF2 were dissolved in water, stirred for 24 hours (Figure 2). After 24 hours, hydrophobic compound (pR-NTF2) was extracted with DCM, then DCM was evaporated via Rotary Evaporator (Rotavap) and the final product was freeze dried in the lyophilizer for 24 hours to remove any excessive moisture. In result, the hydrophobic compounds of pararosaniline bis (trifluoromethanesulfonyl) imide (pR-NTF2) were obtained as depicted in Figure 2.
Synthesis of nanoparticles
Reprecipitation method was used to prepare INMs of pR-NTF2 as described earlier [20,30]. Briefly, a concentrated solution was prepared in ethanol and dropwise added to vials containing DI water while under sonication waves for 5 minutes. The INMs were allowed a 20 minutes rest time and characterized using TEM and DLS. INMs’ photophysical characteristics were studied in detail to investigate their sensitivity towards detection of SO2.
Instrumentation
The Rotavapor Buchi RE 111 was used to evaporate the solvent after the synthesis of the IMs. The newly synthesized hydrophobic derivative is characterized using ESI high-resolution mass spectrometry (Shimadzu IT-TOF) or MS. Nanoparticles are prepared using reprecipitation methods via Fisher Scientific FS20H ultrasonicator. The morphology of nanoparticles are characterized using a FEI Tecnai F20 80kV TEM and a Broo khaven NanoBrook 90plus Zeta for DLS [7]. INMs of pRNTF2 were prepared in water to investigate the hydrodynamic diameter and dry diameter by using DLS and TEM respectively. Thermogravimetric analysis (Mettler Toledo) or TGA was performed to analyze the thermal stability of synthesized material. Samples were heated in air at a rate of 10oC/ min over a range of 25-800 oC and were plotted as a function of mass lost. A plot of the 1st derivative of mass lost is used to determine the onset degradation temperature, Tonset. The single beam Varian Cary 60 UV-Vis-NIR absorption spectrophotometer was utilized for photophysical characterization. A 2-sided quartz cuvette was used for absorption experiment. The changes in the absorption spectra of newly developed IMs and INMs are recorded and compared with parent compounds. All parent samples, IMs and INMs absorption spectra upon exposure with SO2 are recorded to investigate the most sensitive sensor for prompt detection of SO2
Synthesis of SO2 gas
An apparatus was constructed using a triconnected test tubes hooked on a ring stand as depicted in Figure S1 in the supporting information to effectively generate SO2 gas from the chemical reaction presented in Eq. 1. Cu trimmings and H2 SO4 were placed in the middle test tube chamber where it generates copper sulfate (CuSO4), water and SO2 gas as a result of chemical reaction in the presence of heat. A plastic tube is used to connect the test tube with a vial to transfer SO2 gas from the test tube into the vial containing fuchsine parent dyes solution, IM solution, INM dispersion and filter paper coated with these sensor dyes.
Cu + 2H2SO4 Δ→ CuSO4+ 2H2O + SO2 Eq. 1
Preparation of portable paper-based sensor and nanosensor
A solution of R-HCl, pR-HCl were prepared in EtOH and water separately and used to develop filter paper-based sensor. pR-NTF2 was prepared in EtOH and water separately to develop paper-based sensor and nanosensor coated filter paper respectively. Strips of filter paper (ashy) were soaked into the dye solution and nanoparticles of pR-NTF2 for one hour to allow adequate absorption onto the paper. The paper is then removed from the solution, dried and placed into the vial connected to the apparatus generating SO2.
Results and discussions
IMs and INMs Characterizations
MS
Newly synthesized pR-NTF2 IMs was confirmed via high-resolution MS. Based on the mass to charge (M/Z) ratio of fragment ions in positive and negative ion mode, the mass of pR-NTF2 can be evaluated. The observed peaks (Figure S2) from positive ion mode and negative ion mode were 288.15 and 279.92, respectively. These values matched with theoretical molecular weight of pR cation (288.37 g/mol) and NTF2 anion (280.15 g/mol) indicating the pR-NTF2 was successfully synthesized via ionexchange method. Thermal stability of IMs was also investigated and presented it in SI.
TGA
The newly synthesized compound is investigated for its thermal stability using TGA. Thermal stability curve of pR-NTF2 was generated by heating under continuous air flow from 25-800 oC (Figure S3). pR-NTF2 possesses one major degradation occurring at 495 oC. This indicates that upon conversion from chloride salt to NTF2 IM, there is not a significant loss in thermal stability.
TEM
TEM is used to visualize the shape and dimension of nanoparticles in the absence of any media. To prepare the TEM grid, a small aliquot of pR-NTF2 INMs was drop casted onto a hydrophobic copper grid. After drying, the grids are analyzed to determine particle size and morphology using TEM instrument. As shown in Figure S4, the pR-NTF2 INMs are spherical in shape and have an approximate diameter in the range of 180-200 nm.
DLS
DLS is used to determine the size distribution of solvated nanoparticles. Average hydrodynamic diameter was found to be 215.12 ± 3.95 nm with a polydispersity index of 0.178 ± 0.020. Such a low polydispersity index indicates that the nanoparticles are uniform in size. The zeta potential value was found to be -28.46 ± 3.48 mV indicating that the INMs are colloidally stable and possess a negative surface charge. This negative charge indicates the presence of NTF2 anion on the surface of nanoparticles while the pR cation dye is mostly present at the core of the nanoparticles.
Photophysical characterization and sensing of SO2 gas by fuchsine dyes and hydrophobic derivative
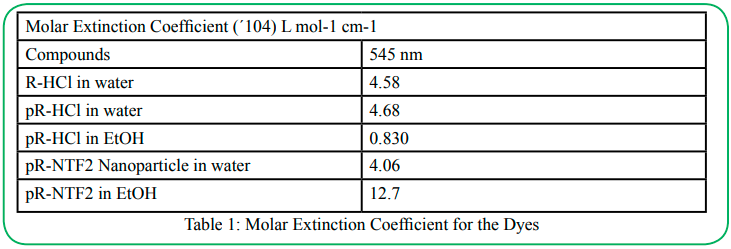
The detailed photophysical properties of all parent dyes and synthesized IM and their nanoparticles are recorded with and without exposure to SO2. Two solvents, water and EtOH were used. The dye absorption wave length maxima of R-HCl, pR-HCl, and pR-NTF2 have been observed primarily at 545 nm wavelengths while a shoulder is observed at 504 nm. The normalized absorption spectra of all compounds are shown in Figure 5a. A slight shift in absorption wavelength maxima is observed from 550 nm to 545 nm upon changing the solvent from EtOH to water. In addition, the changes in molar absorptivity is also recorded. The molar extinction coefficient has been calculated at wavelengths maxima. The results for molar extinction coefficients for all dyes in different solvents are as tabulated in Table 1.
Detailed examination of results revealed that pR-NTF2 exhibited the highest value of molar absorptivity as compared to its parent compound. Thus, it proved that replacement of small chloride ion with a bulky NTF2 ion significantly improved the absorption characteristics of fuchsine dyes. Therefore, it is expected that a highly sensitive sensor can be developed using IMs and INMs which also permit to develop a portable paper-based sensor for the toxic SO2.
Absorption spectra of R-HCl, pR-HCl, pR-NTF2 and INMs were recorded when exposed to SO2 gas to determine the sensitivity of compounds towards SO2 detection in solution. The changes in photophysical properties of the compounds upon exposure of SO2 at different time intervals were recorded.
In EtOH solution, the absorption intensity of R-HCl (Figure 5b) was not changed after increasing the exposure time with SO2 gas. The dye is still very colorful which shows the R-HCl limit to sense SO2. For pR-HCl (Figure 5d), the absorption of pR-HCl was slightly decreased after 30 seconds exposure to SO2 while after exposure of 45 seconds no further decrease in absorbance intensity is observed. This result shows the limitation of pR-HCl for used as a sensor for SO2 detection in ethanolic solution.
In aqueous solution, the absorption of R-HCl (Figure 5c) was dramatically decreased as the exposure time of SO2 increased and the peak maxima was red-shifted. After 60 seconds, the absorbance was quenched completely. The longer the exposure of SO2 to the strong magenta color of R-HCl, the more colorless it becomes. The absorption of pR-HCl (Figure 5e) in water is recorded upon exposure to SO2 at different time interval. A significant decreased in intensity is observed after 45 seconds exposure to SO2. This appeared to be more time sensitive than R-HCl in water. Then at 80 seconds the strong magenta color disappeared suggesting that SO2 completely decolorize the dye.
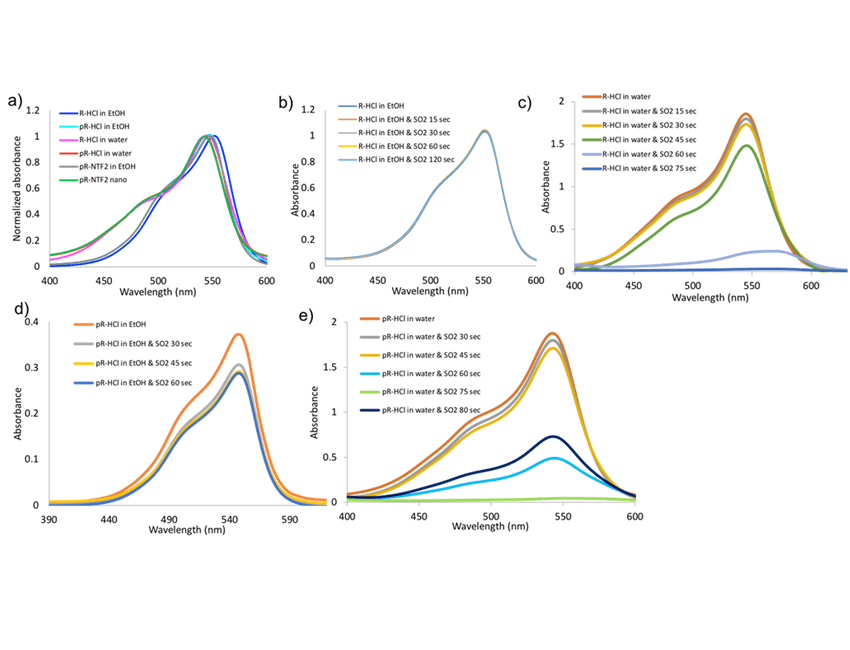
Figure 5. (a) The normalized absorption spectra of parent compounds, IMs and its INMs in different solvents. The absorption spectra of R-HCl in (b) EtOH and (c) water. The absorption spectra of pR-HCl in (c) EtOH and (d) water
In order to prove that decolorization for R-HCl and pR-HCl is caused by SO2 and not because of instability dye in water, photostability test of R-HCl and pR-HCl is also performed in water to prove the stability of the solution over the time span of 60 minutes (Figure S5a, S5b). Minimal to no differences in the absorption spectra are observed which proves R-HCl and pR-HCl dye are stable in water.
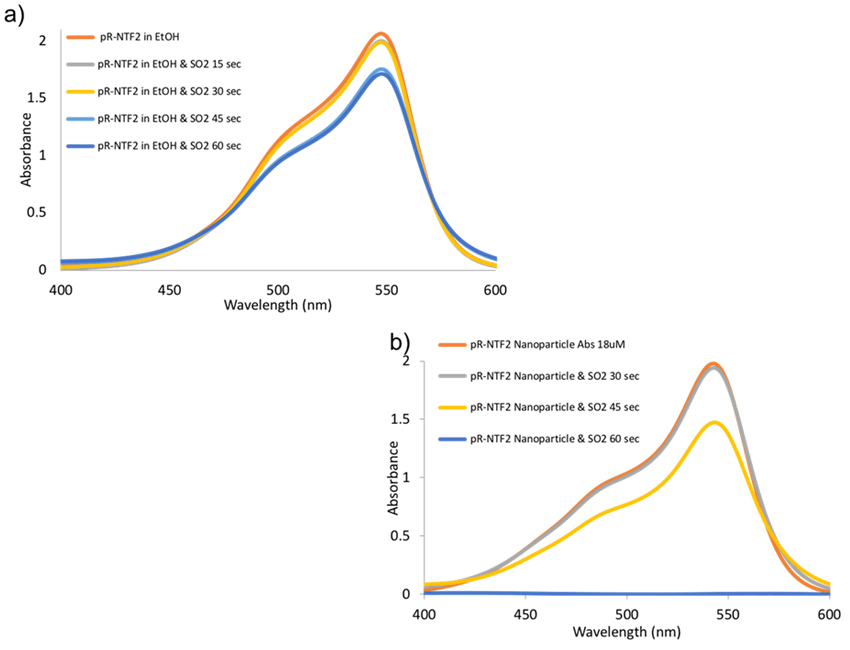
Hydrophobic IMs, pR-NTF2, has demonstrated a higher molar extinction coefficient (Table 1) which could be used as a highly sensitive colorimetric sensor for SO2. After full characterization and performing the sensor response of parent fuchsine dyes, IMs are tested as a sensor for SO2 detection (Figure 6). The absorption ntensity of pR-NTF2 in EtOH solution slowly decreases after prolonged exposure to SO2 (Figure 6a). However, when pR-NTF2 inanoparticle in water (Figure 6b) was exposed to SO2, a significant decrease in the absorbance intensity was observed within 15 seconds time lapse. pR-NTF2 nanoparticles exhibited a tremendous decreased in absorbance intensity after 45 seconds when exposed to SO2 gas. At 60 seconds, the magenta color was completely colorless.It indicates the sensitive performance of nanoparticles towards SO2 sensing. The stability of the pR-NTF2 nanoparticle was also performed and demonstrated in Figure S5c. Absorbance spectra is recorded after every 20 minutes and minimal differences in the absorption spectra has been observed, indicating that the pR-NTF2 nanoparticles are stable.
Sensing results using portable filter paper sensor
The ultimate goal of the study was to develop a low-cost portable sensor to detect SO2. Examination of results obtained using paper-based sensor with parentdyes (prepared in EtOH) (Figure S6) indicated that both R-HCl and pR-HCl took approximately 20 seconds to start reacting with SO2 and became colorless after 30 seconds. However, results of pR-HCl and R-HCl paper-based sensor (prepared in water) (Figure S7) became colorless after 20 seconds when exposed to SO2. In comparison to parent dyes’ sensor, pR-NTF2 IMs paper-based sensor showed a very rapid response and decolorize the dyes in less than 10 seconds when exposed with SO2 (Figure 7a). This proves that the paper-based sensor from the newly synthesized compound, pR-NTF2, was successfully enhanced the sensitivity of colorimetric sensor for SO2 detection due to improved photophysical properties. When the nanosensor of pR-NTF2 were exposed to SO2, an even shorter amount of time was required to detect the SO2 gas as shown in Figure 7b. Furthermore, filter paper-based sensor developed using hydrophobic INMs are more stable towards moisture and can be stored for a long time as compared to their hydrophilic parent compound. Thus, IM approach to develop a hydrophobic compound does not only improve the photophysical properties which enhanced the sensitivity of fuchsine dyes towards SO2 but it also permits to develop a stable, economical, and portable paper-based sensor which can easily be used in the field for rapid detection of SO2.

Figure 7. Exposure of SO2 for 10 seconds (a) pR-NTF2 (prepared in EtOH) and (b) pR-NTF2 nanoparticle (prepared in water)
Conclusion
The hydrophobic derivative pR-HCl, was successfully synthesized via ion-exchange reaction. Absorption characteristics significantly improved by changing the counterion from chloride to NTF2-. The molar extinction coefficient of pR-NTF2 was dramatically enhanced indicating the potential to develop a highly sensitive colorimetric method for SO2 detection. In the study, the hydrophobic derivative, pR-NTF2 was used to develop nanoparticle-based sensor using fuchsine dye for SO2 detection for the first time. INMs of pR-NTF2 has demonstrated the quick response and rapid decolorization of fuchsine dyes when exposed to SO2 gas. The hydrophobic derivative of fuchsine dye, pR-NTF2 permits the synthesis of stable paper-based sensor. A portable sensor was developed using inexpensive filter paper which can be used as an economical and stable sensor for SO2 sensing. In future, many different anions can be combined with R-HCl and pR-HCl using IM approach to alter the hydrophobicity and photophysical characteristics of the synthesized product which can further tune the sensitivity of the sensor. The portable based sensor approach using the hydrophobic nanoparticles is thus successfully demonstrated.
Conflict of interests:
There is no conflict of interest.
Acknowledgements
This material is based upon work supported by the National Science Foundation EPSCoR Research Infrastructure under award number RII Track 4-1833004. We also acknowledge the funding from the UA Little Rock signature experience award and UA Little Rock McNair Scholar program.
References
EPA. (2019). Sulfur Dioxide (SO2) Pollution. Retrieved from United States Environmental Protection Agency website: https:// www.epa.gov/so2-pollution/sulfur-dioxide-basics#what is so2. View
Kumar Jha, D., Sabesan, M., Das, A., Vinithkumar, N. V, & Kirubagaran, R. (2011). Evaluation of Interpolation Technique for Air Quality Parameters in Port Blair, India. Universal Journal of Environmental Research and Technology, 1(3), 301– 310. Retrieved from http://www.environmentaljournal.org/1-3/ ujert-1-3-10.pdfView
Thurston, G. D. (2017). Outdoor Air Pollution: Sources, Atmospheric Transport, and Human Health Effects. In International Encyclopedia of Public Health (Second Edition) (Second Edi, Vol. 5).https://doi.org/10.1016/B978-0-12- 803678-5.003209View
Pohl, H., Liccione, J., & Iannucci, A. (2002). Toxicological Profile for Sulfur Dioxide. In Agency for Toxic Substances and Disease Registry’s Toxicological Profiles.https://doi. org/10.1201/9781420061888_ch144
DINPS. (2018). Sulfur Dioxide Effects on Health. Retrieved from U.S. Department of the Interior National Park Service website: https://www.nps.gov/subjects/air/humanhealth-sulfur. htmView
Spetz, A. L. (2014). SiC-FET based SO2 sensor for power plant emission applications. Sensors and Actuators, B: Chemical, 194(194), 511–520. https://doi.org/10.1016/j.snb.2013.11.089View
Goyal, S. K. (2001). Use of rosaniline hydrochloride dye for atmospheric SO2 determination and method sensitivity analysis. Journal of Environmental Monitoring, 3(6), 666–670. https://doi.org/10.1039/b106209nView
Pate, J. B., Lodge, J. P., & Wartburg, A. F. (1962). Effect of Pararosaniline in the Trace Determination of Sulfur Dioxide. Scientific Communications, 34(12), 1660–1662.View
Warner, I. M., El-Zahab, B., & Siraj, N. (2014). Perspectives on moving ionic liquid chemistry into the solid phase. Analytical Chemistry, 86(15), 7184–7191. https://doi.org/10.1021/ ac501529mView
McNeel, K. E., Siraj, N., Bhattarai, N., & Warner, I. M. (2019). Fluorescence-Based Ratiometric Nanosensor for Selective Imaging of Cancer Cells. ACS Omega, 4(1), 1592–1600. https:// doi.org/10.1021/acsomega.8b02765View
Berton, P., Siraj, N., Das, S., de Rooy, S., Wuilloud, R. G., & Warner, I. M. (2020). Efficient Low-Cost Procedure for Microextraction of Estrogen from Environmental Water Using Magnetic Ionic Liquids. Molecules (Basel, Switzerland), 26(1), 1–12. https://doi.org/10.3390/molecules26010032View
Galpothdeniya, W. I. S., Regmi, B. P., McCarter, K. S., De Rooy, S. L., Siraj, N., & Warner, I. M.(2015). Virtual colorimetric sensor array: Single ionic liquid for solvent discrimination. Analytical Chemistry, 87(8), 4464–4471. https://doi.org/10.1021/acs. analchem.5b00714View
Al Ghafly, H., Siraj, N., Das, S., Regmi, B. P., Magut, P. K. S., Galpothdeniya, W. I. S., … Warner, I.M. (2014). GUMBOS matrices of variable hydrophobicity for matrix-assisted laser desorption/ionization mass spectrometry. Rapid Communications in Mass Spectrometry, 28(21),2307–2314. https://doi.org/10.1002/rcm.7027View
Kolic, P. E., Siraj, N., Cong, M., Regmi, B. P., Luan, X., Wang, Y., & Warner, I. M. (2016). Improving energy relay dyes for dye-sensitized solar cells by use of a group of uniform materials based on organic salts (GUMBOS). RSC Advances, 6(97), 95273–95282. https://doi.org/10.1039/c6ra21980bView
Siraj, N., Hasan, F., Das, S., Kiruri, L. W., Steege Gall, K. E., Baker, G. A., & Warner, I. M. (2014).Carbazole-derived group of uniform materials based on organic salts: Solid state fluorescent analogues of ionic liquids for potential applications in organic-based blue light-emitting diodes.Journal of Physical Chemistry C, 118(5), 2312–2320. https://doi.org/10.1021/jp410784vView
Bhattarai, N., Mathis, J. M., Chen, M., Perez, R. L., Siraj, N., Magut, P. K. S., … Warner, I. M. (2018).Endocytic Selective Toxicity of Rhodamine 6G nanoGUMBOS in Breast Cancer Cells. Molecular Pharmaceutics, 15(9), 3837–3845. https://doi. org/10.1021/acs.molpharmaceut.8b00339View
Hanson, K., Roskop, L., Djurovich, P. I., Zahariev, F., Gordon, M. S., & Thompson, M. E. (2010). A paradigm for blue- or red-shifted absorption of small molecules depending on the site of π-extension. Journal of the American Chemical Society, 132(45), 16247–16255.https://doi.org/10.1021/ja1075162View
Gayton, J. N., Autry, S., Fortenberry, R. C., Hammer, N. I., & Delcamp, J. H. (2018). Counter Anion Effect on the Photophysical Properties of Emissive Indolizine-Cyanine Dyes in Solution and Solid State. Molecules, 23(12). https://doi. org/10.3390/molecules23123051View
Jordan, A. N., Das, S., Siraj, N., De Rooy, S. L., Li, M., El-Zahab, B., … Warner, I. M. (2012). Anioncontrolled morphologies and spectral features of cyanine-based nanoGUMBOS - An improved photosensitizer. Nanoscale, 4(16), 5031–5038. https://doi.org/10.1039/c2nr30432eView
Haber, L. H., Karam, T. E., Siraj, N., Ranasinghe, J. C., Kolic, P. E., Regmi, B. P., & Warner, I. M.(2020). Efficient photoinduced energy transfer in porphyrin-based nanomaterials. Journal of Physical Chemistry C, 124(44), 24533–24541. https://doi. org/10.1021/acs.jpcc.0c08985View
Jeevanandam, J., Barhoum, A., Chan, Y. S., Dufresne, A., & Danquah, M. K. (2018). Review on nanoparticles and nanostructured materials: History, sources, toxicity and regulations. Beilstein Journal of Nanotechnology, 9(1), 1050– 1074. https://doi.org/10.3762/bjnano.9.98View
Jordan, A. N., Siraj, N., Das, S., & Warner, I. M. (2014). Tunable near-infrared emission of binary nanoand mesoscale GUMBOS. RSC Advances, 4(54), 28471–28480. https://doi. org/10.1039/c4ra03256jView
Karam, T. E., Siraj, N., Warner, I. M., & Haber, L. H. (2015). Anomalous Size-Dependent Excited-State Relaxation Dynamics of NanoGUMBOS. Journal of Physical Chemistry C, 119(50), 28206–28213.https://doi.org/10.1021/acs. jpcc.5b09729View
Hung, T.-F., & Liu, R.-S. (2012). Chapter 1: An Introduction of Controlled Sizes and Shapes of Nanostructured Materials and Their Applications. In R.-S. Liu (Ed.), Controlled Nanofabrication: Advances and Applications. https://doi. org/10.4032/9789814364515
R.Kumar, G. Sharma, and M. K. (2013). Effect of size and shape on the vibrational and thermodynamic properties of nanomaterials. Journal of Thermodynamics, 1(1), 1–5.https:// doi.org/10.1155/2013/328051View
Zamiri, R., Zakaria, A., Ahangar, H. A., Darroudi, M., Zak, A. K., & Drummen, G. P. C. (2012). Aqueous starch as a stabilizer in zinc oxide nanoparticle synthesis via laser ablation. Journal of Alloys and Compounds, 516, 41–48. https://doi. org/10.1016/j.jallcom.2011.11.118View
Simon-Deckers, A., Loo, S., Mayne-L’Hermite, M., Herlin-Boime, N., Menguy, N., Reynaud, C., … Carriere, M. (2009). Size-, composition- and shape-dependent toxicological impact of metal oxide nanoparticles and carbon nanotubes toward bacteria. Environmental Science and Technology,43(21), 8423– 8429. https://doi.org/10.1021/es9016975View
Yuan, Z., Li, R., Meng, F., Zhang, J., Zuo, K., & Han, E. (2019). Approaches to enhancing gas sensing properties: A review. Sensors (Switzerland), 19(7). https://doi.org/10.3390/s19071495View
Charitidis, C. A., Georgiou, P., Koklioti, M. A., Trompeta, A. F., & Markakis, V. (2014). Manufacturing nanomaterials: From research to industry.Manufacturing Review, 1(11), 19.View
Kolic, P. E., Siraj, N., Hamdan, S., Regmi, B. P., & Warner, I. M. (2016). Synthesis and Characterization of Porphyrin-Based GUMBOS and NanoGUMBOS as Improved Photosensitizers. Journal of Physical Chemistry C, 120(9), 5155–5163.https:// doi.org/10.1021/acs.jpcc.5b12013View